Glycerol: A Natural Moisturizer!
Glycerol, sometimes referred to as glycerin, is a sugar alcohol used in a variety of personal care products, including toothpaste, hair conditioner, cosmetics, and moisturizers.
It is a colorless, odorless, viscous liquid that is sweet-tasting and non-toxic. The glycerol backbone is found in all lipids known as triglycerides. It is widely used in the food industry as a sweetener and humectant and in pharmaceutical formulations. Glycerol has three hydroxyl groups that are responsible for its solubility in water.
What does Glycerol look like in Chemistry?
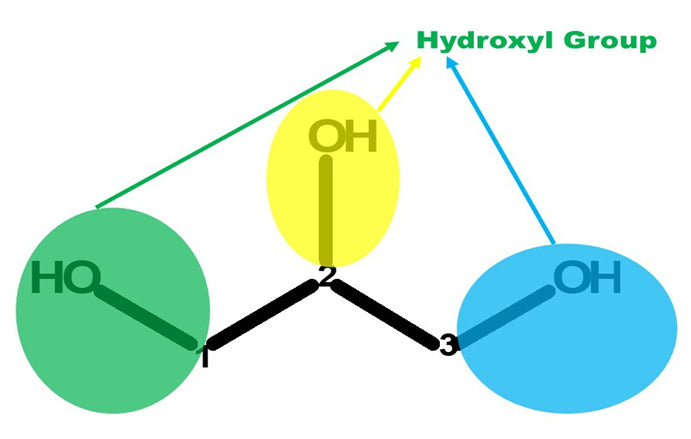
Let’s Get Building!
Using your Student Molecular Set from Duluth Labs let’s create Benzoic Acid! You’ll need:
-
3 Carbon Atoms
-
3 Oxygen atoms
-
8 Hydrogen atoms
-
8Small connectors (compact small bonds for hydrogen)
-
5 Medium Connectors
-
Molecular Tool (for Disassembly)
Put aside all the atoms and connectors needed.
Let’s Start Building With Our First Hydroxyl Group.
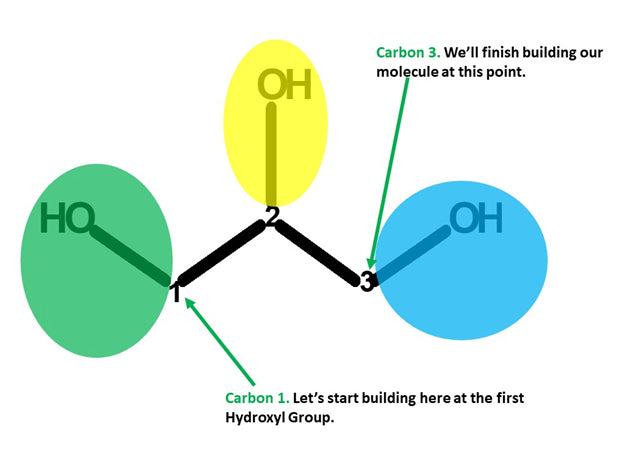
Note: We build this portion in a clockwise direction, starting with Carbon 1 and end with Carbon 3.
Let’s start!
Steps:
-
1

1. Get a carbon atom (Carbon 1) then attach two Hydrogen atoms to Carbon 1using two small connectors. Then use a medium connector and small connector to attach a hydroxyl group.
-
2

2. Get another carbon atom (Carbon 2)then attach this toCarbon 1using a medium connector. Then, using a small connector attach a Hydrogen atom below. The using a medium connector and a short connector attach a hydroxyl group to Carbon 2.
-
3
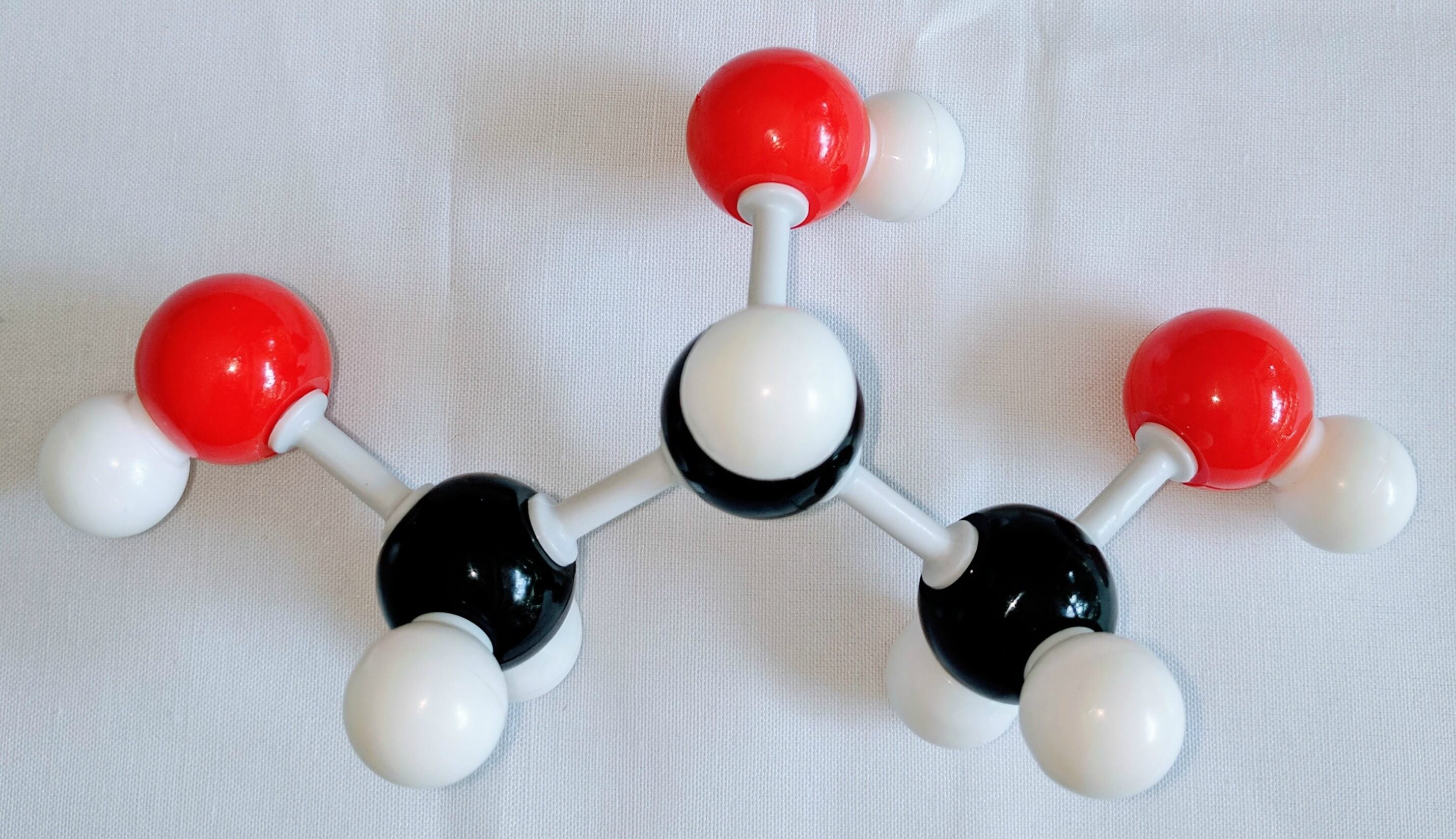
3. Using another carbon atom Carbon 3and attach it to Carbon 2using a medium connector. Then, place two Hydrogen atoms on Carbon 3using two small connectors.Finally, use a medium connector attach an Oxygen atom to Carbon 3. Place a Hydrogen atom on the Oxygen atom.










