URUSHIOL: The Poison Present in Poison Ivy
Poison ivy, also known as the “Eastern Poison Ivy”, is a plant native to Eastern North America. When touched, the secretions present in the leaves causes a condition called contact dermatitis, which is characterized with a severe, itchy and painful inflammation of the skin. The toxic principle present in the secretions is Urushiol and is being produced in certain parts of plants. With its slightly nonvolatile nature, urushiol is easily carried by different media such as clothing, gardening tools, soil, shoes, or even through smoke coming from the burned Poison Ivy plant.
How does Urushiol look like in Chemistry?

Let’s Get Building!
Using your Student Molecular Set from Duluth Labs let’s create Urushiol! You’ll need:
- 21 Carbon atoms
- 2 Oxygen atoms
- 36 Hydrogen atoms
- 9 Small connectors (compact small bonds for hydrogen)
- 6 Medium Connectors
- 8 Long connectors
- Molecular Tool (for Disassembly)
Put aside all the atoms and connectors needed.
We will build Urushiol in a clockwise direction, starting with Carbon 1!
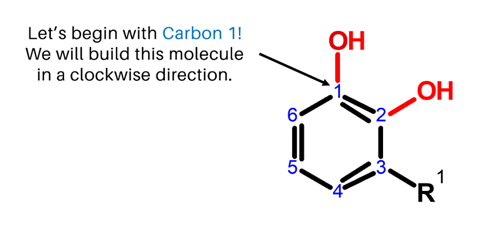
Steps:
-
1

1. Get one carbon atom (Carbon 1) then, attach an Oxygen atom to it using a medium connector. Add 1 hydrogen atom to this oxygen using 1 small connector.
-
2

2. Then, get another carbon atom (Carbon 2) and attach this to Carbon 1 using 2 long connectors.
-
3

3. Attach 1 oxygen atom Carbon 2 using 1 medium connector. Then place 1 hydrogen atom on this oxygen using 1 small connector
-
4

4. Next, get one carbon atom (Carbon 3) then attach this below Carbon 2 using a medium connector.
-
5

5. Then, get another carbon atom (Carbon 4) then attach this to Carbon 3 using 2 long connectors. Place 1 hydrogen atom on Carbon 4 using a small connector.
-
6

6. Grab one Carbon atom (Carbon 5) and attach this next to Carbon 4 using a medium connector. Likewise, place one hydrogen atom on Carbon 5 using a small connector.
-
7

7. Then, get the last carbon atom (Carbon 6) then attach this to Carbon 5 using 2 long connectors. Add one hydrogen atom on Carbon 6 using 1 small connector.
-
8

8. Finally, join Carbon 6 and Carbon 1 together using 1 medium connector.
We now have our Urushiol ring. Let’s try building its R substituent.
Have your 15 Carbon atoms and 31 Hydrogen atoms ready, and let’s start off with Carbon 1 of the alkyl chain!
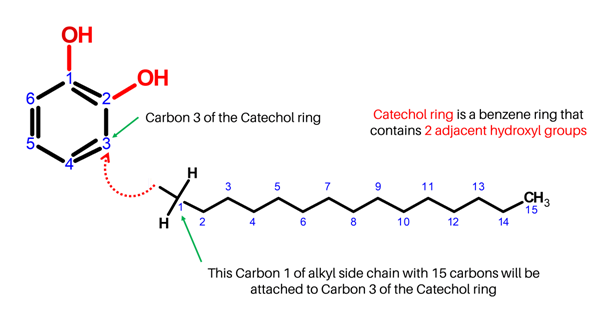
-
1

1. Get one carbon atom (Carbon 1) then, attach this to Carbon 3 of the benzene ring using a medium connector. Attach 2 hydrogen atoms to it with your 2 small connectors.
-
2

2. Get another Carbon atom (Carbon 2) then attach this next to Carbon 1 (of the alkyl chain) using a medium connector. Add 2 hydrogens to Carbon 2 using 2 small connectors.
-
3

3. Repeat step 2 for Carbons 3 to 14. Make sure to attach the succeeding carbons next to each other.
-
4

4. Finally, get the last Carbon atom (Carbon 15) then connect this to Carbon 14 of the alkyl chain using a medium connector. Add 3 hydrogen atoms to Carbon 15 using 3 small connectors.



















https://cutt.ly/i4fyCza
https://bit.ly/3Ln1imY
https://spo337.com/
머니라인247 https://spo337.com/ml247/
황룡카지노 https://spo337.com/gdcasino/
아시안커넥트 https://spo337.com/asianconnect/
스보벳 https://spo337.com/sbobet/
피나클 https://spo337.com/pinbet88/
맥스벳 https://spo337.com/maxbet/
파워볼 https://spo337.com/category/powerball/
BTI Sports https://spo337.com/btisports/
해외배팅사이트 추천 https://spo337.com/
해외스포츠배팅사이트 추천 https://spo337.com/
해외배팅사이트에이전시 추천 https://spo337.com/
https://sites.google.com/view/spo337/
https://sites.google.com/view/spo337/%20
http://sites.google.com/view/spo337
https://sites.google.com/view/spo337/
https://sites.google.com/view/spo337/%2F
http://sites.google.com/view/spo337/%20
http://sites.google.com/view/spo337/
http://www.sites.google.com/view/spo337/
https://www.sites.google.com/view/spo337/
http://www.sites.google.com/view/spo337/%20
https://www.sites.google.com/view/spo337/%20
http://www.sites.google.com/view/spo337/%2F
https://www.sites.google.com/view/spo337/%2F
https://sites.google.com/view/xn—b60by94a1wbra414jbsf-/%EC%95%84%EC%8B%9C%EC%95%88%EC%BB%A4%EB%84%A5%ED%8A%B8
https://sites.google.com/view/xn—b60by94a1wbra414jbsf-/%20
http://sites.google.com/view/xn—b60by94a1wbra414jbsf-/
https://sites.google.com/view/xn—b60by94a1wbra414jbsf-/%2F
http://sites.google.com/view/xn—b60by94a1wbra414jbsf-/%2F
https://www.sites.google.com/view/xn—b60by94a1wbra414jbsf-/
http://www.sites.google.com/view/xn—b60by94a1wbra414jbsf-/
https://ln.run/Cl2oj
https://rb.gy/iv97s
https://apaste.info/qsmr
https://bitbin.it/WBykrHkf/
https://rb.gy/o9da1r
https://commie.io/#20ihmGcp
https://apaste.info/xqvA
https://paste.myst.rs/9lwr05lw
https://commie.io/#WxnN33j6
https://pasteio.com/xzeZLnRsjqhn
https://pasteio.com/xToV5U2uv0g9
https://abelovsky.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://absolon.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://achenbach.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://adamabelovsky.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://adamb-bartos.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://adamburda.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://adamjanak.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://adammikulasek.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://adamrykala.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://adamskala.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://adamtoman.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://adamvanek.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://adamvasina.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://adelaberanova.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://adelavichova.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://agalarov.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://alagia.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://alanni.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://alenabenesova.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://alenapekarova.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://alenapitrova.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://alesbaloun.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://alesbeseda.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://alesmerta.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://alesszabo.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://alesthruby.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://alexandrasynac.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://alexandraudzenija.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://alexandrbenda.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://alexova.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://alicebaresova.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://alik.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://aloisnebel.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://aloukla.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://andreaholopova.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://andreanovotna1.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://andrejbabis.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://andrejruscak.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://andrysova.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://anetamachova.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://annamacickova.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://annanovotna.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://antl.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://antoninkozel.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://antoninkralik.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://antoninkratochvil.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://antoninkvapil.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://antoninmares.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://antoninmazac.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://antoninpecenka.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://antoninsevcik.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://aplikace.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://arnoldlicka.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://audiokrystof.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://auer.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://augustini.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://aulehlova.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://auto.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://aviou.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://axamit.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://babakmichal.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://babel.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://babica.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://babickazvolska.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://babikova.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bacik.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://badman.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bajnar.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://balaban.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://balhar.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://balmetova.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://balusek.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bandik.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://banga.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bangova.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://baoviet.com.vn/Redirect.aspx?url=https%3A%2F%2Fspo337.com/
https://baranek.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://baranka.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://barborakoznarova.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://barborakubatova.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://barborasedlackova.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://barboraskreckova.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://barboratopinkova.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://barboravesela.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bardi.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bardoun.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bares.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://barsa.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bartackova.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bartas.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bartos.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bartosmartin.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bartosova.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bartyzal.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://basket.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://batikova.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://baubin.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://baudys.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bausova.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bayerl.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://becker.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://becvar.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://becvarova.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bederka.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bednarik.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bednarova.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bedrichdvorak.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://beerova.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://behounek.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://belapolaskova.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://belatrebinova.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bellingerova.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://belobradek.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://belova.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://belsanova.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://benesovsky.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://benyacoub.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bercik.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://beres.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bergerova.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bergl.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bergman.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bernasek.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bernkopfova.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://berrezouga.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://berrouche.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://berwidbuquoy.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://beso.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://besser.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bezdek.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bezecny.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://beznoska.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bezrodny.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bicera.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bilek.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://binko.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bittnerova.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://blabolova.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://blaha.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://blahastanislav.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://blanarova.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://blanka.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://blumelova.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://blumkova.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bobek.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bobelova.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bobosikova.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bockova.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bodova.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://boehmova.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://boettinger.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bohac.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bohacek.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bohackova.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bohata.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bohdanprochazka.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bohme.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bohumilatruhlarova.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bohumirkolar.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bohumirsimek.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bohumirzidek.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bohuslavsobotka.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bohusova.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bojko.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bojkova.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://borovicka.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://borovska.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://borovy.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bortel.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bortlik.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://boruvka.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bosak.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://boskova.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bostlova.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://botek.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://boucek.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://boudakasparova.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bouska.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bozenamokrosova.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bradacova.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://bradna.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://www.triathlon.org/?URL=https%3A%2F%2Fspo337.com/
https://www.popcouncil.org/scripts/leaving.asp?URL=https%3A%2F%2Fspo337.com/
https://www.ppa.com/?URL=https%3A%2F%2Fspo337.com/
https://interpals.net/url_redirect.php?href=https%3A%2F%2Fspo337.com/
https://www.siemenstransport.com/careers?redirect=1&url=https%3A%2F%2Fspo337.com/
https://sc.sie.gov.hk/TuniS/https%3A%2F%2Fspo337.com/
https://shorefire.com/?URL=https%3A%2F%2Fspo337.com/
https://www.octranspo.com/en/about-us/confederation-line-1-website?URL=https%3A%2F%2Fspo337.com/
https://www.ait.ie/?URL=https%3A%2F%2Fspo337.com/
https://www.swrve.com/?URL=https%3A%2F%2Fspo337.com/
https://timberlinelodge.com/?URL=https%3A%2F%2Fspo337.com/
https://myemma.com/?URL=https%3A%2F%2Fspo337.com/
https://www.usich.gov/?URL=https%3A%2F%2Fspo337.com/
https://www.malcolmturnbull.com.au/?URL=https%3A%2F%2Fspo337.com/
https://www.hockney.com/?URL=https%3A%2F%2Fspo337.com/
https://www.adminer.org/redirect/?url=https%3A%2F%2Fspo337.com/
https://www.omnigroup.com/omnifocus/?URL=https%3A%2F%2Fspo337.com/
https://www.chuys.com/?URL=https%3A%2F%2Fspo337.com/
https://news.url.google.com/url?q=https%3A%2F%2Fspo337.com/
http://www.winnipegfreepress.com/s?action=doLogout&rurl=https%3A%2F%2Fspo337.com/
https://mypage.syosetu.com/?jumplink=https%3A%2F%2Fspo337.com/
http://yp.ocregister.com/media/js/netsoltrademark.php?d=https%3A%2F%2Fspo337.com/
http://yellowpages.staradvertiser.com/media/js/netsoltrademark.php?d=https%3A%2F%2Fspo337.com/
http://local.lex18.com/media/js/netsoltrademark.php?d=https%3A%2F%2Fspo337.com/
http://yp.ocregister.com/media/js/netsoltrademark.php?d=https%3A%2F%2Fspo337.com/
http://yellowpages.sfexaminer.com/media/js/netsoltrademark.php?d=https%3A%2F%2Fspo337.com/
http://localbusiness.starnewsonline.com/media/js/netsoltrademark.php?d=https%3A%2F%2Fspo337.com/
http://acceletronics.de/media/js/netsoltrademark.php?d=https%3A%2F%2Fspo337.com/
http://cinca.com/media/js/netsoltrademark.php?d=https%3A%2F%2Fspo337.com/
http://borlamufflers.at/media/js/netsoltrademark.php?d=https%3A%2F%2Fspo337.com/
http://www.findingyoudeals007.com/media/js/netsoltrademark.php?d=https%3A%2F%2Fspo337.com/
http://worldoftours.org/media/js/netsoltrademark.php?d=https%3A%2F%2Fspo337.com/
http://1stscotia.com/media/js/netsoltrademark.php?d=https%3A%2F%2Fspo337.com/
http://vitalstats.com/media/js/netsoltrademark.php?d=https%3A%2F%2Fspo337.com/
http://alloysurfcaesinc.info/media/js/netsoltrademark.php?d=https%3A%2F%2Fspo337.com/
http://www.helixturnhelix.com/media/js/netsoltrademark.php?d=https%3A%2F%2Fspo337.com/
http://www.martin-garcia.com/media/js/netsoltrademark.php?d=https%3A%2F%2Fspo337.com/&popup=1
http://www.red-dot.com/media/js/netsoltrademark.php?d=https%3A%2F%2Fspo337.com/
http://www.thegreennest.com/media/js/netsoltrademark.php?d=https%3A%2F%2Fspo337.com/
http://c-uslaw.com/media/js/netsoltrademark.php?d=https%3A%2F%2Fspo337.com/
https://www.invisalign-doctor.com.au/api/redirect?url=https%3A%2F%2Fspo337.com
http://d-click.fmcovas.org.br/u/20636/11/16715/41_0/0c8eb/?url=https%3A%2F%2Fspo337.com
http://uriburner.com/HtmlPivotViewer/?url=https%3A%2F%2Fspo337.com
https://amp.wte.net/t.aspx?S=23&ID=5679&NL=1431&N=6739&SI=881487&url=https%3A%2F%2Fspo337.com
https://ads.atype.jp/track/click.php?aid=10577&linkid=T122&redirect=https%3A%2F%2Fspo337.com&mid=85
https://click.atype.jp/track/click.php?aid=552&linkid=P22&redirect=https%3A%2F%2Fspo337.com
https://www.atype.jp/track/click.php?aid=1663&linkid=b9&redirect=https%3A%2F%2Fspo337.com
http://www.apples4theteacher.com/cgi-bin/redirect.cgi?https%3A%2F%2Fspo337.com
http://scanverify.com/siteverify.php?site=www.https%3A%2F%2Fspo337.com
http://typedia.com/?URL=https%3A%2F%2Fspo337.com
https://www.nennung-online.de/pages/tracker.php?id=431&languageid=1&link=https%3A%2F%2Fspo337.com
http://ir.chartnexus.com/s/p.php?u=1&f=1&c=108&t=https%3A%2F%2Fspo337.com
http://www.ds-360.com/goto_url.asp?url=https%3A%2F%2Fspo337.com
https://inmusicbrands.com/?URL=www.https%3A%2F%2Fspo337.com/
http://courses.test.bg/modules/babel/redirect.php?newlang=bg_BG&newurl=https%3A%2F%2Fspo337.com
http://uproxy.library.dc-uoit.ca/login?url=https%3A%2F%2Fspo337.com
http://www.butikstrender.se/?bsa_pro_id=19&bsa_pro_url=https%3A%2F%2Fspo337.com
https://ch.atomy.com/products/m/SG?prodUrl=https%3A%2F%2Fspo337.com
http://avp.innity.com/click/?campaignid=10933&adid=115198&zoneid=39296&pubid=3194&ex=1412139790&pcu=&auth=3tx88b-1412053876272&url=https%3A%2F%2Fspo337.com
http://wibo.m78.com/rank/rl_out.cgi?id=jjjsss&url=https%3A%2F%2Fspo337.com
http://dlibrary.mediu.edu.my/cgi-bin/koha/tracklinks.pl?uri=https%3A%2F%2Fspo337.com
https://www.dramonline.org/redirect?url=https%3A%2F%2Fspo337.com
http://www.canasvieiras.com.br/redireciona.php?url=https%3A%2F%2Fspo337.com
https://www.otohits.net/home/redirectto?url=https%3A%2F%2Fspo337.com
http://projects.europa.ba/ProjectDetails/Index/PCL_46?returnUrl=https%3A%2F%2Fspo337.com
https://baumspage.com/cc/ccframe.php?path=https%3A%2F%2Fspo337.com
http://www.trackroad.com/conn/garminimport.aspx?returnurl=https%3A%2F%2Fspo337.com
https://atlanticleague.com/tracker/index.html?t=ad&pool_id=11&url=https%3A%2F%2Fspo337.com
https://www.nacogdoches.org/banner-outgoing.php?banner_id=38&b_url=https%3A%2F%2Fspo337.com
https://pixel.sitescout.com/iap/6ad1383b0f81bb61?cookieQ=1&r=https%3A%2F%2Fspo337.com
http://www.myauto.by/away.php?url=https%3A%2F%2Fspo337.com
http://www.yellowmoxie.com/redirect.php?tt=80000&sk=Aspire&ka=Aspire&ourl=https%3A%2F%2Fspo337.com
https://www.e-expo.net/category/click_url.html?url=https%3A%2F%2Fspo337.com
https://dijaski.net/redirect?u=https%3A%2F%2Fspo337.com
https://join.chat/en/powered/?site=THAT+NUTRITIOUS+GLOW&url=https%3A%2F%2Fspo337.com
https://ma.by/away.php?url=https%3A%2F%2Fspo337.com
https://378.hatenablog.com/iframe/hatena_bookmark_comment?canonical_uri=https%3A%2F%2Fspo337.com
https://mercury.postlight.com/amp?url=https%3A%2F%2Fspo337.com
https://www.fuzokubk.com/cgi-bin/LinkO.cgi?u=www.https%3A%2F%2Fspo337.com
http://smtp.mystar.com.my/interx/tracker?op=click&id=995.1e0d&url=https%3A%2F%2Fspo337.com
https://naviking.localking.com.tw/about/redirect.aspx?mid=7&url=https%3A%2F%2Fspo337.com
http://staging.talentegg.ca/redirect/company/224?destination=https%3A%2F%2Fspo337.com
https://bvgrider.onelink.me/JTVb?af_web_dp=https%3A%2F%2Fspo337.com
http://landofvolunteers.com/go.php?https%3A%2F%2Fspo337.com
https://hatenablog-parts.com/embed?url=https%3A%2F%2Fspo337.com
https://timemapper.okfnlabs.org/view?url=https%3A%2F%2Fspo337.com
http://oita.doctor-search.tv/linkchk.aspx?no=1171&link=https%3A%2F%2Fspo337.com
http://orders.gazettextra.com/AdHunter/Default/Home/EmailFriend?url=https%3A%2F%2Fspo337.com
http://whois.hostsir.com/?domain=www.https%3A%2F%2Fspo337.com
http://marillion.com/forum/index.php?thememode=mobile;redirect=https%3A%2F%2Fspo337.com
https://trk.hbomax.com/aff_c?offer_id=5&aff_id=1026&aff_click_id=toptenreviews-us-9916793282449424000&url=https%3A%2F%2Fspo337.com&aff_sub2=https://www.toptenreviews.com/best-tv-streaming-services-sites/best-tv-streaming-services-sites
https://panarmenian.net/eng/tofv?tourl=https%3A%2F%2Fspo337.com
http://www.marchhare.jp/rs.php?url=https%3A%2F%2Fspo337.com
http://egov2.miamigov.com/Office_of_Auditor_General/admin/Portal/LinkClick.aspx?tabid=1&table=Announcements&field=ItemID&id=98&link=https%3A%2F%2Fspo337.com
https://nanos.jp/jmp?url=https%3A%2F%2Fspo337.com
http://hirlevel.mediacenter.hu/click.php?hirlevel_id=12405549102322&url=https%3A%2F%2Fspo337.com
http://parkcities.bubblelife.com/click/c3592/?url=https%3A%2F%2Fspo337.com
http://convertit.com/redirect.asp?to=https%3A%2F%2Fspo337.com
https://www.askart.com/redirect.aspx?ToURL=https%3A%2F%2Fspo337.com
https://www.iaai.com/Images/ViewAllImages?stockNumber=28203522&branchCode=443&branchId=443&salvageId=28652718&VehicleSearchurl=https%3A%2F%2Fspo337.com
https://devot-ee.com/?URL=https%3A%2F%2Fspo337.com
https://w1.websnadno.cz/index.php?menu1r=2&stiznost=true&surl=https%3A%2F%2Fspo337.com&sreferer=https://w1.websnadno.cz/
https://blog.utoledo.edu/feed2js/feed2js_async.php?src=https%3A%2F%2Fspo337.com&num=4&date=y&tz=n&utf=y&targ=blank
http://airkast.weatherology.com/web/lnklog.php?widget_id=1&lnk=https%3A%2F%2Fspo337.com
http://smile.wjp.am/link-free/link3.cgi?mode=cnt&no=8&hpurl=https%3A%2F%2Fspo337.com
http://www.snwebcastcenter.com/event/page/count_download_time.php?url=https%3A%2F%2Fspo337.com
http://d-click.fiemg.com.br/u/18081/131/75411/137_0/82cb7/?url=https%3A%2F%2Fspo337.com
http://nethunt.co/api/v1/track/link/click/5c801d81d23c1b3d70efbe8a/1556808049608/?link=https%3A%2F%2Fspo337.com
http://welcomepage.ca/link.asp?id=58%7Ehttps%3A%2F%2Fspo337.com
http://shp.hu/hpc_uj/click.php?ml=5&url=https%3A%2F%2Fspo337.com
https://www.morhipo.com/shared/partnercookie?k=gort&url=https%3A%2F%2Fspo337.com
http://www.sa-live.com/merror.html?errortype=1&url=https%3A%2F%2Fspo337.com
http://www.bookmerken.de/?url=https%3A%2F%2Fspo337.com
http://www.mejtoft.se/research/?page=redirect&link=https%3A%2F%2Fspo337.com
http://old.evermotion.org/stats.php?url=https%3A%2F%2Fspo337.com
http://davidpawson.org/resources/resource/416?return_url=https%3A%2F%2Fspo337.com
https://www.dcfever.com/click.php?id=lensdb_fbs&url=https%3A%2F%2Fspo337.com
http://pasco.k12.fl.us/?URL=www.https%3A%2F%2Fspo337.com
https://www.freado.com/trackviews.php?action=buy&bookid=16477&buylink=https%3A%2F%2Fspo337.com
https://multiply.co.za/sso/flyover/?url=https%3A%2F%2Fspo337.com
https://bnc.lt/a/key_live_pgerP08EdSp0oA8BT3aZqbhoqzgSpodT?medium=&feature=&campaign=&channel=&$always_deeplink=0&$fallbackurl=www.https%3A%2F%2Fspo337.com&$deeplinkpath=&p=c11429c2860165eee314
https://images.google.com.bo/url?q=https%3A%2F%2Fspo337.com/
https://cse.google.com.bo/url?sa=i&url=https%3A%2F%2Fspo337.com/
https://cse.google.bs/url?sa=i&url=https%3A%2F%2Fspo337.com/
https://cse.google.mu/url?sa=i&url=https%3A%2F%2Fspo337.com/
https://maps.google.mk/url?q=https%3A%2F%2Fspo337.com/
https://images.google.mk/url?q=https%3A%2F%2Fspo337.com/
https://cse.google.mk/url?sa=i&url=https%3A%2F%2Fspo337.com/
https://cse.google.al/url?sa=i&url=https%3A%2F%2Fspo337.com/
https://cse.google.li/url?sa=i&url=https%3A%2F%2Fspo337.com/
https://cse.google.mn/url?sa=i&url=https%3A%2F%2Fspo337.com/
https://cse.google.com.bh/url?sa=i&url=https%3A%2F%2Fspo337.com/
https://cse.google.com.kh/url?sa=i&url=https%3A%2F%2Fspo337.com/
https://cse.google.com.lb/url?sa=i&url=https%3A%2F%2Fspo337.com/
https://cse.google.tt/url?sa=i&url=https%3A%2F%2Fspo337.com/
https://maps.google.tt/url?q=https%3A%2F%2Fspo337.com/
https://cse.google.ci/url?sa=i&url=https%3A%2F%2Fspo337.com/
https://images.google.ci/url?q=https%3A%2F%2Fspo337.com/
https://maps.google.dj/url?q=https%3A%2F%2Fspo337.com/
https://images.google.dj/url?q=https%3A%2F%2Fspo337.com/
https://cse.google.dj/url?sa=i&url=https%3A%2F%2Fspo337.com/
https://cse.google.hn/url?sa=i&url=https%3A%2F%2Fspo337.com/
https://maps.google.hn/url?q=https%3A%2F%2Fspo337.com/
https://images.google.hn/url?q=https%3A%2F%2Fspo337.com/
https://maps.google.co.tz/url?q=https%3A%2F%2Fspo337.com/
https://cse.google.co.tz/url?sa=i&url=https%3A%2F%2Fspo337.com/
https://images.google.co.tz/url?q=https%3A%2F%2Fspo337.com/
https://images.google.gm/url?q=https%3A%2F%2Fspo337.com/
https://cse.google.gm/url?sa=i&url=https%3A%2F%2Fspo337.com/
https://maps.google.gm/url?q=https%3A%2F%2Fspo337.com/
https://maps.google.com.py/url?q=https%3A%2F%2Fspo337.com/
https://images.google.com.py/url?q=https%3A%2F%2Fspo337.com/
http://ijbssnet.com/view.php?u=spo337.com
https://caminhoesecarretas.com.br/redirect.aspx?id=1083&url=spo337.com
http://www.humanbrainmapping.org/i4a/etrack/track.cfm?rType=2&campaignID=3572&contactID=4524&origurl=spo337.com
http://www.iqads.ro/bitrix/rk.php?goto=spo337.com
https://clra.member365.com/ecommunication/api/click/k8UH1S35jdDig9F-Y-f_jQ/nyfRYG7i4IX2z99VcvAhJw?r=spo337.com
https://slenderierecord.futureartist.net/external_redirect?text_lnk=spo337.com
http://eva-dmc4.halfmoon.jp/eva-dmc4/cutlinks/rank.php?url=spo337.com
https://hondanews.eu/mobileredirect/2?url=spo337.com
http://forward.zillertal.at/?url=spo337.com
https://www.bioguiden.se/redirect.aspx?url=spo337.com
https://www.feedroll.com/rssviewer/feed2js.php?src=spo337.com
https://www.anybeats.jp/jump/?spo337.com
https://www.kyrktorget.se/includes/statsaver.php?type=kt&id=4209&url=spo337.com
https://www.yesasia.com/global/0-0-0-cid.null_cuc.null_ac.XDYDPHR1-en/assocred.html?rdl=spo337.com
https://www.cityrealty.com/ad-server?id=14509&adg=B&url=spo337.com&p=1&i=600
https://zippyapp.com/redir?u=spo337.com
https://www.tshirthell.com/store/clicks.php?partner=sbgerlinkd&page=spo337.com
https://www.sodertalje.se/find_v2/click?t_id=1B2M2Y8AsgTpgAmY7PhCfg==&t_q=852&t_tags=language:sv,siteid:67f9c486-281d-4765-ba72-ba3914739e3b&t_ip=193.181.21.1&t_hit.id=Livs_Common_Model_PageTypes_ArticlePage/a30e9a85-39dc-40fd-babd-9e9452bc561b_sv&t_hit.pos=1&t_redirect=spo337.com
https://screenmediafilms.net/out/watch/486?link=spo337.com
https://barbados.org/al/?event=ad.logClickampadvert=DC6FF007FAD78E23C54A673E3258DDC0EE638CB31CFE6FB9D0F4E0C53EF6B1276EC9DDCA3D10A7EA5E5F52955053E7F2A0C5D4D51F5050E21EC0B7F8CDCDA1EB3BBEEEDAB3EBDC25114C276741BA028E&webAddress=spo337.com
https://dat.2chan.net/bin/jump.php?spo337.com
http://novalogic.com/remote.asp?nlink=spo337.com
https://www.teacherlists.com/files/banner.php?title=Mrs.%20Roberts&link=spo337.com
http://www3.valueline.com/vlac/logon.aspx?lp=spo337.com
https://forssanlehti.portal.worldoftulo.com/Registration/Lookup/?returnUrl=spo337.com
https://gamergen.com/redirect/?url=spo337.com
https://www.hobowars.com/game/linker.php?url=spo337.com
http://www.arakhne.org/redirect.php?url=spo337.com
http://www.peterblum.com/releasenotes.aspx?returnurl=spo337.com
http://us.member.uschoolnet.com/register_step1.php?from=www.https%3A%2F%2Fspo337.com
https://www.esato.com/go.php?url=spo337.com
https://www.checkraka.com/click.php?url=spo337.com
http://aboutlincolncenter.org/component/dmms/handoff?backurl=spo337.com
http://www.americantourister.com/disneyside/bumper.php?r=spo337.com
https://pireminder.com/redirect/?to=spo337.com
https://brief.promax.org/revive/www/delivery/ck.php?ct=1&oaparams=2bannerid=99zoneid=3cb=04a8d0f9edoadest=spo337.com
http://www.buddssaab.ca/wp-content/plugins/wp-js-external-link-info/redirect.php?blog=Budds20Oakville&url=spo337.com
http://www.cross-a.net/go_out.php?url=spo337.com
https://www.lnfcu.com/helpers/choice.asp?h=spo337.com
https://www.textise.net/showtext.aspx?strurl=www.https%3A%2F%2Fspo337.com
http://members.asoa.org/sso/logout.aspx?returnurl=spo337.com
http://www.antiqueweek.com/scripts/sendoffsite.asp?url=https://https%3A%2F%2Fspo337.com
http://go.eniro.dk/lg/rejse-guide/europa/cat-1694/https://https%3A%2F%2Fspo337.com
http://m.shopinsanantonio.com/redirect.aspx?url=https%3A%2F%2Fspo337.com/
http://www.cqfuzhuang.com/url.asp?url=https%3A%2F%2Fspo337.com/
https://www.svenskaracefans.com/ex.aspx?t=https%3A%2F%2Fspo337.com/
https://joomlinks.org/?url=https%3A%2F%2Fspo337.com/
http://www.startgames.ws/friend.php?url=https%3A%2F%2Fspo337.com/product/usa-email-database/&title=Xo%20Wars%20-%20tic%20tac%20too%20flash%20game.