https://emcmee.blogspot.com/2022/08/transfer-furniture-jeddah.html
https://www.khedmeh.com/wall/blogs/post/12991
https://www.khedmeh.com/wall/blogs/12905
https://www.b-webdesign.org/dir-wowonder/read-blog/95246
https://www.b-webdesign.org/dir-wowonder/read-blog/95225
https://joyrulez.com/blogs/168166/%D8%B4%D8%B1%D9%83%D8%A7%D8%AA-%D9%86%D9%82%D9%84-%D8%A7%D9%84%D8%B9%D9%81%D8%B4-%D8%A8%D8%AC%D8%AF%D8%A9
https://joyrulez.com/blogs/168162/%D8%B4%D8%B1%D9%83%D8%A7%D8%AA-%D9%84%D9%86%D9%82%D9%84-%D8%A7%D9%84%D8%B9%D9%81%D8%B4-%D9%81%D9%8A-%D8%AC%D8%AF%D8%A9
https://demo.sngine.com/blogs/102320/%D9%86%D9%82%D9%84-%D8%B9%D9%81%D8%B4-%D8%A8%D8%AC%D8%AF%D8%A9
https://demo.sngine.com/blogs/101980/%D8%A7%D9%81%D8%B6%D9%84-%D8%B4%D8%B1%D9%83%D8%A9-%D9%86%D9%82%D9%84-%D8%B9%D9%81%D8%B4-%D8%A8%D8%AC%D8%AF%D8%A9-emc-mee-com-transfer-furniture
https://emc-mee.page.tl/%26%231575%3B%26%231585%3B%26%231602%3B%26%231575%3B%26%231605%3B-%26%231588%3B%26%231585%3B%26%231603%3B%26%231575%3B%26%231578%3B-%26%231606%3B%26%231602%3B%26%231604%3B-%26%231593%3B%26%231601%3B%26%231588%3B-%26%231576%3B%26%231580%3B%26%231583%3B%26%231577%3B.htm
https://ataralmadinah662300791.wordpress.com/2022/09/05/%d8%b4%d8%b1%d9%83%d8%a9-%d9%86%d9%82%d9%84-%d8%b9%d9%81%d8%b4-%d9%85%d9%86-%d8%ac%d8%af%d8%a9-%d8%a7%d9%84%d9%89-%d8%aa%d8%a8%d9%88%d9%83-%d9%85%d8%b9-%d8%a7%d9%84%d8%aa%d8%ba%d9%84%d9%8a%d9%81/
https://atar-almadinah.over-blog.com/2022/09/transfer-furniture-jeddah-tabuk.html
https://eslamiatview.blogspot.com/2022/09/transfer-furniture-.html
https://is.gd/bJ2Dmy
https://is.gd/ypnMEL
https://is.gd/BLejy1
https://is.gd/nHp6N4
https://is.gd/BtLzld
https://is.gd/zxECuM
https://is.gd/i4mxfM
https://is.gd/DsIPbe
https://cutt.ly/tCzFRi0
https://cutt.ly/ECzF3dA
https://cutt.ly/oCzGwW6
https://is.gd/GfoG2V
https://is.gd/mX0WTb
https://cutt.ly/ACzGRPF
https://is.gd/SLAkcT
https://cutt.ly/TCzGPlc
https://rb.gy/doq4qv
https://rb.gy/f6lina
https://rb.gy/lfqg9r
https://rb.gy/nrko3x
https://rb.gy/xhuggi
https://rb.gy/lbp0ea
https://rb.gy/wsgbj0
https://rb.gy/eumloo
https://rb.gy/foooha
https://rb.gy/po1lxc
https://rb.gy/fqpdsz
https://is.gd/9SdaEc
https://cutt.ly/qCzJbUt
https://www.vingle.net/posts/4729609
https://www.vingle.net/posts/4729610
https://rb.gy/eimwki
https://rb.gy/ionvkh
https://cutt.ly/7CzZUSh
https://is.gd/DEo87A
https://cutt.ly/eCzZAiQ
https://is.gd/rUreg8
https://hackmd.io/moversjeddah/By88RRJxs https://jeddah.peatix.com/ https://unsplash.com/moversjeddah
https://moversriyadhcom.wordpress.com/2022/09/09/%d8%b4%d8%b1%d9%83%d8%a7%d8%aa-%d9%86%d9%82%d9%84-%d8%b9%d9%81%d8%b4-%d8%a8%d9%85%d9%83%d8%a9/
https://moversriyadhcom.wordpress.com/2022/09/09/%d8%a7%d9%81%d8%b6%d9%84-%d8%b4%d8%b1%d9%83%d8%a9-%d9%86%d9%82%d9%84-%d8%b9%d9%81%d8%b4-%d8%a8%d8%a7%d9%84%d8%a7%d8%ad%d8%b3%d8%a7%d8%a1/
https://moversriyadhcom.wordpress.com/2022/09/10/%d8%a7%d9%81%d8%b6%d9%84-%d8%b4%d8%b1%d9%83%d8%a9-%d9%86%d9%82%d9%84-%d8%b9%d9%81%d8%b4-%d8%a8%d8%a7%d9%84%d8%ae%d8%a8%d8%b1/
https://moversriyadhcom.wordpress.com/2022/09/10/%d8%a7%d9%81%d8%b6%d9%84-%d8%b4%d8%b1%d9%83%d8%a9-%d9%86%d9%82%d9%84-%d8%b9%d9%81%d8%b4-%d8%a8%d8%ac%d8%af%d8%a9/
https://moversfurniture.over-blog.com/transfer-jeddah.html
https://moversfurniture.over-blog.com/movers-furniture-jeddah.html
https://hackmd.io/@jeddahmovers/rJMNqojxj
https://www.skreebee.com/read-blog/115087
https://oktob.io/posts/43829
https://telegra.ph/%D8%B4%D8%B1%D9%83%D8%A9-%D9%86%D9%82%D9%84-%D8%B9%D9%81%D8%B4-%D9%85%D9%86-%D8%AC%D8%AF%D8%A9-%D8%A7%D9%84%D9%89-%D8%AA%D8%A8%D9%88%D9%83-09-12
https://ataralmadinah662300791.wordpress.com/2022/09/12/%d8%a7%d8%b1%d9%82%d8%a7%d9%85-%d8%b4%d8%b1%d9%83%d8%a7%d8%aa-%d9%86%d9%82%d9%84-%d8%a7%d9%84%d8%b9%d9%81%d8%b4-%d8%a8%d8%ac%d8%af%d8%a9/
https://rb.gy/rl7jzc
https://cutt.ly/NCZt6Jm
https://is.gd/A89d52
https://atar-almadinah.over-blog.com/numbers-company-transfer-furniture.html
https://oktob.io/posts/43903
https://telegra.ph/%D8%A7%D8%B1%D9%82%D8%A7%D9%85-%D8%B4%D8%B1%D9%83%D8%A7%D8%AA-%D9%86%D9%82%D9%84-%D8%B9%D9%81%D8%B4-%D9%88%D8%A7%D8%AB%D8%A7%D8%AB-%D8%A8%D8%AC%D8%AF%D8%A9-09-12
https://www.skreebee.com/read-blog/115193
https://hackmd.io/jeddahmovers/B1Is9m6li https://hackmd.io/jeddahmovers/B1Ux7Vpxj
https://hackmd.io/@jeddahmovers/S1g3xLaxj
https://addandclick.com/read-blog/94597
https://webyourself.eu/blogs/36223/%D8%A7%D8%B1%D9%82%D8%A7%D9%85-%D8%B4%D8%B1%D9%83%D8%A7%D8%AA-%D9%86%D9%82%D9%84-%D8%B9%D9%81%D8%B4-%D9%88%D8%A7%D8%AB%D8%A7%D8%AB-%D8%A8%D8%AC%D8%AF%D8%A9
https://moversriyadhcom.wordpress.com/2022/09/13/%d9%90%d8%b4%d8%b1%d9%83%d8%a9-%d9%86%d9%82%d9%84-%d8%b9%d9%81%d8%b4-%d8%a8%d8%ac%d8%a7%d8%b2%d8%a7%d9%86/
https://www.scoop.it/topic/rcutioni/p/4135811249/2022/09/19/25-0545970806-65
https://www.scoop.it/topic/rcutioni/p/4135811200/2022/09/19/0500175959-55
https://www.scoop.it/topic/rcutioni/p/4135809640/2022/09/19/0500175959-45
https://www.scoop.it/topic/rcutioni/p/4135810312/2022/09/19/0500175959-50
https://ko-fi.com/jumperads49641
https://hackmd.io/jumperads/Syzi0PCei https://hackmd.io/jumperads/B1Z2f_Cgj
https://khairyayman85.wixsite.com/jumperads
https://allmoversinriyadh.wordpress.com/2022/09/15/%d8%a7%d9%81%d8%b6%d9%84-%d8%b4%d8%b1%d9%83%d8%a9-%d8%aa%d9%86%d8%b8%d9%8a%d9%81-%d8%a8%d9%8a%d9%86%d8%a8%d8%b9/
https://allmoversinriyadh.wordpress.com/2022/09/14/%d8%a7%d9%81%d8%b6%d9%84-%d8%b4%d8%b1%d9%83%d8%a9-%d8%aa%d9%86%d8%b8%d9%8a%d9%81-%d8%a8%d8%ae%d9%85%d9%8a%d8%b3-%d9%85%d8%b4%d9%8a%d8%b7/
https://allmoversinriyadh.wordpress.com/2022/09/14/%d8%a7%d9%81%d8%b6%d9%84-%d8%b4%d8%b1%d9%83%d8%a9-%d8%aa%d9%86%d8%b8%d9%8a%d9%81-%d8%a8%d8%a7%d9%84%d8%b1%d9%8a%d8%a7%d8%b6-2/
https://allmoversinriyadh.wordpress.com/2022/04/07/%d8%a7%d9%81%d8%b6%d9%84-%d8%b4%d8%b1%d9%83%d8%a9-%d8%aa%d9%86%d8%b8%d9%8a%d9%81-%d8%a8%d8%a7%d9%84%d8%b1%d9%8a%d8%a7%d8%b6/
https://www.atar-almadinah.com/%D8%B4%D8%B1%D9%83%D8%A9-%D9%86%D9%82%D9%84-%D8%B9%D9%81%D8%B4-%D8%A8%D8%AC%D8%AF%D8%A9-%D8%B9%D9%85%D8%A7%D9%84%D8%A9-%D9%81%D9%84%D8%A8%D9%8A%D9%86%D9%8A%D8%A9.html
https://www.atar-almadinah.com/%D8%B4%D8%B1%D9%83%D8%A9-%D9%86%D9%82%D9%84-%D8%B9%D9%81%D8%B4-%D8%AC%D8%AF%D8%A9-%D8%A7%D8%A8%D8%AD%D8%B1-%D8%A7%D9%84%D8%B4%D9%85%D8%A7%D9%84%D9%8A%D8%A9.html
https://ataralmadinah662300791.wordpress.com/2022/09/20/%d8%af%d9%84%d9%8a%d9%84-%d8%b4%d8%b1%d9%83%d8%a7%d8%aa-%d9%86%d9%82%d9%84-%d8%a7%d9%84%d8%b9%d9%81%d8%b4-%d8%a8%d8%ac%d8%af%d8%a9/
https://atar-almadinah.over-blog.com/guide-to-moving-furniture-companies-in-jeddah.html
https://ataralmadinah662300791.wordpress.com/2022/09/20/%d8%b4%d8%b1%d9%83%d8%a9-%d9%86%d9%82%d9%84-%d8%b9%d9%81%d8%b4-%d8%a8%d8%ac%d8%af%d8%a9-%d8%b9%d9%85%d8%a7%d9%84%d8%a9-%d9%81%d9%84%d8%a8%d9%8a%d9%86%d9%8a%d8%a9/
https://atar-almadinah.over-blog.com/furniture-moving-company-in-jeddah-filipino.html
https://ataralmadinah662300791.wordpress.com/2022/09/23/%d8%a7%d9%81%d8%b6%d9%84-%d8%b4%d8%b1%d9%83%d8%a9-%d9%86%d9%82%d9%84-%d8%b9%d9%81%d8%b4-%d8%a8%d9%85%d9%83%d8%a9/
https://allmoversinriyadh.wordpress.com/2022/09/24/%d8%a7%d9%81%d8%b6%d9%84-%d8%b4%d8%b1%d9%83%d8%a9-%d8%aa%d9%86%d8%b8%d9%8a%d9%81-%d8%ae%d8%b2%d8%a7%d9%86%d8%a7%d8%aa-%d8%a8%d8%a7%d9%84%d8%ac%d8%a8%d9%8a%d9%84/
https://transferefurniture.hatenablog.com/entry/2022/09/25/054311
https://justpaste.it/99zuq
https://allmoversinriyadh.wordpress.com/2022/09/25/%d8%a7%d9%81%d8%b6%d9%84-%d8%b4%d8%b1%d9%83%d8%a9-%d9%86%d9%82%d9%84-%d8%b9%d9%81%d8%b4-%d8%a8%d8%a7%d9%84%d8%ac%d8%a8%d9%8a%d9%84/
https://anotepad.com/note/read/qrdb39sk
https://anotepad.com/note/read/mhenxycs
https://anotepad.com/note/read/3tpk27dm
https://anotepad.com/note/read/g8hggmdp
https://anotepad.com/note/read/a4thkdsf
https://slashdot.org/submission/16646847/the-best-furniture-moving-company-in-jeddah
https://www.reddit.com/user/MinimumLong6251/comments/xo1edo/%D8%B4%D8%B1%D9%83%D8%A9_%D9%86%D9%82%D9%84_%D8%B9%D9%81%D8%B4_%D8%A8%D8%AC%D8%AF%D8%A9_%D8%B9%D9%85%D8%A7%D9%84%D8%A9_%D9%81%D9%84%D8%A8%D9%8A%D9%86%D9%8A%D8%A9/
https://www.reddit.com/user/MinimumLong6251/
https://www.crokes.com/ataralmadinah/info/