https://www.pennergame.de/redirect/?site=https%3A%2F%2Fspo337.com/
http://www.jkes.tyc.edu.tw/dyna/netlink/hits.php?id=527&url=https%3A%2F%2Fspo337.com/
https://tanganrss.com/rsstxt/cushion.php?url=spo337.com/
https://getpocket.com/redirect?url=https%3A%2F%2Fspo337.com/
http://doodle.com/r?url=https%3A%2F%2Fspo337.com/
https://g.i.ua/?userID=6897361&userID=6897361&url=https%3A%2F%2Fspo337.com/
http://lexicon.arvindlexicon.com/Pages/RedirectHostPage.aspx?language=English&word=multidecker&redirect_to=https%3A%2F%2Fspo337.com/
https://prosports-shop.com/shop/display_cart?return_url=https%3A%2F%2Fspo337.com/
https://bbs.pku.edu.cn/v2/jump-to.php?url=https%3A%2F%2Fspo337.com/
https://www.ask.com/web?qo=extensionSearchBox&o=1473171&ad=semA&q=spo337.com/
https://adactio.com/extras/talklikeapirate/translate.php?filename=https%3A%2F%2Fspo337.com/
https://www.copyscape.com/view.php?u=spo337.com/
http://ww11.aitsafe.com/cf/review.cfm?userid=d0223865&return=spo337.com/
http://nitrogen.sub.jp/php/Viewer.php?URL=https%3A%2F%2Fspo337.com/
https://articulate.com/support/article/Articulate-Online-End-of-Life-Transition-Guide?Returnurl=https%3A%2F%2Fspo337.com/
https://forex-blog-uk.blogspot.com/search/?label=https%3A%2F%2Fspo337.com/
http://svenvanbolt.de/topframe.php?https%3A%2F%2Fspo337.com/
https://id.nan-net.jp/system/login/link.cgi?jump=https%3A%2F%2Fspo337.com/
https://kinhtexaydung.net/redirect/?url=https%3A%2F%2Fspo337.com/
http://givc.by/bitrix/rk.php?goto=https%3A%2F%2Fspo337.com/
http://reisenett.no/annonsebanner.tmpl?url=https%3A%2F%2Fspo337.com/
https://www.ahewar.org/links/dform.asp?url=https%3A%2F%2Fspo337.com/
https://jla.drmuller.net/r.php?url=https%3A%2F%2Fspo337.com/
https://www.earthlost.de/deref.php?url=https%3A%2F%2Fspo337.com/
https://www.smore.com/app/reporting/out/f677?u=https%3A%2F%2Fspo337.com/
https://www.ning.com/xn/authenticate?target=https%3A%2F%2Fspo337.com/
https://www.repubblica.it/social/sites/repubblica/d/boxes/shares/sharebar.cache.php?t=float-2017-v1&url=https%3A%2F%2Fspo337.com
https://www.gouv.ci/banniere/adclick.php?bannerid=595&zoneid=2&dest=https%3A%2F%2Fspo337.com
https://liquidmaps.org/users_fichas_items/index/626/440?return=https%3A%2F%2Fspo337.com
https://infosmi.com/redirect.php?url=https%3A%2F%2Fspo337.com
http://www.kidscat.ch/linkoutphp/o.php?out=https%3A%2F%2Fspo337.com
https://www.kirschenmarkt-gladenbach.de/go.php?go=https%3A%2F%2Fspo337.com
https://www.manacomputers.com/redirect.php?blog=%E0%B8%A1%E0%B8%B2%E0%B8%99%E0%B8%B2%E0%B8%84%E0%B8%AD%E0%B8%A1%E0%B8%9E%E0%B8%B4%E0%B8%A7%E0%B9%80%E0%B8%95%E0%B8%AD%E0%B8%A3%E0%B9%8C&url=https%3A%2F%2Fspo337.com
https://gogvo.com/redir.php?url=https%3A%2F%2Fspo337.com
http://jbbs.shitaraba.net/bbs/link.cgi?url=https%3A%2F%2Fspo337.com
https://www.hfmmagazine.com/user/logout?referer=https%3A%2F%2Fspo337.com
https://top.hange.jp/linkdispatch/dispatch?targetUrl=https%3A%2F%2Fspo337.com
https://camberwellpark-manchester.secure-dbprimary.com/manchester/primary/camberwellpark/arenas/schoolwebsite/calendar/calendar?backto=https%3A%2F%2Fspo337.com
https://www.foodengineeringmag.com/gdpr-policy?url=https%3A%2F%2Fwww.foodengineeringmag.com%2Fuser%2Fpostlogin%3Fredirect%3Dhttps%3A%2F%2Fspo337.com
http://come-on.rdy.jp/wanted/cgi-bin/rank.cgi?mode=link&id=9066&url=https%3A%2F%2Fspo337.com
https://kudago.com/go/?to=https%3A%2F%2Fspo337.com
https://sylt.pennergame.de/redirect/?site=https%3A%2F%2Fspo337.com
http://www.isuperpage.co.kr/kwclick.asp?id=senplus&url=https%3A%2F%2Fspo337.com
https://desarrolloruralysostenibilidad.dip-badajoz.es/ir.php?url=https%3A%2F%2Fspo337.com&d=eyJ0YWJsYSI6InByb3llY3Rvc192aXNpdGFzIiwiY2FtcG9SZWwiOiJpZFByb3llY3RvIiwidmFsb3IiOiIyNiJ9
https://catalogue.ceda.ac.uk/uuid/6ba75d14b4159cb5f1daec7a3f2472f6?jump=related-anchor&search_url=https%3A%2F%2Fspo337.com
http://www.gses.tyc.edu.tw/dyna/netlink/hits.php?id=153&url=https%3A%2F%2Fspo337.com
https://id.dpa-system.dk/Home/Culture?culture=en&returnurl=https%3A%2F%2Fspo337.com
https://vcc.iljmp.com/1/f-00163?lp=https%3A%2F%2Fspo337.com
https://sync.adtelligent.com/csync?redir=https%3A%2F%2Fspo337.com
https://www.ighome.com/Redirect.aspx?url=https%3A%2F%2Fspo337.com
https://mudcat.org/link.cfm?url=https%3A%2F%2Fspo337.com
http://d-click.sociesc.org.br/u/20840/36/829763/103_0/4b7fb/?url=https%3A%2F%2Fspo337.com
https://hudsonltd.com/?URL=https%3A%2F%2Fspo337.com
http://admin.kpsearch.com/active/admin/customer/customer_email1_birthday.asp?item=&chname=gnc&strhomeurl=https%3A%2F%2Fspo337.com
https://www.play.net/bounce/redirect.asp?URL=https%3A%2F%2Fspo337.com
http://ten.rash.jp/muryou/ys4/rank.cgi?mode=link&id=72&url=https%3A%2F%2Fspo337.com
http://mailstat.us/tr/t/la7sfb3srlik9hzemvgrw/c/https%3A%2F%2Fspo337.com
http://vancouver-webpages.com/cgi-bin/node-info?https%3A%2F%2Fspo337.com
https://www.ehso.com/ehsord.php?URL=https%3A%2F%2Fspo337.com
https://php-api.engageya.com/oper/https_redirect.php?url=https%3A%2F%2Fspo337.com/
http://www.iatn.net/redirect?url=https%3A%2F%2Fspo337.com/
https://www.chicagolandchamber.org/LinkClick.aspx?link=https%3A%2F%2Fspo337.com/
https://berlin.pennergame.de/redirect/?site=https%3A%2F%2Fspo337.com/
https://muenchen.pennergame.de/redirect/?site=https%3A%2F%2Fspo337.com/
http://click.app4mobile-services.biz/storelink/?url=https%3A%2F%2Fspo337.com/
http://riotits.net/cgi-bin/a2/out.cgi?id=121&l=top4&u=https%3A%2F%2Fspo337.com/
http://d-click.vxcontact.com/u/2012/508/68946/1671_0/3626c/?url=https%3A%2F%2Fspo337.com/
http://www.funds-sp.jp/link/link.php?url=https%3A%2F%2Fspo337.com/
http://golfy.jp/log/log.php?id=1&obj_id=16&url=https%3A%2F%2Fspo337.com/
https://element.lv/go?url=https%3A%2F%2Fspo337.com/
http://www.bquest.org/Links/Redirect.aspx?ID=164&url=https%3A%2F%2Fspo337.com/
https://ukrainochka.ua/go.php?to=https%3A%2F%2Fspo337.com/
http://www.mishizhuti.com/114/export.php?url=https%3A%2F%2Fspo337.com/
http://navigate.ims.ca/default.aspx?id=1211260&mailingid=37291&redirect=https%3A%2F%2Fspo337.com/
https://prod1.airage.com/cirrata/www/delivery/ck.php?ct=1&oaparams=2__bannerid=150__zoneid=50__cb=27f996991c__oadest=https%3A%2F%2Fspo337.com/
http://biyougeka.esthetic-esthe.com/rank.cgi?mode=link&id=848&url=https%3A%2F%2Fspo337.com/
http://www.brownsberrypatch.farmvisit.com/redirect.jsp?urlr=https%3A%2F%2Fspo337.com/
https://www.floridafilmofficeinc.com/?goto=https%3A%2F%2Fspo337.com/
http://www.hyzsh.com/link/link.asp?id=10&url=https%3A%2F%2Fspo337.com/
http://www.imxyd.com/urlredirect.php?go=https%3A%2F%2Fspo337.com/
https://app.newsatme.com/emt/ses/814/33cgGz8Ee6U4F8kmECGeHZDSDgbTFjgWU231be54/click?url=https%3A%2F%2Fspo337.com/
http://w-ecolife.com/feed2js/feed2js.php?src=https%3A%2F%2Fspo337.com/
https://webreel.com/api/1/click?url=https%3A%2F%2Fspo337.com/
https://www.shihou-syoshi.jp/details/linkchk.aspx?type=p&url=https%3A%2F%2Fspo337.com/
http://www.redeletras.com.ar/show.link.php?url=https%3A%2F%2Fspo337.com/
https://union.diexun.com/market/?action=click&area=A-h-02-b&id=561&url=https%3A%2F%2Fspo337.com/
https://www.fortrucker-env.com/leaving.aspx?ext=https%3A%2F%2Fspo337.com/
https://www.livecmc.com/?lang=fr&id=Ld9efT&url=https%3A%2F%2Fspo337.com/
https://www.ittrade.cz/redir.asp?WenId=107&WenUrllink=https%3A%2F%2Fspo337.com/
http://www.cnainterpreta.it/redirect.asp?url=https%3A%2F%2Fspo337.com/
http://m.shopinlasvegas.net/redirect.aspx?url=https%3A%2F%2Fspo337.com/
https://www.voxlocalis.net/enlazar/?url=https%3A%2F%2Fspo337.com/
http://m.shopinboston.com/redirect.aspx?url=https%3A%2F%2Fspo337.com/
http://m.shopinsanjose.com/redirect.aspx?url=https%3A%2F%2Fspo337.com/
http://m.shopinsacramento.com/redirect.aspx?url=https%3A%2F%2Fspo337.com/
http://m.shopinraleigh.com/redirect.aspx?url=https%3A%2F%2Fspo337.com/
http://ra-blog.net/outgoing.php?url=https%3A%2F%2Fspo337.com/
http://m.shopinphilly.com/redirect.aspx?url=https%3A%2F%2Fspo337.com/
http://bbs.diced.jp/jump/?t=https%3A%2F%2Fspo337.com/
https://aritc.yru.ac.th/redirect/78?url=https%3A%2F%2Fspo337.com/
https://bookbuzzr.com/trackviews.php?action=buy&bookid=16363&buylink=https%3A%2F%2Fspo337.com/
http://jpn1.fukugan.com/rssimg/cushion.php?url=www.spo337.com/product/usa-email-database/
http://www.ixawiki.com/link.php?url=https%3A%2F%2Fspo337.com/
http://www.arcadepod.com/games/gamemenu.php?id=2027&name=Idiot%27s+Delight+Solitaire+Games&url=https%3A%2F%2Fspo337.com/
http://www.mech.vg/gateway.php?url=https%3A%2F%2Fspo337.com/
http://www.arch.iped.pl/artykuly.php?id=1&cookie=1&url=https%3A%2F%2Fspo337.com/
http://click.mobile.conduit-services.com/storeLink/?url=https%3A%2F%2Fspo337.com/
https://www.auburnapartmentguide.com/MobileDefault.aspx?reff=https%3A%2F%2Fspo337.com/
https://contractorsupplymagazine.com/tracking/tracking_link.php?e=info@dpxsystems.com&s=688&u=3576&a=724&r=https%3A%2F%2Fspo337.com/
http://www.findingfarm.com/redir?url=https%3A%2F%2Fspo337.com/
http://www.fmisrael.com/Error.aspx?url=https%3A%2F%2Fspo337.com/
http://www.e-tsuyama.com/cgi-bin/jump.cgi?jumpto=https%3A%2F%2Fspo337.com/
http://www.humaniplex.com/jscs.html?hj=y&ru=https%3A%2F%2Fspo337.com/
http://www.dresscircle-net.com/psr/rank.cgi?mode=link&id=14&url=https%3A%2F%2Fspo337.com/
http://sv2.deqwas.net/edge/codezine/standard/item/4742/choose?destination=https%3A%2F%2Fspo337.com/
https://www.tricitiesapartmentguide.com/MobileDefault.aspx?reff=https%3A%2F%2Fspo337.com/
http://banatanama.ir/banatanama.ir/viewpage.aspx?url=https%3A%2F%2Fspo337.com/
https://www.f5uii.net/link.php?site=https%3A%2F%2Fspo337.com/
http://www.ssnote.net/link?q=https%3A%2F%2Fspo337.com/student-database
http://www.infohelp.com/infohelp/jump.php?url=https%3A%2F%2Fspo337.com/
https://www.lepetitcornillon.fr/externe.php?site=https%3A%2F%2Fspo337.com/
http://www.fullerccim.com/Dot_EmailFriend.asp?referURL=https%3A%2F%2Fspo337.com/
https://www.sharps.se/redirect?url=https%3A%2F%2Fspo337.com/
https://seafood.media/fis/shared/redirect.asp?banner=6158&url=https%3A%2F%2Fspo337.com/
http://sc25.com/log_viewing.php?id=374&type=source&url=https%3A%2F%2Fspo337.com/
http://www.toku-jp.com/Rouge/minibbs.cgi?https%3A%2F%2Fspo337.com/
https://www.odeki.de/bw/redirect?external=https%3A%2F%2Fspo337.com/
http://yoshio.noizm.com/jump.php?u=https%3A%2F%2Fspo337.com/
http://enews2.sfera.net/newsletter/redirect.php?id=luigi.bottazzi@libero.it_0000004670_73&link=https%3A%2F%2Fspo337.com/
http://webredirect.garenanow.com/?p=gp&lang=en&url=https%3A%2F%2Fspo337.com/
http://www.irwebcast.com/cgi-local/report/redirect.cgi?url=https%3A%2F%2Fspo337.com/
http://ch1.artemisweb.jp/linkout.cgi?url=https%3A%2F%2Fspo337.com/
http://performance-appraisals.org/appraisal-library/topframe2014.php?goto=https%3A%2F%2Fspo337.com/
http://www.burstek.com/RedirectPage.php?reason=4&value=Anonymizers&proctoblocktimeout=1&ip=89.78.118.181&url=https%3A%2F%2Fspo337.com/