https://companyhouseservice.wordpress.com/2022/09/27/%d8%b4%d8%b1%d9%83%d8%a9-%d8%aa%d9%86%d8%b8%d9%8a%d9%81-%d8%a8%d8%a7%d9%84%d8%ac%d8%a8%d9%8a%d9%84/
https://www.evernote.com/shard/s588/client/snv?noteGuid=5f0e6065-a56d-cc5c-1335-9270d70deb11¬eKey=f04bcc20e270300aefd136ec88db4377&sn=https%3A%2F%2Fwww.evernote.com%2Fshard%2Fs588%2Fsh%2F5f0e6065-a56d-cc5c-1335-9270d70deb11%2Ff04bcc20e270300aefd136ec88db4377&title=The%2Bbest%2Bfurniture%2Bmoving%2Bcompany%2Bin%2BJeddah
https://www.pearltrees.com/moverscompany
http://www.pearltrees.com/moverscompany/item471149920
https://www.instapaper.com/p/11269590
https://zlidein.com/read-blog/97062
https://zlidein.com/read-blog/97063
https://zlidein.com/read-blog/97071
https://zlidein.com/read-blog/99326
https://www.skreebee.com/read-blog/110085
https://telegra.ph/%D8%B4%D8%B1%D9%83%D8%A9-%D9%86%D9%82%D9%84-%D8%B9%D9%81%D8%B4-%D9%85%D9%86-%D8%AC%D8%AF%D8%A9-%D8%A7%D9%84%D9%89-%D8%AC%D8%A7%D8%B2%D8%A7%D9%86-07-12
https://webyourself.eu/blogs/25649/%D8%B4%D8%B1%D9%83%D8%A9-%D9%86%D9%82%D9%84-%D8%B9%D9%81%D8%B4-%D9%85%D9%86-%D8%AC%D8%AF%D8%A9-%D8%A7%D9%84%D9%89-%D8%AC%D8%A7%D8%B2%D8%A7%D9%86
https://www.merexpression.com/read-blog/21528
https://www.rolonet.com/blogs/89146/%D8%B4%D8%B1%D9%83%D8%A9-%D9%86%D9%82%D9%84-%D8%B9%D9%81%D8%B4-%D9%85%D9%86-%D8%AC%D8%AF%D8%A9-%D8%A7%D9%84%D9%89-%D8%AC%D8%A7%D8%B2%D8%A7%D9%86
https://oktob.io/posts/41814
https://webyourself.eu/blogs/26016/%D8%A7%D9%81%D8%B6%D9%84-%D8%B4%D8%B1%D9%83%D8%A9-%D9%86%D9%82%D9%84-%D8%B9%D9%81%D8%B4-%D8%AC%D8%AF%D8%A9-%D8%A7%D8%A8%D8%AD%D8%B1-%D8%A7%D9%84%D8%B4%D9%85%D8%A7%D9%84%D9%8A%D8%A9
https://oktob.io/posts/41908
https://www.domyate.com/2022/06/14/why-emc-mee-company-to-transfer-furniture-to-to-jeddah/
https://www.domyate.com/2022/06/12/best-nakl-afsh-jeddah/
https://www.domyate.com/2022/06/12/%d9%83%d9%8a%d9%81%d9%8a%d8%a9-%d9%81%d9%83-%d9%88%d8%aa%d8%b1%d9%83%d9%8a%d8%a8-%d8%a7%d9%84%d8%b9%d9%81%d8%b4-%d9%88%d8%a7%d9%84%d8%a7%d8%ab%d8%a7%d8%ab-%d8%a8%d8%ac%d8%af%d8%a9/
https://zlidein.com/read-blog/99328
https://telegra.ph/%D8%A7%D9%81%D8%B6%D9%84-%D8%B4%D8%B1%D9%83%D8%A9-%D8%AA%D9%86%D8%B8%D9%8A%D9%81-%D9%85%D9%83%D9%8A%D9%81%D8%A7%D8%AA-%D8%A8%D9%85%D9%83%D8%A9-07-17
http://html.net.linux17.wannafindserver.dk/forums/viewtopic.php?f=6&t=447291&sid=d49bfa79507d1da5e63e18cadb51c609
https://www.carookee.de/forum/Retinoblastom-Forum/32167082?mp=49083704862d5f02883b054f8275606a980b0ddf6bde4bc1a0c4b5&mps=%26%231588%3B%26%231585%3B%26%231603%3B%26%231577%3B+%26%231606%3B%26%231602%3B%26%231604%3B+%26%231593%3B%26%231601%3B%26%231588%3B+%26%231575%3B%26%231604%3B%26%231589%3B%26%231602%3B%26%231585%3B+%26%231575%3B%26%231604%3B%26%231583%3B%26%231608%3B%26%231604%3B%26%231610%3B#32167082
http://html.net.linux17.wannafindserver.dk/forums/viewtopic.php?f=5&t=447319&sid=02b7bb5d454a63d0454daad5ff0e8fa3
https://www.carookee.de/forum/Retinoblastom-Forum/32167083?mp=156822404062d6023635638289af31b90d4f5bbea03aea79dc802ca&mps=%26%231588%3B%26%231585%3B%26%231603%3B%26%231577%3B+%26%231578%3B%26%231606%3B%26%231592%3B%26%231610%3B%26%231601%3B+%26%231605%3B%26%231603%3B%26%231610%3B%26%231601%3B%26%231575%3B%26%231578%3B+%26%231576%3B%26%231605%3B%26%231603%3B%26%231577%3B#32167083
https://www.carookee.de/forum/Retinoblastom-Forum/32167084?mp=156822404062d604da7583a085d6df59451e00fbf675ffe4d767997&mps=%26%231588%3B%26%231585%3B%26%231603%3B%26%231577%3B+%26%231578%3B%26%231606%3B%26%231592%3B%26%231610%3B%26%231601%3B+%26%231582%3B%26%231586%3B%26%231575%3B%26%231606%3B%26%231575%3B%26%231578%3B+%26%231576%3B%26%231582%3B%26%231605%3B%26%231610%3B%26%231587%3B+%26%231605%3B%26%231588%3B%26%231610%3B%26%231591%3B#32167084
https://www.carookee.de/forum/Retinoblastom-Forum/32167085?mp=156822404062d60ab56d77ab711e09fefce9f81dfa2064fdad6969e&mps=%26%231588%3B%26%231585%3B%26%231603%3B%26%231577%3B+%26%231606%3B%26%231602%3B%26%231604%3B+%26%231593%3B%26%231601%3B%26%231588%3B+%26%231605%3B%26%231606%3B+%26%231580%3B%26%231583%3B%26%231577%3B+%26%231575%3B%26%231604%3B%26%231609%3B+%26%231580%3B%26%231575%3B%26%231586%3B%26%231575%3B%26%231606%3B#32167085
https://elasakr-jeddah.jimdosite.com/
https://business.go.tz/web/rashid.ndimbo/~/86020/home/-/message_boards/message/24415721
https://business.go.tz/web/rashid.ndimbo/~/86020/home/-/message_boards/message/24415731
https://business.go.tz/web/rashid.ndimbo/~/86020/home/-/message_boards/message/24458120
https://business.go.tz/web/rashid.ndimbo/~/86020/home/-/message_boards/message/24456486
https://bestmoversfurniture.wordpress.com/2022/04/05/transfer-furniture-jeddah/
https://www.smore.com/ps2zt
https://www.smore.com/s9rz8q
https://www.smore.com/0kthj
https://www.muratshriners.com/profile/elsakrjeddah/profile
https://www.medmotion.org/profile/elsakrjeddah/profile
https://ello.co/atar-almadinah
https://ello.co/atar-almadinah/post/9biwwplb4odbgeboq8xcgw
https://nowewyrazy.uw.edu.pl/profil/atar-almadinah
http://findfriendz.com/profile/173148.html
https://companyhouseservice.wordpress.com/2022/08/06/%d8%a7%d9%81%d8%b6%d9%84-%d8%b4%d8%b1%d9%83%d8%a9-%d8%aa%d9%86%d8%b8%d9%8a%d9%81-%d8%a8%d8%a7%d9%84%d8%b1%d9%8a%d8%a7%d8%b6/
https://www.edocr.com/v/d9xod4ar/atar-almadinah/shrka-nkl-aafsh-bgda
https://www.edocr.com/user/atar-almadinah
https://www.tuugo.us/Companies/atar-almadinah/0310006888242
https://www.tuugo.us/SiteViewer/0310006888242
https://topsitenet.com/startpage/ataralmadinah/859508/
https://topsitenet.com/profile/ataralmadinah/859508/
https://en.gravatar.com/elsakrjeddah
https://610b31f1e425e.site123.me/about
https://www.kickstarter.com/profile/atar-almadinah/about
WhiskeyPeak77@gmail.com
12/22/22
https://casinobulk.com/
https://casinobulk.com/ 카지노사이트
https://casinobulk.com/ 바카라사이트
https://casinobulk.com/ 온라인카지노
https://casinobulk.com/ 온라인바카라
https://casinobulk.com/ 온라인슬롯사이트
https://casinobulk.com/ 카지노사이트게임
https://casinobulk.com/ 카지노사이트검증
https://casinobulk.com/ 카지노사이트추천
https://casinobulk.com/ 안전카지노사이트
https://casinobulk.com/ 안전카지노사이트도메인
https://casinobulk.com/ 안전한 카지노사이트 추천
https://casinobulk.com/ 바카라사이트게임
https://casinobulk.com/ 바카라사이트검증
https://casinobulk.com/ 바카라사이트추천
https://casinobulk.com/ 안전바카라사이트
https://casinobulk.com/ 안전바카라사이트도
https://casinobulk.com/ 안전한 바카라사이트
http://toolbarqueries.google.com.uy/url?sa=t&url=https://casinobulk.com/
http://toolbarqueries.google.com.tw/url?sa=t&url=https://casinobulk.com/
http://toolbarqueries.google.com.tr/url?sa=t&url=https://casinobulk.com/
http://toolbarqueries.google.com.sa/url?sa=t&url=https://casinobulk.com/
http://toolbarqueries.google.com.py/url?sa=t&url=https://casinobulk.com/
http://toolbarqueries.google.com.pr/url?sa=t&url=https://casinobulk.com/
http://toolbarqueries.google.com.pk/url?sa=t&url=https://casinobulk.com/
http://toolbarqueries.google.com.pe/url?sa=t&url=https://casinobulk.com/
http://toolbarqueries.google.com.my/url?sa=t&url=https://casinobulk.com/
http://toolbarqueries.google.com.hk/url?sa=t&url=https://casinobulk.com/
http://toolbarqueries.google.com.gt/url?sa=t&url=https://casinobulk.com/
http://toolbarqueries.google.com.gh/url?sa=t&url=https://casinobulk.com/
https://clients1.google.com.ar/url?sa=t&url=https://casinobulk.com/
https://clients1.google.com.ag/url?sa=t&url=https://casinobulk.com/
https://clients1.google.co.zm/url?sa=t&url=https://casinobulk.com/
https://clients1.google.co.za/url?sa=t&url=https://casinobulk.com/
https://clients1.google.co.ve/url?sa=t&url=https://casinobulk.com/
https://clients1.google.co.uz/url?sa=t&url=https://casinobulk.com/
https://clients1.google.co.ug/url?sa=t&url=https://casinobulk.com/
https://clients1.google.co.th/url?sa=t&url=https://casinobulk.com/
https://clients1.google.co.nz/url?sa=t&url=https://casinobulk.com/
https://clients1.google.co.kr/url?sa=t&url=https://casinobulk.com/
https://clients1.google.co.ke/url?sa=t&url=https://casinobulk.com/
https://clients1.google.co.il/url?sa=t&url=https://casinobulk.com/
https://clients1.google.co.id/url?sa=t&url=https://casinobulk.com/
https://clients1.google.co.cr/url?sa=t&url=https://casinobulk.com/
https://clients1.google.co.ck/url?sa=t&url=https://casinobulk.com/
https://cse.google.co.ck/url?sa=t&url=https://casinobulk.com/
https://cse.google.co.bw/url?sa=t&url=https://casinobulk.com/
https://cse.google.cm/url?sa=t&url=https://casinobulk.com/
https://cse.google.cl/url?sa=t&url=https://casinobulk.com/
https://cse.google.ci/url?sa=t&url=https://casinobulk.com/
https://cse.google.ch/url?sa=t&url=https://casinobulk.com/
https://cse.google.ch/url?sa=i&url=https://casinobulk.com/
https://cse.google.cg/url?sa=t&url=https://casinobulk.com/
https://cse.google.cd/url?sa=t&url=https://casinobulk.com/
https://cse.google.by/url?sa=t&url=https://casinobulk.com/
https://cse.google.bs/url?sa=t&url=https://casinobulk.com/
https://cse.google.bi/url?sa=t&url=https://casinobulk.com/
https://cse.google.bg/url?sa=t&url=https://casinobulk.com/
https://cse.google.be/url?sa=t&url=https://casinobulk.com/
https://cse.google.be/url?sa=i&url=https://casinobulk.com/
https://cse.google.ba/url?sa=t&url=https://casinobulk.com/
https://cse.google.az/url?sa=t&url=https://casinobulk.com/
https://cse.google.at/url?sa=t&url=https://casinobulk.com/
https://images.google.ca/url?sa=t&url=https://blogskorcasino1.blogspot.com/2022/12/philippines-advises-pogos-to-settle.html
https://images.google.by/url?sa=t&url=https://blogskorcasino1.blogspot.com/2022/12/philippines-advises-pogos-to-settle.html
https://images.google.bs/url?sa=t&url=https://blogskorcasino1.blogspot.com/2022/12/philippines-advises-pogos-to-settle.html
https://images.google.bi/url?sa=t&url=https://blogskorcasino1.blogspot.com/2022/12/philippines-advises-pogos-to-settle.html
https://images.google.bg/url?sa=t&url=https://blogskorcasino1.blogspot.com/2022/12/philippines-advises-pogos-to-settle.html
https://images.google.bf/url?sa=t&url=https://blogskorcasino1.blogspot.com/2022/12/philippines-advises-pogos-to-settle.html
https://images.google.be/url?sa=t&url=https://blogskorcasino1.blogspot.com/2022/12/philippines-advises-pogos-to-settle.html
https://images.google.ba/url?sa=t&url=https://blogskorcasino1.blogspot.com/2022/12/philippines-advises-pogos-to-settle.html
https://images.google.ba/url?q=https://blogskorcasino1.blogspot.com/2022/12/philippines-advises-pogos-to-settle.html
https://images.google.az/url?sa=t&url=https://blogskorcasino1.blogspot.com/2022/12/philippines-advises-pogos-to-settle.html
https://images.google.at/url?sa=t&url=https://blogskorcasino1.blogspot.com/2022/12/philippines-advises-pogos-to-settle.html
https://images.google.as/url?sa=t&url=https://blogskorcasino1.blogspot.com/2022/12/philippines-advises-pogos-to-settle.html
https://images.google.am/url?sa=t&url=https://blogskorcasino1.blogspot.com/2022/12/philippines-advises-pogos-to-settle.html
https://images.google.al/url?sa=t&url=https://blogskorcasino1.blogspot.com/2022/12/philippines-advises-pogos-to-settle.html
https://images.google.ae/url?sa=t&url=https://blogskorcasino1.blogspot.com/2022/12/philippines-advises-pogos-to-settle.html
https://images.google.ae/url?q=https://blogskorcasino1.blogspot.com/2022/12/philippines-advises-pogos-to-settle.html
https://images.google.ad/url?sa=t&url=https://blogskorcasino1.blogspot.com/2022/12/philippines-advises-pogos-to-settle.html
https://maps.google.co.ke/url?sa=t&url=https://blogskorcasino1.blogspot.com/2022/12/philippines-advises-pogos-to-settle.html
https://maps.google.co.jp/url?sa=t&url=https://blogskorcasino1.blogspot.com/2022/12/philippines-advises-pogos-to-settle.html
https://maps.google.co.in/url?sa=t&url=https://blogskorcasino1.blogspot.com/2022/12/philippines-advises-pogos-to-settle.html
https://maps.google.co.il/url?sa=t&url=https://blogskorcasino1.blogspot.com/2022/12/philippines-advises-pogos-to-settle.html
https://maps.google.co.id/url?sa=t&url=https://blogskorcasino1.blogspot.com/2022/12/philippines-advises-pogos-to-settle.html
https://maps.google.co.cr/url?sa=t&url=https://blogskorcasino1.blogspot.com/2022/12/philippines-advises-pogos-to-settle.html
https://maps.google.co.bw/url?sa=t&url=https://blogskorcasino1.blogspot.com/2022/12/philippines-advises-pogos-to-settle.html
https://maps.google.cm/url?sa=t&url=https://blogskorcasino1.blogspot.com/2022/12/philippines-advises-pogos-to-settle.html
https://maps.google.cl/url?sa=t&url=https://blogskorcasino1.blogspot.com/2022/12/philippines-advises-pogos-to-settle.html
https://maps.google.ci/url?sa=t&url=https://blogskorcasino1.blogspot.com/2022/12/philippines-advises-pogos-to-settle.html
https://maps.google.ch/url?sa=t&url=https://blogskorcasino1.blogspot.com/2022/12/philippines-advises-pogos-to-settle.html
https://maps.google.cd/url?sa=t&url=https://blogskorcasino1.blogspot.com/2022/12/philippines-advises-pogos-to-settle.html
https://maps.google.cat/url?sa=t&url=https://blogskorcasino1.blogspot.com/2022/12/philippines-advises-pogos-to-settle.html
https://maps.google.ca/url?sa=t&url=https://blogskorcasino1.blogspot.com/2022/12/philippines-advises-pogos-to-settle.html
https://www.google.co.tz/url?sa=t&url=https://blogskorcasino1.blogspot.com/2022/12/philippines-advises-pogos-to-settle.html
https://www.google.co.th/url?sa=t&url=https://blogskorcasino1.blogspot.com/2022/12/philippines-advises-pogos-to-settle.html
https://www.google.co.nz/url?sa=t&url=https://blogskorcasino1.blogspot.com/2022/12/philippines-advises-pogos-to-settle.html
https://www.google.co.ma/url?sa=t&url=https://blogskorcasino1.blogspot.com/2022/12/philippines-advises-pogos-to-settle.html
https://www.google.co.ls/url?sa=t&url=https://blogskorcasino1.blogspot.com/2022/12/philippines-advises-pogos-to-settle.html
https://www.google.co.kr/url?sa=t&url=https://blogskorcasino1.blogspot.com/2022/12/philippines-advises-pogos-to-settle.html
https://www.google.co.ke/url?sa=t&url=https://blogskorcasino1.blogspot.com/2022/12/philippines-advises-pogos-to-settle.html
https://www.google.co.jp/url?sa=t&url=https://blogskorcasino1.blogspot.com/2022/12/philippines-advises-pogos-to-settle.html
https://www.google.co.in/url?sa=t&url=https://blogskorcasino1.blogspot.com/2022/12/philippines-advises-pogos-to-settle.html
https://www.google.co.il/url?sa=t&url=https://blogskorcasino1.blogspot.com/2022/12/philippines-advises-pogos-to-settle.html
https://www.google.co.id/url?sa=t&url=https://blogskorcasino1.blogspot.com/2022/12/philippines-advises-pogos-to-settle.html
https://www.google.co.cr/url?sa=t&url=https://blogskorcasino1.blogspot.com/2022/12/philippines-advises-pogos-to-settle.html
https://www.google.co.bw/url?sa=t&url=https://blogskorcasino1.blogspot.com/2022/12/philippines-advises-pogos-to-settle.html
https://www.google.cm/url?sa=t&url=https://blogskorcasino1.blogspot.com/2022/12/philippines-advises-pogos-to-settle.html
https://www.google.cl/url?sa=t&url=https://blogskorcasino1.blogspot.com/2022/12/philippines-advises-pogos-to-settle.html
https://www.google.ci/url?sa=t&url=https://blogskorcasino1.blogspot.com/2022/12/philippines-advises-pogos-to-settle.html
https://www.google.ch/url?sa=t&url=https://blogskorcasino1.blogspot.com/2022/12/philippines-advises-pogos-to-settle.html
https://www.google.cd/url?sa=t&url=https://blogskorcasino1.blogspot.com/2022/12/philippines-advises-pogos-to-settle.html
https://www.google.cat/url?sa=t&url=https://blogskorcasino1.blogspot.com/2022/12/philippines-advises-pogos-to-settle.html
https://www.google.ca/url?sa=t&url=https://blogskorcasino1.blogspot.com/2022/12/philippines-advises-pogos-to-settle.html
WhiskeyPeak77@gmail.com 11/28/22
https://opviewer.com/
http://www.erotikplatz.at/redirect.php?id=939&mode=fuhrer&url=https://opviewer.com
http://www.imsnet.at/LangChange.aspx?uri=https://opviewer.com
https://www.kath-kirche-kaernten.at/pfarren/pfarre/C3014?URL=https://opviewer.com
http://gs.matzendorf.at/includes/linkaufruf.asp?art=kapitel&link=https://opviewer.com
http://www.nuttenzone.at/jump.php?url=https://opviewer.com
https://cms.oeav-events.at/wGlobal/nessyEDVapps/layout/fancybox.php?link=https://opviewer.com
https://www.oebb.at/nightjet_newsletter/tc/xxxx?url=https://opviewer.com
https://www.gardensonline.com.au/Global/Players/YouTube.aspx?VideoURL=https://opviewer.com
http://www2.golflink.com.au/out.aspx?frm=gglcmicrosite&target=https://opviewer.com