https://blog.hybridhealth-shinjuku.jp/?wptouch_switch=desktop&redirect=https%3A%2F%2Fspo337.com
https://hometutorbd.com/goto.php?directoryid=201&href=https%3A%2F%2Fspo337.com
https://jobatron.com/jobclick/?RedirectURL=https%3A%2F%2Fspo337.com
http://www.node-1.net/cgi-bin/cgi-local/bhi_extlinkclicktocntl.cgi?https%3A%2F%2Fspo337.com
https://darulifta.info/ask/to_dar_ask_url/?dar_id=1&url=https%3A%2F%2Fspo337.com
http://kentuckyheadhunters.net/gbook/go.php?url=https%3A%2F%2Fspo337.com
http://www.muppetsauderghem.be/?URL=spo337.com
http://sportwelt.log.checkeffect.at/da/?f=pclick&u=sportwelt&tkn=BFC393C7-4148-4CB1-AA7A-2519D80CBCC1&tknt=1&redirect=https%3A%2F%2Fspo337.com
http://kokubunsai.fujinomiya.biz/cgi/acc/acc.cgi?REDIRECT=https%3A%2F%2Fspo337.com
https://www.abcplus.biz/cartform.aspx?returnurl=https%3A%2F%2Fspo337.com
http://azy.com.au/index.php/goods/index/golink?url=https%3A%2F%2Fspo337.com
https://www.hayward-pool.ca/webapp/wcs/stores/servlet/CompareProductsDisplayView?storeId=11201&catalogId=11551&langId=-2&compareReturnName=&searchTerm=&returnUrl=https%3A%2F%2Fspo337.com
http://www.burgenkunde.at/links/klixzaehler.php?url=https%3A%2F%2Fspo337.com
http://tiroldorfsee.log.checkeffect.at/da/?f=pclick&u=tiroldorfsee&tkn=B0B61998-29E6-4432-8718-9F23AC27463E&tknt=1&redirect=https%3A%2F%2Fspo337.com
https://www.tppm.by/bitrix/redirect.php?event1=&event2=&event3=&goto=https%3A%2F%2Fspo337.com
http://hotelsaccommodation.com.au/centralsite/redirect.asp?dest=https%3A%2F%2Fspo337.com
http://pinki.nbbs.biz/kusyon.php?url=https%3A%2F%2Fspo337.com
http://my.effairs.at/austriatech/link/t?i=2504674541756&v=0&c=anonym&e=anonym@anonym.at&href=https%3A%2F%2Fspo337.com
http://www.trasportopersone.it/redirect.aspx?url=https%3A%2F%2Fspo337.com
http://ekonomka.dn.ua/out.php?link=https%3A%2F%2Fspo337.com
https://www.acecontrol.biz/link.php?u=https%3A%2F%2Fspo337.com
http://www.fsg-zihlschlacht.ch/sponsoren/sponsoren-weiter.asp?name=RaiffeisenbankZihlschlacht-Muolen-Bischofszell&url=https%3A%2F%2Fspo337.com
http://wal-land.cn/ucenter_home/link.php?url=https%3A%2F%2Fspo337.com
http://www.pingfarm.com/index.php?action=ping&urls=https%3A%2F%2Fspo337.com
https://www.vermont.com/linkclickcounts.cfm?linksId=6287&url=https%3A%2F%2Fspo337.com
http://www.dauntless-soft.com/products/android/beforeyougo.asp?U=https%3A%2F%2Fspo337.com
http://www.tstz.com/link.php?url=https%3A%2F%2Fspo337.com
http://ad.eanalyzer.de/10008728?url=https%3A%2F%2Fspo337.com
http://japan.road.jp/navi/navi.cgi?jump=226&url=https%3A%2F%2Fspo337.com
https://www.azlawhelp.org/externalsite.cfm?url=https%3A%2F%2Fspo337.com
https://www.naturum.co.jp/ad/linkshare/?siteID=p_L785d6UQY-V4Fh4Rxs7wNzOPgtzv95Tg&lsurl=https%3A%2F%2Fspo337.com
https://auth.she.com/logout/?client_id=8&callback=https%3A%2F%2Fspo337.com
http://enseignants.flammarion.com/Banners_Click.cfm?ID=86&URL=spo337.com
http://mlc.vigicorp.fr/link/619-1112492/?link=https%3A%2F%2Fspo337.com
https://www.dltk-teach.com/p.asp?p=https%3A%2F%2Fspo337.com
https://www.benissa.net/boletines/redir?cod_bol=CODENVBOLETIN&dir=spo337.com
https://movil.todorelatos.com/url.php?q=https%3A%2F%2Fspo337.com
https://fachowiec.com/zliczanie-bannera?id=24&url=https%3A%2F%2Fspo337.com
http://furusato-kirishima.com/cutlinks/rank.php?url=https%3A%2F%2Fspo337.com
https://slashwrestling.com/cgi-bin/redirect.cgi?https%3A%2F%2Fspo337.com
https://edusearch.ir/Goto.aspx?url=https%3A%2F%2Fspo337.com
https://ichi-up.net/finish?experiment=END_BANNER_DIGI&url=https%3A%2F%2Fspo337.com
https://morelia.estudiantil.mx/redirect?url=https%3A%2F%2Fspo337.com
https://lavery.sednove.com/extenso/module/sed/directmail/fr/tracking.snc?u=W5PV665070YU0B&url=https%3A%2F%2Fspo337.com
https://bza.co/dt-click.php?id=160&url=https%3A%2F%2Fspo337.com
https://maned.com/scripts/lm/lm.php?tk=CQkJZWNuZXdzQGluZm90b2RheS5jb20JW05ld3NdIE1FSSBBbm5vdW5jZXMgUGFydG5lcnNoaXAgV2l0aCBUd2l4bCBNZWRpYQkxNjcyCVBSIE1lZGlhIENvbnRhY3RzCTI1OQljbGljawl5ZXMJbm8=&url=https%3A%2F%2Fspo337.com
https://regie.hiwit.org/clic.cgi?id=1&zoned=a&zone=5&url=https%3A%2F%2Fspo337.com
https://comingoutspb.com/bitrix/redirect.php?event1=&event2=&event3=&goto=https%3A%2F%2Fspo337.com
https://search.mrcpl.org/webbridge~S19*eng/showresource?resurl=https%3A%2F%2Fspo337.com&linkid=15922799&noframe=1
https://access.bridges.com/externalRedirector.do?url=spo337.com
https://thairesidents.com/l.php?b=85&p=2,5&l=https%3A%2F%2Fspo337.com
https://www.joeshouse.org/booking?link=https%3A%2F%2Fspo337.com&ID=1112
https://cas.mon-ent-occitanie.fr/login?service=https%3A%2F%2Fspo337.com&gateway=true
https://gfb.gameflier.com/func/actionRewriter.aspx?pro=http&url=spo337.com
https://www.mendocino.com/?id=4884&url=spo337.com
https://sso.uic.fr/cas/login?service=https%3A%2F%2Fspo337.com&gateway=true
https://www.latestnigeriannews.com/link_channel.php?channel=https%3A%2F%2Fspo337.com
https://pdhonline.com/cgi-bin/quiz/refersite/refersite.cgi?refer_site_name=AAEE&site_url=spo337.com%20
https://affiliate.asknow.com/adservice/click/2806167/spo337.com
https://d.cungcap.net/d/spo337.com
https://www.crmsoftwareblog.com/flow/post_click.php?bid=1&pid=24970&destination=https%3A%2F%2Fspo337.com
https://gazetablic.com/ads/www/delivery/ck.php?ct=1&oaparams=2__bannerid=34__zoneid=26__cb=0e0dfef92b__oadest=https%3A%2F%2Fspo337.com
https://barcelo.ingenioustech.biz/ts/i2879670/tsc?amc=IGTtrack&trg=spo337.com/
https://travel98.com/redirect.php?url=https%3A%2F%2Fspo337.com
https://pdcn.co/e/spo337.com/
https://www.nexgam.de/ref.php?nxref=amazon&nxlink=https%3A%2F%2Fspo337.com
https://oversea.cnki.net/kcms/logout.aspx?url=https%3A%2F%2Fspo337.com
https://www.sign-in-china.com/newsletter/statistics.php?type=mail2url&bs=88&i=114854&url=https%3A%2F%2Fspo337.com
http://www.priegeltje.nl/gastenboek/go.php?url=https%3A%2F%2Fspo337.com
https://kabu-sokuhou.com/redirect/head/?u=https%3A%2F%2Fspo337.com
https://www.trackeame.com/sem-tracker-web/track?kw=14270960094&c=1706689156&mt=p&n=b&u=https%3A%2F%2Fspo337.com
http://miyagi.lawyer-search.tv/details/linkchk.aspx?type=o&url=https%3A%2F%2Fspo337.com
https://bigjobslittlejobs.com/jobclick/?RedirectURL=https%3A%2F%2Fspo337.com&Domain=bigjobslittlejobs.com&rgp_m=title23&et=4495
https://communicatedcareers.com/jobclick/?RedirectURL=https%3A%2F%2Fspo337.com
http://www.brillkids.com/ra.php?id=BKAFF64558&s=spo337.com
https://mobile.thomasandfriends.jp/TRF001/?url=https%3A%2F%2Fspo337.com
https://wasitviewed.com/index.php?href=https%3A%2F%2Fspo337.com
https://saht.mobi/Profile/Login?returnUrl=https%3A%2F%2Fspo337.com
https://reson-ltd.co.jp/navi/navi.cgi?&mode=jump&id=0009&url=spo337.com
https://s-p.me/template/pages/station/redirect.php?url=https%3A%2F%2Fspo337.com
http://craftbeverageinsights.com/jump.php?url=https%3A%2F%2Fspo337.com
http://ww.thesteelbrothers.com/buy.php?store=iBooks&url=https%3A%2F%2Fspo337.com
http://thdt.vn/convert/convert.php?link=https%3A%2F%2Fspo337.com
http://wvw.aldia.cr/servicios/phps/load2.php?url=https%3A%2F%2Fspo337.com
http://www.nigeriannewspapersonline.net/cgi-bin/redirect.pl?link=https%3A%2F%2Fspo337.com
http://demo.logger.co.kr/source.php?url=https%3A%2F%2Fspo337.com
https://click.fitminutes.com/?prod_id=-2924339127316720883&psid=136&auth=Hrbpx&kw=&env=2&subid=organic_fitminutes_us_blog&fct=true&passback=https%3A%2F%2Fspo337.com
http://www.doitweb365.de/scripts/doitweb.exe/rasklickzaehler2?https%3A%2F%2Fspo337.com
https://www.guadamur.eu/template/pages/station/redirect.php?url=https%3A%2F%2Fspo337.com
https://uk.kindofbook.com/redirect.php/?red=https%3A%2F%2Fspo337.com
http://www.mckinneyfarm.com/template/plugins/stationExtremes/redirect.php?url=https%3A%2F%2Fspo337.com
http://search.pointcom.com/k.php?ai=&url=https%3A%2F%2Fspo337.com
http://www.cercasostituto.it/index.php?name=GestBanner&file=counter&idbanner=40&dir_link=https%3A%2F%2Fspo337.com
http://31.gregorinius.com/index/d1?diff=0&source=og&campaign=4397&content=&clickid=hrx9nw9psafm4g9v&aurl=https%3A%2F%2Fspo337.com&an=&term=&site=&darken=1#
https://www.rsedatanews.net/amp?url=https%3A%2F%2Fspo337.com
http://1.dranationius.com/index/c1?diff=1&source=og&campaign=17149&content=&clickid=sxyfhidcjh3bqphk&aurl=https%3A%2F%2Fspo337.com&an=&term=&site=&darken=1&allFull=0&isubs=1#
http://www.meteo-leran.fr/meteotemplate/template/plugins/deviations/redirect.php?url=https%3A%2F%2Fspo337.com
https://www.eurobichons.com/fda%20alerts.php?url=https%3A%2F%2Fspo337.com
https://mydojo.at/de_AT/karate/weiterleitung?redirect=https%3A%2F%2Fspo337.com
http://click.phosphodiesterase4.com/k.php?ai=&url=https%3A%2F%2Fspo337.com
https://tneahelp.in/redirect.php?l=https%3A%2F%2Fspo337.com
http://www.mariahownersclub.com/forum/redirect-to/?redirect=https%3A%2F%2Fspo337.com
http://www.wildromance.com/buy.php?url=https%3A%2F%2Fspo337.com&store=iBooks&book=omk-ibooks-us
http://mapleriverweather.com/mobile/pages/station/redirect.php?url=https%3A%2F%2Fspo337.com
http://www.archijob.co.il/index/comp_website.asp?companyId=1469&website=https%3A%2F%2Fspo337.com
https://student-helpr.rminds.dev/redirect?redirectTo=https%3A%2F%2Fspo337.com
http://www.airnav.com/depart?https%3A%2F%2Fspo337.com
https://3db.moy.su/go?https%3A%2F%2Fspo337.com
https://bnc.lt/a/key_live_pgerP08EdSp0oA8BT3aZqbhoqzgSpodT?medium=&feature=&campaign=&channel=&$always_deeplink=0&$fallback_url=spo337.com&$deeplink_path=&p=c11429c2860165eee314
http://ad.gunosy.com/pages/redirect?location=https%3A%2F%2Fspo337.com
http://goldankauf-engelskirchen.de/out.php?link=https%3A%2F%2Fspo337.com
https://com7.jp/ad/?https%3A%2F%2Fspo337.com
http://devicedoctor.com/driver-feedback.php?device=PCI%20bus&url=https%3A%2F%2Fspo337.com
https://www.mega-show.com/redirect-nonssl.php?sslurl=https%3A%2F%2Fspo337.com
https://www.surinenglish.com/backend/conectar.php?url=https%3A%2F%2Fspo337.com
https://loadus.exelator.com/load/?p=258&g=244&clk=1&crid=porscheofnorth&stid=rennlist&j=r&ru=https%3A%2F%2Fspo337.com
http://www.mastermason.com/MakandaLodge434/guestbook/go.php?url=https%3A%2F%2Fspo337.com
https://flamingo.moy.su/go?https%3A%2F%2Fspo337.com
http://t.raptorsmartadvisor.com/.lty?url=https%3A%2F%2Fspo337.com
https://www.misadventures.com/buy.php?store=Kindle&url=https%3A%2F%2Fspo337.com
http://pharmacist-job-hikakuandsearch.net/cta/r.php?link=https%3A%2F%2Fspo337.com
http://wetter.wassersport-warendorf.de/meteotemplate/pages/station/redirect.php?url=https%3A%2F%2Fspo337.com
http://redirect.sgtips.com/redirect.php?url=https%3A%2F%2Fspo337.com
http://www.experty.com/l.php?u=https%3A%2F%2Fspo337.com
http://sanmariano.lineameteo.it/plugins/stationExtremes/redirect.php?url=https%3A%2F%2Fspo337.com/
https://pvtistes.net/forum/redirect-to/?redirect=https%3A%2F%2Fspo337.com/