http://www2.golflink.com.au/out.aspx?frm=logo&target=https://opviewer.com
https://www.golfselect.com.au/redirect?activityType_cd=WEB-LINK&course_id=2568&tgturl=https://opviewer.com
https://www.malcolmturnbull.com.au/?URL=https://opviewer.com
http://march-hare.com.au/library/default.asp?PP=/library/toc/lib-12.xml&tocPath=&URL=https://https://opviewer.com
https://www.oliverhume.com.au/enquiry/thank-you/?redirectTo=https://opviewer.com
http://www.parents-guide-illawarra.com.au/Redirect.aspx?destination=https://https://opviewer.com
https://ramset.com.au/Document/Url/?url=https://opviewer.com
https://ramset.com.au/document/url/?url=https://opviewer.com
http://rubyconnection.com.au/umbraco/newsletterstudio/tracking/trackclick.aspx?url=https://opviewer.com
http://southburnett.com.au/movies/movie.php?url=https://opviewer.com
https://www.vicsport.com.au/analytics/outbound?url=https://opviewer.com
https://www.vwwatercooled.com.au/forums/redirect-to/?redirect=https://https://opviewer.com
http://clients3.weblink.com.au/clients/aluminalimited/priceframe1.aspx?link=https://opviewer.com
https://maps.google.lt/url?sa=t&url=https://opviewer.com
https://ref.gamer.com.tw/redir.php?url=https://opviewer.com
https://images.google.com.sa/url?sa=t&url=https://opviewer.com
https://maps.google.com.sa/url?sa=t&url=https://opviewer.com
https://www.google.com.sa/url?sa=t&url=https://opviewer.com
https://images.google.hr/url?sa=t&url=https://opviewer.com
https://www.google.hr/url?sa=t&url=https://opviewer.com
https://maps.google.hr/url?sa=t&url=https://opviewer.com
https://images.google.com.pe/url?sa=t&url=https://opviewer.com
https://www.google.com.pe/url?sa=t&url=https://opviewer.com
https://maps.google.ae/url?sa=t&url=https://opviewer.com
https://images.google.ae/url?sa=t&url=https://opviewer.com
https://www.google.ae/url?sa=t&url=https://opviewer.com
https://www.google.co.ve/url?sa=t&url=https://opviewer.com
https://maps.google.co.ve/url?sa=t&url=https://opviewer.com
https://images.google.co.ve/url?sa=t&url=https://opviewer.com
http://onlinemanuals.txdot.gov/help/urlstatusgo.html?url=https://opviewer.com
https://www.google.com.pk/url?sa=t&url=https://opviewer.com
https://images.google.com.pk/url?sa=t&url=https://opviewer.com
https://community.rsa.com/t5/custom/page/page-id/ExternalRedirect?url=https://opviewer.com
https://www.google.com.eg/url?sa=t&url=https://opviewer.com
https://maps.google.com.eg/url?sa=t&url=https://opviewer.com
https://images.google.com.eg/url?sa=t&url=https://opviewer.com
https://www.google.si/url?sa=t&url=https://opviewer.com
https://maps.google.si/url?sa=t&url=https://opviewer.com
https://images.google.si/url?sa=t&url=https://opviewer.com
http://www.pickyourown.org/articles.php?NAME=Visit+Us&URL=https://opviewer.com
https://maps.google.lv/url?sa=t&url=https://opviewer.com
https://www.google.lv/url?sa=t&url=https://opviewer.com
https://images.google.lv/url?sa=t&url=https://opviewer.com
https://community.cypress.com/t5/custom/page/page-id/ExternalRedirect?url=https://opviewer.com
https://www.google.ee/url?sa=t&url=https://opviewer.com
https://cms.oeav-events.at/wGlobal/nessyEDVapps/layout/fancybox.php?link=https://opviewer.com
https://www.oebb.at/nightjet_newsletter/tc/xxxx?url=https://opviewer.com
https://www.gardensonline.com.au/Global/Players/YouTube.aspx?VideoURL=https://opviewer.com
http://www2.golflink.com.au/out.aspx?frm=gglcmicrosite&target=https://opviewer.com
http://www2.golflink.com.au/out.aspx?frm=logo&target=https://opviewer.com
https://www.golfselect.com.au/redirect?activityType_cd=WEB-LINK&courseid=2568&tgturl=https://opviewer.com
https://www.malcolmturnbull.com.au/?URL=https://opviewer.com
http://march-hare.com.au/library/default.asp?PP=/library/toc/lib-12.xml&tocPath=&URL=https://https://opviewer.com
https://www.oliverhume.com.au/enquiry/thank-you/?redirectTo=https://opviewer.com
http://www.parents-guide-illawarra.com.au/Redirect.aspx?destination=https://https://opviewer.com
https://ramset.com.au/Document/Url/?url=https://opviewer.com
https://ramset.com.au/document/url/?url=https://opviewer.com
http://rubyconnection.com.au/umbraco/newsletterstudio/tracking/trackclick.aspx?url=https://opviewer.com
http://southburnett.com.au/movies/movie.php?url=https://opviewer.com
https://www.vicsport.com.au/analytics/outbound?url=https://opviewer.com
https://www.vwwatercooled.com.au/forums/redirect-to/?redirect=https://https://opviewer.com
http://clients3.weblink.com.au/clients/aluminalimited/priceframe1.aspx?link=https://opviewer.com
https://clients1.google.ad/url?q=https://opviewer.com
https://cse.google.ad/url?q=https://opviewer.com
https://images.google.ad/url?q=https://opviewer.com
https://maps.google.ad/url?q=https://opviewer.com
https://www.google.ad/url?q=https://opviewer.com
https://emaratyah.ae/new-redirect.php?w=https://opviewer.com
http://mbrf.ae/knowledgeaward/language/ar/?redirecturl=https://opviewer.com
http://rafco.ae/container.asp?url=https://opviewer.com
http://for-css.ucoz.ae/go?https://opviewer.com
https://clients1.google.com.af/url?q=https://opviewer.com
https://cse.google.com.af/url?q=https://opviewer.com
https://images.google.com.af/url?q=https://opviewer.com
http://toolbarqueries.google.com.af/url?sa=t&url=https://opviewer.com
https://www.google.com.af/url?q=https://opviewer.com
https://www.snek.ai/redirect?url=https://opviewer.com
http://www.torrent.ai/lt/redirect.php?url=https://opviewer.com
http://avto.al/az/home/redirect?carId=1639612&url=https://opviewer.com
https://clients1.google.al/url?q=https://opviewer.com
https://cse.google.al/url?q=https://opviewer.com
https://images.google.al/url?q=https://opviewer.com
https://images.google.al/url?q=https://opviewer.com
http://toolbarqueries.google.al/url?q=https://opviewer.com
https://www.google.al/url?q=https://opviewer.com
http://tido.al/vazhdo.php?url=https://opviewer.com
http://smile.wjp.am/link-free/link3.cgi?mode=cnt&no=8&hpurl=https://opviewer.com
https://oxleys.app/friends.php?q=https://opviewer.com
http://www.ain.com.ar/openpop.php?url=https://opviewer.com
http://www.ain.com.ar/openpop.php?url=https://opviewer.com
https://www.google.nl/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.mw/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.mv/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.mu/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.ms/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.mn/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.mk/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.mg/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.me/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.md/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.lv/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.lu/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.lt/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.lk/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.li/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.la/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.kz/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.kg/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.jo/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.je/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.it/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.is/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.iq/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.ie/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.hu/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.ht/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.hr/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.hn/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.gr/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.gp/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.gm/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.gl/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.gg/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.ge/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.fr/