https://www.katholische-sonntagszeitung.de/anzeigen_redirect.php?name=Schnitzerei%20Schinner%20-%20Osterkrippen&target=https%3A%2F%2Fspo337.com
http://inquiry.princetonreview.com/away/?value=cconntwit&category=FS&url=https%3A%2F%2Fspo337.com
http://www.fimmgcagliari.org/index.php?name=GestBanner&file=counter&idbanner=28&dir_link=https%3A%2F%2Fspo337.com
http://www.fimmgviterbo.org/mobfimmgviterbo/index.php?nametm=counter&idbanner=4&dir_link=https%3A%2F%2Fspo337.com
http://bjornagain.com.au/redirect.php?location=https%3A%2F%2Fspo337.com
http://www.konto-testsieger.de/goto/abgelehnt/beratung/?url=https%3A%2F%2Fspo337.com/
http://www.goformore.ca/fr/commerciaux/includes/redirector.php?strURL=https%3A%2F%2Fspo337.com/
http://www.lilyandtheduke.com/buy.php?url=https%3A%2F%2Fspo337.com/&store=iBooks
http://www.bitded.com/redir.php?url=https%3A%2F%2Fspo337.com
http://vejr.arloese.dk/template/plugins/deviations/redirect.php?url=https%3A%2F%2Fspo337.com
https://server-system.jp/nordson/redirect.php?targeturl=https%3A%2F%2Fspo337.com&title=%E3%83%8E%E3%83%BC%E3%83%89%E3%82%BD%E3%83%B3%E5%90%91%E3%81%91%E6%83%85%E5%A0%B1
http://www.kontoexperte.de/goto/tagesgeld/?url=https%3A%2F%2Fspo337.com
http://www.friedo.nl/template/pages/station/redirect.php?url=https%3A%2F%2Fspo337.com
https://forum.gsmhosting.com/vbb/redirect-to/?redirect=https%3A%2F%2Fspo337.com
https://dento.itot.jp/ref/?bnrno=03&url=https%3A%2F%2Fspo337.com
http://www.exactshot.at/redir.php?url=https%3A%2F%2Fspo337.com
https://www.ferrol.gal/educacion/visor_pdf.aspx?url_pdf=https%3A%2F%2Fspo337.com
https://ao-inc.com/?URL=https%3A%2F%2Fspo337.com
https://www.woodworker.de/?URL=https%3A%2F%2Fspo337.com
https://www.kranten.com/redirect/nd.html?u=https%3A%2F%2Fspo337.com
http://www.artistar.it/ext/topframe.php?link=https%3A%2F%2Fspo337.com
http://networksolutionssux.com/media/js/netsoltrademark.php?d=spo337.com
http://local.rongbachkim.com/rdr.php?url=https%3A%2F%2Fspo337.com
https://news.myseldon.com/away?to=https%3A%2F%2Fspo337.com
http://bachecauniversitaria.it/link/frm_top.php?url=https%3A%2F%2Fspo337.com
https://www.stcwdirect.com/redirect.php?url=https%3A%2F%2Fspo337.com
https://finanzplaner-deutschland.de/fpdeu/index.asp?source=/fpdeu/inc/mitglieder_form.asp@nr=24@referer=https%3A%2F%2Fspo337.com
http://forum.vcoderz.com/externalredirect.php?url=https%3A%2F%2Fspo337.com
https://www.momentumstudio.com/?URL=https%3A%2F%2Fspo337.com
http://90.gregorinius.com/index/d1?diff=0&source=og&campaign=5796&content=&clickid=6glaagrcny71ype6&aurl=https%3A%2F%2Fspo337.com
http://28.restonovius.com/index/s1?diff=1&source=og&campaign=16004&content=somedude3&clickid=m7nz4apsasighm85&aurl=https%3A%2F%2Fspo337.com&an=&term=6876&site=
http://accglobal.net/fr/commerciaux/includes/redirector.php?strURL=https%3A%2F%2Fspo337.com
http://25.quarenafius.com/index/s1?diff=0&source=og&campaign=16004&content=rediskin&clickid=opcg4radtqjz1bgu&aurl=https%3A%2F%2Fspo337.com&an=&term=5353&site
http://1.dranationius.com/index/c1?diff=1&source=og&campaign=17149&content=&clickid=sxyfhidcjh3bqphk&aurl=https%3A%2F%2Fspo337.com&an=&term=&si
https://misechko.com.ua/go?url=spo337.com
http://click.localpages.com/k.php?ai=9788&url=https%3A%2F%2Fspo337.com
http://massimopoletti.altervista.org/template/pages/station/redirect.php?url=https%3A%2F%2Fspo337.com
http://30.crouchserf.com/index/c3?diff=0&source=og&campaign=16004&content=&clickid=lqnt8jsq37o93l3p&aurl=https%3A%2F%2Fspo337.com&an=o
http://www.week.co.jp/skion/cljump.php?clid=129&url=https%3A%2F%2Fspo337.com
http://go.e-frontier.co.jp/rd2.php?uri=https%3A%2F%2Fspo337.com
https://forum.419eater.com/forum/ref.php?url=https%3A%2F%2Fspo337.com
http://dineview.com/redirect.fwx?type=menu&id=R068134&url=https%3A%2F%2Fspo337.com
http://images.ttacorp.com/linktracker.aspx?u=https%3A%2F%2Fspo337.com
http://career-first.net/?page=2&board=QJF&load-url=https%3A%2F%2Fspo337.com
http://urls.tsa.2mes4.com/amazon_product.php?ASIN=B07211LBSP&page=10&url=https%3A%2F%2Fspo337.com
http://boltsaga.com/buy.php?book=bolt-volume-1&store=Waterstones&url=https%3A%2F%2Fspo337.com/
http://foreneset.no/template/plugins/windDirection/redirect.php?url=https%3A%2F%2Fspo337.com/
http://ssearch.jp/books/amazonUS.php?url=https%3A%2F%2Fspo337.com
https://www.gazzettadellevalli.it/gdv/advredirect.php?url=https%3A%2F%2Fspo337.com
http://bolt-saga.com/buy.php?url=https%3A%2F%2Fspo337.com/&store=iBooks&book=bolt-volume-1-ibooks-us
http://link03.net/redirect.cgi?url=https%3A%2F%2Fspo337.com
https://www.thesamba.com/vw/bin/banner_click.php?redirect=spo337.com
http://meteo-cugy.ch/template/plugins/deviations/redirect.php?url=https%3A%2F%2Fspo337.com
http://cryptocurrency-hikakuandsearch.net/cta/r.php?link=https%3A%2F%2Fspo337.com
http://craftbeverageinsight.com/jump.php?url=https%3A%2F%2Fspo337.com/
http://www.oraichi.com/link/?url=https%3A%2F%2Fspo337.com
http://www.doitweb.de/scripts/doitweb.exe/rasklickzaehler?https%3A%2F%2Fspo337.com
http://spillarkivet.no/i.php?url=https%3A%2F%2Fspo337.com/
https://stara.biblioteka.jelenia-gora.pl/dalej.php?adres=https%3A%2F%2Fspo337.com
https://www.uia.no/linktools/redirect?url=https%3A%2F%2Fspo337.com
http://meteo-villers-bretonneux.fr/meteo_template/template/pages/station/redirect.php?url=https%3A%2F%2Fspo337.com
http://www.zanzana.net/goto.asp?goto=https%3A%2F%2Fspo337.com
https://shiftup.ca/view.aspx?Site=spo337.com
https://fresh-jobs.uk/click/click_site?url=https%3A%2F%2Fspo337.com
https://www.fnnews.com/redirect?url=https%3A%2F%2Fspo337.com&utm_ca
https://compedia.jp/conversion.php?type=official&url=https%3A%2F%2Fspo337.com
http://hai.byjeanne.com/member/login.html?noMemberOrder=&returnUrl=https%3A%2F%2Fspo337.com
http://hampus.biz/?URL=spo337.com
http://hatenablog-parts.com/embed?url=https%3A%2F%2Fspo337.com
http://hcr233.azurewebsites.net/url?q=https%3A%2F%2Fspo337.com
http://hipposupport.de/url?q=https%3A%2F%2Fspo337.com
http://historisches-festmahl.de/go.php?url=https%3A%2F%2Fspo337.com
http://hockey-now.stage.publishwithagility.com/account/logout?returnUrl=https%3A%2F%2Fspo337.com
http://house.speakingsame.com/cn/floorplan.php?sta=vic&addr=91+arthurton+road&q=northcote&url=spo337.com
http://hramacek.de/url?q=https%3A%2F%2Fspo337.com
http://hufschlag-foto.de/gallery2/main.php?g2_view=core.UserAdmin&g2_subView=core.UserLogin&g2_return=https%3A%2F%2Fspo337.com
http://hydronics-solutions.com/bitrix/rk.php?goto=https%3A%2F%2Fspo337.com
http://icecap.us/?URL=spo337.com
http://ighaleb.ir/redirect/redirect.php?url=https%3A%2F%2Fspo337.com
http://ikonet.com/en/visualdictionary/static/us/blog_this?id=https%3A%2F%2Fspo337.com
http://imagelibrary.asprey.com/?URL=spo337.com
http://ime.nu/https%3A%2F%2Fspo337.com
http://informatief.financieeldossier.nl/index.php?url=https%3A%2F%2Fspo337.com
http://interflex.biz/url?q=https%3A%2F%2Fspo337.com
http://ivvb.de/url?q=https%3A%2F%2Fspo337.com
http://j.lix7.net/?https%3A%2F%2Fspo337.com
http://jacobberger.com/?URL=spo337.com
http://jahn.eu/url?q=https%3A%2F%2Fspo337.com
http://jamesvelvet.com/?URL=spo337.com
http://jamrefractory.com/default.aspx?key=4KOasVkDUpczQmigaUsZswe-qe-q&out=forgotpassword&sys=user&cul=fa-IR&returnurl=https%3A%2F%2Fspo337.com
http://jewelrybay.co.kr/member/login.html?noMemberOrder=&returnUrl=https%3A%2F%2Fspo337.com
http://jla.drmuller.net/r.php?url=https%3A%2F%2Fspo337.com
http://jump.pagecs.net/https%3A%2F%2Fspo337.com
http://karkom.de/url?q=https%3A%2F%2Fspo337.com
http://kenkyuukai.jp/event/event_detail_society.asp?id=52212&ref=calendar&rurl=https%3A%2F%2Fspo337.com
http://kens.de/url?q=https%3A%2F%2Fspo337.com
http://kikikifigure.com/member/login.html?noMemberOrder&returnUrl=https%3A%2F%2Fspo337.com
http://kinderundjugendpsychotherapie.de/url?q=https%3A%2F%2Fspo337.com
http://kinhtexaydung.net/redirect/?url=https%3A%2F%2Fspo337.com
http://www.kalinna.de/url?q=https%3A%2F%2Fspo337.com
http://www.hartmanngmbh.de/url?q=https%3A%2F%2Fspo337.com
https://www.the-mainboard.com/proxy.php?link=https%3A%2F%2Fspo337.com
https://www.betamachinery.com/?URL=https%3A%2F%2Fspo337.com
http://webradio.fm/webtop.cfm?site=https%3A%2F%2Fspo337.com
http://www.sprang.net/url?q=https%3A%2F%2Fspo337.com
http://www.insidearm.com/email-share/send/?share_title=MBNA%20to%20Acquire%20Mortage%20BPO%20Provider%20Nexstar&share_url=https%3A%2F%2Fspo337.com
https://img.2chan.net/bin/jump.php?https%3A%2F%2Fspo337.com
http://www.is.kyusan-u.ac.jp/htmllint/htmllint.cgi?ViewSource=on;URL=https%3A%2F%2Fspo337.com
http://sahakorn.excise.go.th/form_view_activity.php?new_id=NEW20170315185851&url=https%3A%2F%2Fspo337.com
https://forum.everleap.com/proxy.php?link=https%3A%2F%2Fspo337.com
https://secure.nationalimmigrationproject.org/np/clients/nationalimmigration/tellFriend.jsp?subject=Attending%202020+Annual+Pre-AILA+Crimes+and+Immigration+Virtual+CLE&url=https%3A%2F%2Fspo337.com
https://www.vsfs.cz/?id=1758&gal=216&img=15315&back=https%3A%2F%2Fspo337.com
http://www.mosig-online.de/url?q=https%3A%2F%2Fspo337.com
http://www.hccincorporated.com/?URL=https%3A%2F%2Fspo337.com
http://fatnews.com/?URL=https%3A%2F%2Fspo337.com
https://ruddingtongrange.com/?URL=https%3A%2F%2Fspo337.com
https://csirealty.com/?URL=https%3A%2F%2Fspo337.com
http://asadi.de/url?q=https%3A%2F%2Fspo337.com
http://treblin.de/url?q=https%3A%2F%2Fspo337.com
https://kentbroom.com/?URL=https%3A%2F%2Fspo337.com
http://0845.boo.jp/cgi/mt3/mt4i.cgi?id=24&mode=redirect&no=15&ref_eid=3387&url=https%3A%2F%2Fspo337.com
http://110.164.66.211/ULIB6//dublin.linkout.php?url=https%3A%2F%2Fspo337.com
http://110.164.92.12/ULIB//dublin.linkout.php?url=https%3A%2F%2Fspo337.com
http://198.54.125.86.myopenlink.net/describe/?url=https%3A%2F%2Fspo337.com
https://1st-p.jp/responsive-sample?url=https%3A%2F%2Fspo337.com
http://202.144.225.38/jmp?url=https%3A%2F%2Fspo337.com
http://2cool2.be/url?q=https%3A%2F%2Fspo337.com
http://39.farcaleniom.com/index/d2?diff=0&source=og&campaign=8220&content=&clickid=w7n7kkvqfyfppmh5&aurl=https%3A%2F%2Fspo337.com
http://4coma.net/cgi/mt4/mt4i.cgi?cat=12&mode=redirect&ref_eid=3231&url=https%3A%2F%2Fspo337.com
http://4travel.jp/dynamic/redirect.php?mode=dm_tour&url=https%3A%2F%2Fspo337.com
http://4vn.eu/forum/vcheckvirus.php?url=https%3A%2F%2Fspo337.com
http://hufschlag-foto.de/gallery2/main.php?g2view=core.UserAdmin&g2subView=core.UserLogin&g2return=https%3A%2F%2Fspo337.com
https://btng.org/tiki-tell_a_friend.php?url=https%3A%2F%2Fspo337.com
https://de.flavii.de/index.php?flavii=linker&link=https%3A%2F%2Fspo337.com
https://dbpedia.org/describe/?url=https%3A%2F%2Fspo337.com
https://boowiki.info/go.php?go=https%3A%2F%2Fspo337.com
https://www.otinasadventures.com/index.php?w_img=spo337.com
https://savvylion.com/?bmDomain=spo337.com
http://vivadoo.es/jump.php?idbd=2052&url=https%3A%2F%2Fspo337.com
http://www.country-retreats.com/cgi-bin/redirectpaid.cgi?URL=spo337.com
http://linkanalyse.durad.de/?ext_url=spo337.com
https://map.thai-tour.com/re.php?url=https%3A%2F%2Fspo337.com
https://familie-huettler.de/link.php?link=spo337.com
https://www.socializer.info/follow.asp?docurlf=https%3A%2F%2Fspo337.com
http://sameas.org/html?uri=https%3A%2F%2Fspo337.com
https://www.dynonames.com/buy-expired-or-pre-owned-domain-name.php?url=spo337.com
https://www.trainorders.com/discussion/warning.php?forum_id=1&url=https%3A%2F%2Fspo337.com
https://walkpittsburgh.org/?URL=https%3A%2F%2Fspo337.com
https://whois.zunmi.com/?d=spo337.com/cities%2Ftampa-fl%2F.com
http://panchodeaonori.sakura.ne.jp/feed/aonori/feed2js.php?src=https%3A%2F%2Fspo337.com
https://chofu.keizai.biz/banner.php?type=text_banner&position=right&id=3&uri=https%3A%2F%2Fspo337.com
https://clipperfund.com/?URL=https%3A%2F%2Fspo337.com
http://centre.org.au/?URL=https%3A%2F%2Fspo337.com
http://www.restaurant-zahnacker.fr/?URL=https%3A%2F%2Fspo337.com
https://sensationalsoy.ca/?URL=https%3A%2F%2Fspo337.com
https://www.myrtlebeachnational.com/?URL=https%3A%2F%2Fspo337.com
https://www.readconstruction.co.uk/?URL=https%3A%2F%2Fspo337.com
http://www.skoladesignu.sk/?URL=https%3A%2F%2Fspo337.com
https://logick.co.nz/?URL=https%3A%2F%2Fspo337.com
http://www.cafeteriatrend.hu/?URL=https%3A%2F%2Fspo337.com
http://bigline.net/?URL=https%3A%2F%2Fspo337.com
https://www.ticrecruitment.com/?URL=https%3A%2F%2Fspo337.com
http://aquaguard.com/?URL=https%3A%2F%2Fspo337.com
http://cim.bg/?URL=https%3A%2F%2Fspo337.com
https://www.aikenslake.com/?URL=https%3A%2F%2Fspo337.com
https://www.shinobi.jp/etc/goto.html?https%3A%2F%2Fspo337.com
http://www.addtoinc.com/?URL=spo337.com
http://0120-74-4510.com/redirect.php?program=medipa_orange_pc&rd=off&codename=&channel=&device=&url=https%3A%2F%2Fspo337.com
http://11.ernorvious.com/index/d1?diff=0&source=og&campaign=5944&content=&clickid=2aqzrzl2knl1pmit&aurl=ttps://spo337.com&an=&te=&pushMode=popup
http://1.glawandius.com/index/c2?diff=6&source=og&campaign=18410&content=kirill2005&clickid=tpg69ftnn9vtevf9&aurl=https%3A%2F%2Fspo337.com&an=&term=NCR
http://10.faranharbarius.com/index/c1?diff=0&source=og&campaign=16917&content=&clickid=9ymbp6hz0jpb0x49&aurl=https%3A%2F%2Fspo337.com&an=&term=&site=
http://80.inspiranius.com/index/l1?diff=9&source=og&campaign=8464&content=1627&clickid=l44a32xdmkttt9gt&aurl=https%3A%2F%2Fspo337.com&an=&term=&site=
http://happy-lands.com/bitrix/redirect.php?event1=&event2=&event3=&goto=https%3A%2F%2Fspo337.com
http://fr.knubic.com/redirect_to?url=https%3A%2F%2Fspo337.com
http://fewiki.jp/link.php?https%3A%2F%2Fspo337.com
https://www.grungejohn.com/bitrix/redirect.php?event1=&event2=&event3=&goto=https%3A%2F%2Fspo337.com
https://bio2rdf.org/describe/?url=https%3A%2F%2Fspo337.com
https://eve-search.com/externalLink.asp?l=https%3A%2F%2Fspo337.com
https://blaze.su/bitrix/redirect.php?event1=&event2=&event3=&goto=https%3A%2F%2Fspo337.com
https://rev1.reversion.jp/redirect?url=https%3A%2F%2Fspo337.com
http://markiza.me/bitrix/rk.php?goto=https%3A%2F%2Fspo337.com
https://cdn.iframe.ly/api/iframe?url=https%3A%2F%2Fspo337.com
https://bluecorkscrew.com/store/webdevelopment/tabid/522/ctl/compareitems/mid/1909/default.aspx?returnurl=https%3A%2F%2Fspo337.com
https://www.musicpv.jp/music.cgi?order=&class=&keyword=&FF=&price_sort=&pic_only=&mode=p_wide&id=11143&superkey=1&back=https%3A%2F%2Fspo337.com
https://www.bro-bra.jp/entry/kiyaku.php?url=https%3A%2F%2Fspo337.com
https://www.woodlist.us/delete-company?nid=13964&element=https%3A%2F%2Fspo337.com
https://www.bausch.co.nz/en-nz/redirect/?url=https%3A%2F%2Fspo337.com
https://www.funeralunion.org/delete-company?nid=39&element=https%3A%2F%2Fspo337.com
https://www.poringa.net/?go=https%3A%2F%2Fspo337.com
http://www.hon-cafe.net/cgi-bin/re.cgi?lid=hmw&url=https%3A%2F%2Fspo337.com
https://www.linkytools.com/basic_link_entry_form.aspx?link=entered&returnurl=https%3A%2F%2Fspo337.com&AspxAutoDetectCookieSupport=1
https://www.kikuya-rental.com/bbs/jump.php?url=https%3A%2F%2Fspo337.com
https://chipcart.shop/bitrix/redirect.php?event1=&event2=&event3=&goto=https%3A%2F%2Fspo337.com
https://www.steuerberaterinbruehl.de/ext_link?url=https%3A%2F%2Fspo337.com
https://www.sinara-group.com/bitrix/rk.php?goto=https%3A%2F%2Fspo337.com
https://ggurl.gdgdocs.org/url?q=https%3A%2F%2Fspo337.com
https://www.knipsclub.de/weiterleitung/?url=https%3A%2F%2Fspo337.com
https://www.bildungslandschaft-pulheim.de/redirect.php?url=https%3A%2F%2Fspo337.com
https://bangdream.gamerch.com/gamerch/external_link/?url=https%3A%2F%2Fspo337.com
https://www.morgeneyer.de/ahnen/login/default.aspx?returnurl=https%3A%2F%2Fspo337.com
https://belco.org/exit/?url=https%3A%2F%2Fspo337.com
https://www.office-mica.com/ebookmb/index.cgi?id=1&mode=redirect&no=49&ref_eid=587&url=https%3A%2F%2Fspo337.com
http://www.fouillez-tout.com/cgi-bin/redirurl.cgi?https%3A%2F%2Fspo337.com
http://mcclureandsons.com/Projects/FishHatcheries/Baker_Lake_Spawning_Beach_Hatchery.aspx?Returnurl=https%3A%2F%2Fspo337.com
https://account.eleavers.com/signup.php?user_type=pub&login_base_url=https%3A%2F%2Fspo337.com
http://www.zhaoyunpan.cn/transfer.php?url=https%3A%2F%2Fspo337.com
https://unikom.org/bitrix/redirect.php?event1=&event2=&event3=&goto=https%3A%2F%2Fspo337.com
http://talesofasteria.cswiki.jp/index.php?cmd=jumpto&r=https%3A%2F%2Fspo337.com
http://yahoo-mbga.jp/r?url=//spo337.com
http://www.sitedossier.com/site/spo337.com
http://daidai.gamedb.info/wiki/?cmd=jumpto&r=https%3A%2F%2Fspo337.com
https://jpn1.fukugan.com/rssimg/cushion.php?url=spo337.com
https://www.thaiall.com/cgi/clicko.pl?20819&spo337.com
https://yapy.jp/?F=contact&t=1&d=spo337.com&fc=FFFFFF&tc=C30046&hc=CCCCCC
https://mozakin.com/bbs-link.php?url=spo337.com
http://images.google.de/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://maps.google.be/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://maps.google.ru/url?sa=t&url=https%3A%2F%2Fspo337.com/
http://www.google.se/url?sa=t&source=web&cd=1&ved=0CBcQFjAA&url=https%3A%2F%2Fspo337.com/
https://www.google.se/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://maps.google.com.tr/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.dk/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.com.hk/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://images.google.com.mx/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.hu/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://images.google.com.au/url?q=https%3A%2F%2Fspo337.com/
https://maps.google.pt/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.co.nz/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202766&url=http://https%3A%2F%2Fspo337.com/
https://www.google.com.ar/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.co.th/url?sa=i&rct=j&q=&esrc=s&source=images&cd=&cad=rja&uact=8&ved=0ahUKEwjB4_-A3tnWAhWHOY8KHTcgDxMQjRwIBw&url=https%3A%2F%2Fspo337.com/
https://www.google.co.th/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.com.ua/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.no/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.co.za/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.ro/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://images.google.com.vn/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://guru.sanook.com/?URL=https%3A%2F%2Fspo337.com/
https://images.google.com.ph/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://maps.google.cl/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.ie/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://fjb.kaskus.co.id/redirect?url=https%3A%2F%2Fspo337.com/
https://maps.google.com.my/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.sk/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://images.google.com/url?sa=t&url=https%3A%2F%2Fspo337.com/
http://www.google.com.tw/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.co.il/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.rs/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://maps.google.com.ni/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.lt/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.ae/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.si/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://images.google.com.co/url?sa=t&url=https%3A%2F%2Fspo337.com/
http://www.google.fi/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://cse.google.fi/url?q=https%3A%2F%2Fspo337.com/
http://www.google.com.sg/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.hr/url?sa=t&url=https%3A%2F%2Fspo337.com/
http://images.google.co.nz/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://qatar.vcu.edu/?URL=https%3A%2F%2Fspo337.com/
https://images.google.com.pe/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.ee/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.lv/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.com.pk/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://maps.google.com.np/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://maps.google.co.ve/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.lk/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.com.bd/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://images.google.com.ec/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://images.google.by/url?sa=t&url=https%3A%2F%2Fspo337.com/
http://maps.google.cz/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.com.ng/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.lu/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://images.google.com.uy/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.co.cr/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://images.google.tn/url?sa=t&url=https%3A%2F%2Fspo337.com/
http://www.london.umb.edu/?URL=https%3A%2F%2Fspo337.com/
https://www.google.com.do/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://images.google.com.pr/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://ceskapozice.lidovky.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://www.google.ba/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.mu/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://images.google.co.ke/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.is/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://images.google.com.lb/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.com.gt/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://maps.google.com.py/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://maps.google.dz/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://images.google.cat/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.hn/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.com.mt/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.kz/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://images.google.com.kh/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://images.google.cm/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://ilns.ranepa.ru/bitrix/rk.php?goto=https%3A%2F%2Fspo337.com/