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.fm/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.fi/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.es/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.ee/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.dz/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.dk/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.dj/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.de/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.cz/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.com/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.com.vn/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.com.uy/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.com.ua/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.com.tw/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.com.tr/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.com.sv/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.com.sg/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.com.sa/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.com.qa/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.com.py/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.com.pr/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.com.pk/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.com.ph/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.com.pe/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.com.pa/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.com.om/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.com.ni/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.com.ng/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.com.na/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.com.mz/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.com.my/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.com.mx/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.com.mt/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.com.ly/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.com.lb/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.com.kw/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.com.kh/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.com.jm/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.com.hk/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.com.gt/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.com.gi/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.com.gh/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.com.fj/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.com.et/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.com.eg/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.com.ec/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.com.do/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.com.cy/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.com.cu/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.com.co/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.com.bz/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.com.br/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.com.bo/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.com.bn/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.com.bh/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.com.bd/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.com.au/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.com.ar/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.com.ag/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.com.af/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.co.za/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.co.ve/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.co.uk/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.co.ug/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.co.tz/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.co.th/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.co.nz/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.co.ma/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.co.ls/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.co.kr/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.co.ke/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.co.jp/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.co.in/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.co.il/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.co.id/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.co.cr/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.co.bw/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.cm/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.cl/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.ci/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.ch/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.cd/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.cat/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.ca/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.by/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.bs/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.bi/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.bg/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.bf/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.be/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.ba/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.az/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.at/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.as/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.am/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.al/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.ae/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.google.ad/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://plus.google.com/url?q=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.tn/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.sn/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.sk/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.si/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.sh/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.se/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.rw/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.ru/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.rs/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.ro/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.pt/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.pl/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.no/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.nl/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.mw/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.mv/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.mu/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.ms/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.mn/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.mk/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.mg/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.lv/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.lu/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.lt/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.lk/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.li/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.la/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.kz/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.kg/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.jo/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.je/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.it/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.is/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.iq/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.ie/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.hu/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.ht/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.hr/