https://inva.gov.kz/ru/redirect?url=https%3A%2F%2Fspo337.com/
https://m.fishki.net/go/?url=https%3A%2F%2Fspo337.com/
https://www.rospotrebnadzor.ru/bitrix/redirect.php?event1=file&event2=download&event3=prilozheniya-k-prikazu-1018.doc&goto=https%3A%2F%2Fspo337.com/
http://76-rus.ru/bitrix/redirect.php?event1=catalog_out&event2=http2FEEECEBEEE3%EEEE-E5F1FBEDF0&goto=https%3A%2F%2Fspo337.com/
http://cenproxy.mnpals.net/login?url=https%3A%2F%2Fspo337.com/
http://oca.ucsc.edu/login?url=https%3A%2F%2Fspo337.com/
http://www.hyiphistory.com/visit.php?url=https%3A%2F%2Fspo337.com/
http://www.liveinternet.ru/journal_proc.php?action=redirect&url=https%3A%2F%2Fspo337.com/
https://old.fishki.net/go/?url=https%3A%2F%2Fspo337.com/
https://www.banki.ru/away/?url=https%3A%2F%2Fspo337.com/
https://www.institutoquinquelamartin.edu.ar/Administracion/top-10-cuadros-mas-famosos6-1/?unapproved=10807https%3A%2F%2Fspo337.com/
https://mcpedl.com/leaving/?url=https%3A%2F%2Fwww.statusvideosongs.in%2F&cookie_check=1https%3A%2F%2Fspo337.com/
http://sasisa.ru/forum/out.php?link=%3F&yes=1https%3A%2F%2Fspo337.com/
http://usolie.info/bitrix/redirect.php?event1=&event2=&event3=&goto=https%3A%2F%2Fspo337.com/
http://vstu.ru/bitrix/rk.php?goto=https%3A%2F%2Fspo337.com/
http://www.bsaa.edu.ru/bitrix/rk.php?goto=https%3A%2F%2Fspo337.com/
https://academy.1c-bitrix.ru/bitrix/redirect.php?event1=acsdemy&event2=usable&event3=&goto=https%3A%2F%2Fspo337.com/
https://adamb-bartos.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://adamburda.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://adammikulasek.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://adamvanek.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://adamvasina.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://agalarov.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://alenapekarova.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://alenapitrova.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://alesbeseda.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://alesmerta.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://alexova.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://andreanovotna1.blog.idnes.cz/redir.aspx?url=https%3A%2F%2Fspo337.com/
https://maps.google.com/url?sa=t&url=https%3A%2F%2Fspo337.com/
http://www.google.com/url?q=https%3A%2F%2Fspo337.com/
https://www.google.co.uk/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.fr/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://maps.google.es/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://maps.google.ca/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.nl/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.marcellusmatters.psu.edu/?URL=https%3A%2F%2Fspo337.com/
https://www.google.com.au/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://v1.addthis.com/live/redirect/?url=https%3A%2F%2Fspo337.com/
http://daidai.gamedb.info/wiki/?cmd=jumpto&r=https%3A%2F%2Fspo337.com/
https://www.hobowars.com/game/linker.php?url=https%3A%2F%2Fspo337.com/
http://www.clevelandbay.com/?URL=https%3A%2F%2Fspo337.com/
https://www.wheretoskiandsnowboard.com/?URL=https%3A%2F%2Fspo337.com/
http://archive.paulrucker.com/?URL=https%3A%2F%2Fspo337.com/
http://openroadbicycles.com/?URL=https%3A%2F%2Fspo337.com/
http://www.onesky.ca/?URL=https%3A%2F%2Fspo337.com/
http://jc-log.jmirus.de/?URL=https%3A%2F%2Fspo337.com/
http://www.shamelesstraveler.com/?URL=https%3A%2F%2Fspo337.com/
https://www.google.com.sv/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://maps.google.ci/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.jo/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://maps.google.com.bh/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.com.pa/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://images.google.co.bw/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://images.google.am/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://images.google.az/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://maps.google.ge/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.com.cu/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.com.kw/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://images.google.co.ma/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.com.gh/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.mk/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.co.ug/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://maps.google.as/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.ad/url?sa=t&url=https%3A%2F%2Fspo337.com/
http://www.astro.wisc.edu/?URL=https%3A%2F%2Fspo337.com/
http://www.astro.wisc.edu/?URL=/https%3A%2F%2Fspo337.com/
https://maps.google.cd/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.com.cy/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://images.google.bs/url?sa=t&url=https%3A%2F%2Fspo337.com/
http://www.google.com.mx/url?sa=t&url=https%3A%2F%2Fspo337.com/
http://images.google.hu/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.li/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://images.google.com.bz/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.com.af/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://images.google.com.ag/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://images.google.bi/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.mn/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://maps.google.tt/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://images.google.com.na/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://images.google.com.qa/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://images.google.sn/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://images.google.al/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://images.google.fm/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.iq/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://images.google.com.gi/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.je/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.mg/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.com.om/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.dj/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://images.google.com.ly/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.com.jm/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://images.google.com.et/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.md/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.sh/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.co.tz/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.me/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.kg/url?sa=t&url=https%3A%2F%2Fspo337.com/
http://www.google.mw/url?q=https%3A%2F%2Fspo337.com/
https://www.google.mw/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.ht/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.rw/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.ms/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.ps/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.com.bn/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://cse.google.pt/url?q=https%3A%2F%2Fspo337.com/
http://images.google.pt/url?sa=t&url=https%3A%2F%2Fspo337.com/
http://www.google.co.id/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://images.google.co.ls/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://images.google.bf/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.la/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://maps.google.com.fj/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.mv/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.gg/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.triathlon.org/?URL=/https%3A%2F%2Fspo337.com/
http://www.google.com.ar/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.co.mz/url?sa=t&url=https%3A%2F%2Fspo337.com/
http://www.google.co.th/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.gm/url?sa=t&url=https%3A%2F%2Fspo337.com/
http://www.google.no/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.gl/url?sa=t&url=https%3A%2F%2Fspo337.com/
http://maps.google.co.za/url?q=https%3A%2F%2Fspo337.com/
http://images.google.ro/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://cse.google.com.vn/url?q=https%3A%2F%2Fspo337.com/
http://cse.google.com.ph/url?q=https%3A%2F%2Fspo337.com/
http://images.google.com.pk/url?q=https%3A%2F%2Fspo337.com/
http://maps.google.gr/url?sa=t&url=https%3A%2F%2Fspo337.com/
https://www.google.gp/url?sa=t&url=https%3A%2F%2Fspo337.com/
http://www.google.ie/url?q=https%3A%2F%2Fspo337.com/
https://images.google.bg/url?q=https%3A%2F%2Fspo337.com/
https://images.google.com/url?q=https%3A%2F%2Fspo337.com/
http://www.cssdrive.com/?URL=/https%3A%2F%2Fspo337.com/
http://www.google.sk/url?q=https%3A%2F%2Fspo337.com/
http://www.google.co.il/url?q=https%3A%2F%2Fspo337.com/
http://cse.google.rs/url?q=https%3A%2F%2Fspo337.com/
http://www.earth-policy.org/?URL=https%3A%2F%2Fspo337.com/
http://maps.google.lt/url?q=https%3A%2F%2Fspo337.com/
http://maps.google.ae/url?q=https%3A%2F%2Fspo337.com/
http://www.google.com.co/url?q=https%3A%2F%2Fspo337.com/
http://maps.google.hr/url?q=https%3A%2F%2Fspo337.com/
https://community.cypress.com/external-link.jspa?url=http://https%3A%2F%2Fspo337.com/
https://community.rsa.com/external-link.jspa?url=https%3A%2F%2Fspo337.com/
http://www.pasco.k12.fl.us/?URL=https%3A%2F%2Fspo337.com/
https://cse.google.com/url?q=https%3A%2F%2Fspo337.com/
https://maps.google.ee/url?q=https%3A%2F%2Fspo337.com/
http://maps.google.lv/url?q=https%3A%2F%2Fspo337.com/
http://www.google.com.np/url?q=https%3A%2F%2Fspo337.com/
http://www.bookmerken.de/?url=https%3A%2F%2Fspo337.com/
http://maps.google.co.ve/url?q=https%3A%2F%2Fspo337.com/
http://maps.google.com.ec/url?q=https%3A%2F%2Fspo337.com/
http://cse.google.com.bd/url?q=https%3A%2F%2Fspo337.com/
http://maps.google.by/url?q=https%3A%2F%2Fspo337.com/
http://maps.google.lu/url?q=https%3A%2F%2Fspo337.com/
http://images.google.com.uy/url?q=https%3A%2F%2Fspo337.com/
http://www.google.co.cr/url?q=https%3A%2F%2Fspo337.com/
http://cse.google.tn/url?q=https%3A%2F%2Fspo337.com/
http://www.google.mu/url?q=https%3A%2F%2Fspo337.com/
https://www.fuzokubk.com/cgi-bin/LinkO.cgi?u=https%3A%2F%2Fspo337.com/
http://images.google.com.pr/url?q=https%3A%2F%2Fspo337.com/