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.hn/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.gr/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.gm/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.gl/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.gg/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.ge/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.fr/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.fm/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.fi/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.es/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.ee/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.dz/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.dk/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.dj/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.de/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.cz/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.com/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.com.uy/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.com.ua/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.com.tw/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.com.tr/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.com.sv/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.com.sg/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.com.sa/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.com.qa/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.com.py/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.com.pr/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.com.ph/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.com.pe/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.com.pa/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.com.om/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.com.ni/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.com.ng/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.com.na/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.com.mz/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.com.my/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.com.mx/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.com.mt/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.com.ly/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.com.lb/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.com.kw/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.com.kh/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.com.jm/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.com.hk/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.com.gt/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.com.gi/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.com.gh/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.com.fj/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.com.et/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.com.eg/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.com.ec/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.com.do/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.com.cu/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.com.co/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.com.bz/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.com.br/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.com.bo/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.com.bn/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.com.bh/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.com.bd/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.com.au/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.com.ar/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.com.ag/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.co.za/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.co.ve/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.co.uk/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.co.ug/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.co.tz/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.co.th/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.co.nz/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.co.ls/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.co.kr/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.co.ke/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.co.jp/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.co.in/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.co.il/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.co.id/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.co.cr/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.co.bw/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.cm/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.cl/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.ci/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.ch/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.cd/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.cat/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.ca/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.by/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.bs/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.bi/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.bg/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.bf/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.be/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.ba/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.at/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.as/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.ae/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://maps.google.ad/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.tn/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.sn/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.sk/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.si/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.sh/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.se/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.rw/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.ru/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.rs/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.ro/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.pt/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.ps/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.pl/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.no/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.nl/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.mw/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.mv/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.mu/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.ms/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.mn/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.mk/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.mg/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.me/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.md/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.lv/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.lu/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.lt/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.lk/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.li/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.la/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.kz/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.kg/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.jo/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.je/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.it/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.is/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.iq/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.ie/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.hu/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.ht/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.hr/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.hn/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.gr/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.gp/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.gm/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.gl/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.gg/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.ge/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.fr/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.fm/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.fi/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.es/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.ee/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.dz/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.dm/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.dk/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.dj/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.de/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.cz/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.com/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.com.vn/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.com.vc/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.com.uy/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.com.ua/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.com.tw/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.com.tr/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.com.sv/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.com.sg/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.com.sa/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.com.qa/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.com.py/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.com.pr/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.com.pk/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.com.ph/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.com.pe/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.com.pa/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.com.om/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.com.np/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.com.ni/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.com.ng/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.com.na/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.com.mz/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.com.my/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.com.mx/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.com.mt/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.com.ly/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.com.lb/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.com.kw/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.com.kh/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.com.jm/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.com.hk/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.com.gt/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.com.gi/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.com.gh/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.com.fj/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.com.et/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.com.eg/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.com.ec/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.com.do/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.com.cy/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.com.cu/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.com.co/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.com.bz/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.com.br/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.com.bo/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.com.bn/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.com.bh/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.com.bd/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.com.au/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.com.ar/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.com.ag/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.com.af/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.co.zm/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.co.za/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.co.za/url?q=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.co.ve/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.co.uz/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.co.uk/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.co.ug/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.co.tz/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.co.th/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.co.th/url?q=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.co.nz/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.co.ma/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.co.ls/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.co.kr/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.co.kr/url?q=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.co.ke/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.co.jp/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.co.in/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.co.il/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.co.id/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.co.id/url?q=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.co.cr/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.co.cr/url?q=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.co.ck/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.co.bw/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.cm/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.cl/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.ci/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.ch/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.cg/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.cd/url?sa?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.ca/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.by/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.bs/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.bi/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.bg/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.bf/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.be/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.ba/url?sa=t&url=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://images.google.ba/url?q=https://massagestudionews.blogspot.com/2022/11/9-astonishing-advantages-of-massage.html
https://www.gamja888.com/ – 바카라사이트,카지노사이트,카지노게임사이트,온라인바카라,온라인카지노,Gamja888
https://gamja888.com/ – 퀸즈슬롯
https://gamja888.com/ – 맥스카지노
https://gamja888.com/ – 비바카지노
https://gamja888.com/ – 카지노주소
https://gamja888.com/ – 바카라추천
https://gamja888.com/ – 온라인바카라게임
https://gamja888.com/ – 안전한 바카라사이트
https://gamja888.com/ – 바카라
https://gamja888.com/ – 카지노
https://gamja888.com/ – 퀸즈슬롯 카지노
https://gamja888.com/ – 바카라게임사이트
https://gamja888.com/onlinebaccarat/ – 온라인바카라
https://gamja888.com/millionclubcasino/ – 밀리언클럽카지노
https://gamja888.com/safecasinosite/ – 안전카지노사이트
https://gamja888.com/baccaratsiterecommendation/ – 바카라사이트추천
https://gamja888.com/ourcasino/ – 우리카지노계열
https://gamja888.com/slotmachine777/ – 슬롯머신777
https://gamja888.com/royalcasinosite/ – 로얄카지노사이트
https://gamja888.com/crazyslot/ – 크레이지슬롯
https://gamja888.com/onlineblackjack/ – 온라인블랙잭
https://gamja888.com/internetroulette/ – 인터넷룰렛
https://gamja888.com/casinoverificationsite/ – 카지노검증사이트
https://gamja888.com/safebaccaratsite/ – 안전바카라사이트
https://gamja888.com/mobilebaccarat/ – 모바일바카라
https://gamja888.com/howtowin-baccarat/ – 바카라 필승법
https://gamja888.com/meritcasino/ – 메리트카지노
https://gamja888.com/baccarat-howto/ – 바카라 노하우
youube.me
instagrme.com
youubbe.me
Instagrm.me
Instagrme.net
internetgame.me
instagrme.live
naverom.me
facebokom.me