https://legacy.aom.org/verifymember.asp?nextpage=http://https%3A%2F%2Fspo337.com/
http://www.novalogic.com/remote.asp?NLink=https%3A%2F%2Fspo337.com/
http://www.orthodoxytoday.org/?URL=https%3A%2F%2Fspo337.com/
https://bukkit.org/proxy.php?link=https%3A%2F%2Fspo337.com/
http://www.searchdaimon.com/?URL=https%3A%2F%2Fspo337.com/
http://icecap.us/?URL=https%3A%2F%2Fspo337.com/
https://www.adminer.org/redirect/?url=https%3A%2F%2Fspo337.com/
http://www.arakhne.org/redirect.php?url=https%3A%2F%2Fspo337.com/
https://www.raincoast.com/?URL=https%3A%2F%2Fspo337.com/
http://4vn.eu/forum/vcheckvirus.php?url=https%3A%2F%2Fspo337.com/
http://holidaykitchens.com/?URL=https%3A%2F%2Fspo337.com/
http://www.adhub.com/cgi-bin/webdata_pro.pl?cgifunction=clickthru&url=https%3A%2F%2Fspo337.com/
https://client.paltalk.com/client/webapp/client/External.wmt?url=https%3A%2F%2Fspo337.com/
http://www.virtual-egypt.com/framed/framed.cgi?url==https%3A%2F%2Fspo337.com/
http://www.webclap.com/php/jump.php?url=https%3A%2F%2Fspo337.com/
http://urlxray.com/display.php?url=https%3A%2F%2Fspo337.com/
http://ime.nu/sc.hkexnews.hk/TuniS/spo337.com/
http://www.allods.net/redirect/davidcouperconsulting.com/?URL=spo337.com
https://jump.5ch.net/?morrowind.ru/redirect/spo337.com
http://www.morrowind.ru/redirect/ucrca.org/?URL=spo337.com/
https://jump.5ch.net/?wagyu.org/?URL=spo337.com/
https://jump.5ch.net/?unbridledbooks.com/?URL=spo337.com/feft/ref/xiswi/
http://www.allods.net/redirect/jongeriuslab.com/?URL=spo337.com
https://smmry.com/woodforestcharitablefoundation.org/?URL=spo337.com/
https://cwcab.com/?URL=judiisrael.com/?URL=spo337.com
http://www.morrowind.ru/redirect/wdvstudios.be/?URL=spo337.com/feft/ref/xiswi/
https://jump.5ch.net/?frienddo.com/out.php?url=spo337.com
https://jump.5ch.net/?ctlimo.com/?URL=spo337.com/feft/ref/xiswi/
http://www.ut2.ru/redirect/bvilpcc.com/?URL=spo337.com
https://cwcab.com/?URL=reedring.com/?URL=spo337.com/
http://www.ut2.ru/redirect/lbaproperties.com/?URL=spo337.com/feft/ref/xiswi/
http://ime.nu/kingswelliesnursery.com/?URL=spo337.com/
https://smmry.com/miloc.hr/?URL=spo337.com/
https://smmry.com/nslgames.com/?URL=spo337.com/
http://bios.edu/?URL=thebigmo.nl/?URL=spo337.com/
http://www.nwnights.ru/redirect/steamcommunity.com/linkfilter/?url=spo337.com
http://2ch.io/barrypopik.com/index.php?URL=spo337.com/
https://cwcab.com/?URL=batterybusiness.com.au/?URL=spo337.com/
https://smmry.com/blingguard.com/?URL=spo337.com/feft/ref/xiswi/
https://cwcab.com/?URL=professor-murmann.info/?URL=spo337.com
http://www.allods.net/redirect/burkecounty-ga.gov/?URL=spo337.com/
http://www.morrowind.ru/redirect/grillages-wunschel.fr/?URL=spo337.com/feft/ref/xiswi/
https://smmry.com/xn-herzrhythmusstrungen-hbc.biz/goto.php?site=spo337.com/
http://www.ut2.ru/redirect/slrc.org/?URL=spo337.com/
https://jump.5ch.net/?blingguard.com/?URL=spo337.com
https://jump.5ch.net/?ponsonbyacupunctureclinic.co.nz/?URL=spo337.com
https://cwcab.com/?URL=eaglesgymnastics.com/?URL=spo337.com
http://www.ut2.ru/redirect/weburg.net/redirect?url=spo337.com/
http://2ch.io/assertivenorthwest.com/?URL=spo337.com/
http://www.morrowind.ru/redirect/slighdesign.com/?URL=spo337.com
http://ime.nu/gumexslovakia.sk/?URL=spo337.com/
http://www.allods.net/redirect/magenta-mm.com/?URL=spo337.com/
http://bios.edu/?URL=shavermfg.com/?URL=spo337.com
http://www.allods.net/redirect/healthyeatingatschool.ca/?URL=spo337.com/feft/ref/xiswi/
http://www.gta.ru/redirect/dcfossils.org/?URL=spo337.com
https://jump.5ch.net/?pro-net.se/?URL=spo337.com/
https://jump.5ch.net/?arbor-tech.be/?URL=spo337.com
http://www.allods.net/redirect/theaustonian.com/?URL=spo337.com/
http://ime.nu/basebusiness.com.au/?URL=spo337.com/
https://cwcab.com/?URL=hfw1970.de/redirect.php?url=spo337.com
http://www.ut2.ru/redirect/firma.hr/?URL=spo337.com/
http://www.nwnights.ru/redirect/giruna.hu/redirect.php?url=spo337.com
http://www.gta.ru/redirect/emotional.ro/?URL=spo337.com/
http://www.gta.ru/redirect/couchsrvnation.com/?URL=spo337.com/feft/ref/xiswi/
http://www.allods.net/redirect/turbo-x.hr/?URL=spo337.com/
http://www.gta.ru/redirect/albins.com.au/?URL=spo337.com/
http://www.ut2.ru/redirect/emophilips.com/?URL=spo337.com
http://2ch.io/md-technical.com/?URL=spo337.com/feft/ref/xiswi/
http://www.ut2.ru/redirect/stcroixblades.com/?URL=spo337.com/
https://jump.5ch.net/?sassyj.net/?URL=spo337.com/
http://www.nwnights.ru/redirect/applicationadvantage.com/?URL=spo337.com
http://www.ut2.ru/redirect/hotyoga.co.nz/?URL=spo337.com/
http://www.morrowind.ru/redirect/accord.ie/?URL=spo337.com/
http://ime.nu/s79457.gridserver.com/?URL=spo337.com/
http://2ch.io/morrowind.ru/redirect/spo337.com/feft/ref/xiswi/
https://smmry.com/centre.org.au/?URL=spo337.com/
http://ime.nu/pulaskiticketsandtours.com/?URL=spo337.com/
http://www.nwnights.ru/redirect/hs-events.nl/?URL=spo337.com
http://www.ut2.ru/redirect/chivemediagroup.com/?URL=spo337.com/feft/ref/xiswi/
http://www.nwnights.ru/redirect/horizon-environ.com/?URL=spo337.com/
http://www.morrowind.ru/redirect/yesfest.com/?URL=spo337.com/
http://www.morrowind.ru/redirect/t.me/iv?url=spo337.com
http://www.allods.net/redirect/boosterblog.com/vote-815901-624021.html?adresse=spo337.com/
http://www.ut2.ru/redirect/client.paltalk.com/client/webapp/client/External.wmt?url=spo337.com
http://www.morrowind.ru/redirect/minecraft-galaxy.ru/redirect/?url=spo337.com
http://www.nwnights.ru/redirect/rescuetheanimals.org/?URL=spo337.com/
http://ime.nu/foosball.com/?URL=spo337.com
http://www.ut2.ru/redirect/supertramp.com/?URL=spo337.com
http://www.allods.net/redirect/gmmdl.com/?URL=spo337.com/feft/ref/xiswi/
http://2ch.io/icecap.us/?URL=spo337.com/feft/ref/xiswi/
http://www.nwnights.ru/redirect/labassets.com/?URL=spo337.com/
https://cwcab.com/?URL=acceleweb.com/register?aw_site_id=spo337.com/
http://www.allods.net/redirect/cim.bg/?URL=spo337.com/
http://ime.nu/rawseafoods.com/?URL=spo337.com
http://2ch.io/2ch.io/spo337.com
http://www.nwnights.ru/redirect/mikropul.com/?URL=spo337.com/
https://cwcab.com/?URL=bytecheck.com/results?resource=spo337.com
http://www.nwnights.ru/redirect/youtube.com/redirect?q=spo337.com
http://www.nwnights.ru/redirect/salonfranchise.com.au/?URL=spo337.com
https://cwcab.com/?URL=couchsrvnation.com/?URL=spo337.com/
http://ime.nu/usich.gov/?URL=spo337.com
http://www.nwnights.ru/redirect/sostrategic.com.au/?URL=spo337.com/
http://www.allods.net/redirect/boosterblog.net/vote-146-144.html?adresse=spo337.com/
http://www.allods.net/redirect/wilsonlearning.com/?URL=spo337.com/
http://ime.nu/nerida-oasis.com/?URL=spo337.com/feft/ref/xiswi/
http://2ch.io/aldonauto.com/?URL=spo337.com/
http://www.allods.net/redirect/spot-car.com/?URL=spo337.com
http://ime.nu/hornbeckoffshore.com/?URL=spo337.com/
https://cwcab.com/?URL=dentalcommunity.com.au/?URL=spo337.com/
http://www.morrowind.ru/redirect/peter.murmann.name/?URL=spo337.com
http://www.morrowind.ru/redirect/firma.hr/?URL=spo337.com
https://jump.5ch.net/?labassets.com/?URL=spo337.com/feft/ref/xiswi/
http://ime.nu/progressprinciple.com/?URL=spo337.com
http://www.nwnights.ru/redirect/mbcarolinas.org/?URL=spo337.com/
https://jump.5ch.net/?fotka.com/link.php?u=spo337.com
http://bios.edu/?URL=restaurant-zahnacker.fr/?URL=spo337.com
https://cwcab.com/?URL=accord.ie/?URL=spo337.com
http://www.nwnights.ru/redirect/morrisparks.net/?URL=spo337.com
https://cwcab.com/?URL=fishidy.com/go?url=spo337.com
http://www.nwnights.ru/redirect/blingguard.com/?URL=spo337.com/feft/ref/xiswi/
http://www.ut2.ru/redirect/ria-mar.com/?URL=spo337.com/
https://jump.5ch.net/?batterybusiness.com.au/?URL=spo337.com/
http://www.gta.ru/redirect/healthyeatingatschool.ca/?URL=spo337.com/feft/ref/xiswi/
https://cwcab.com/?URL=ntltyres.com.au/?URL=spo337.com/
https://jump.5ch.net/?romanodonatosrl.com/?URL=spo337.com
http://www.morrowind.ru/redirect/pcrnv.com.au/?URL=spo337.com/feft/ref/xiswi/
https://jump.5ch.net/?bluewatergrillri.com/?URL=spo337.com
http://2ch.io/washburnvalley.org/?URL=spo337.com/
https://smmry.com/crspublicity.com.au/?URL=spo337.com/
http://www.allods.net/redirect/vectechnologies.com/?URL=spo337.com/
https://smmry.com/troxellwebdesign.com/?URL=spo337.com/
https://jump.5ch.net/?jacobberger.com/?URL=spo337.com/feft/ref/xiswi/
http://www.ut2.ru/redirect/oncreativity.tv/?URL=spo337.com
http://www.allods.net/redirect/ww2.torahlab.org/?URL=spo337.com/
http://2ch.io/essencemusicagency.com/?URL=spo337.com
http://www.ut2.ru/redirect/livingtrustplus.com/?URL=spo337.com/
https://cwcab.com/?URL=giruna.hu/redirect.php?url=spo337.com/
http://www.nwnights.ru/redirect/cssdrive.com/?URL=spo337.com/feft/ref/xiswi/
http://bios.edu/?URL=basebusiness.com.au/?URL=spo337.com
http://www.allods.net/redirect/2ch.io/spo337.com/ http://2ch.io/slrc.org/?URL=spo337.com
http://www.nwnights.ru/redirect/life-church.com.au/?URL=spo337.com/
http://bios.edu/?URL=precisioncomponents.com.au/?URL=spo337.com/
http://www.nwnights.ru/redirect/okellymoylan.ie/?URL=spo337.com/
pnevmopodveska-club.ru/index.php?app=core&module=system&controller=redirect&do=redirect&url=https%3A%2F%2Fspo337.com
www.darussalamciamis.or.id/redirect/?alamat=http%3A%2F%2Fspo337.com
agama.su/go.php?url=https%3A%2F%2Fspo337.com
bas-ip.ru/bitrix/rk.php?goto=https%3A%2F%2Fspo337.com
college.captainu.com/college_teams/1851/campaigns/51473/tracking/click?contact_id=1154110&email_id=1215036&url=https%3A%2F%2Fspo337.com/
www.oldfold.com/g?u=https%3A%2F%2Fspo337.com
www.anibox.org/go?https%3A%2F%2Fspo337.com
https://acejobs.net/jobclick/?RedirectURL=http%3A%2F%2Fspo337.com&Domain=acejobs.net
www.m.mobilegempak.com/wap_api/get_msisdn.php?URL=https%3A%2F%2Fspo337.com/
sitesdeapostas.co.mz/track/odd?url-id=11&game-id=1334172&odd-type=draw&redirect=https%3A%2F%2Fspo337.com
www.autaabouracky.cz/plugins/guestbook/go.php?url=https%3A%2F%2Fspo337.com/
www.escapers-zone.net/ucp.php?mode=logout&redirect=http%3A%2F%2Fspo337.com
members.practicegreenhealth.org/eweb/Logout.aspx?RedirectURL=https%3A%2F%2Fspo337.com/
http://fiinpro.com/Home/ChangeLanguage?lang=vi-VN&returnUrl=https%3A%2F%2Fspo337.com/
https://visit-thassos.com/index.php/language/en?redirect=https%3A%2F%2Fspo337.com
adserverv6.oberberg.net/adserver/www/delivery/ck.php?ct=1&oaparams=2bannerid=2zoneid=35cb=88915619faoadest=https%3A%2F%2Fspo337.com
www.avilas-style.com/shop/affiche.php?ad_id=132&from=&uri=spo337.com
www.hentaicrack.com/cgi-bin/atx/out.cgi?s=95&u=https%3A%2F%2Fspo337.com/
www.voxlocalis.net/enlazar/?url=https%3A%2F%2Fspo337.com/
www.ferrosystems.com/setLocale.jsp?language=en&url=https%3A%2F%2Fspo337.com
test.healinghealth.com/?wptouch_switch=desktop&redirect=https%3A%2F%2Fspo337.com/
www.lastdates.com/l/?spo337.com
kaimono-navi.jp/rd?u=http%3A%2F%2Fspo337.com
www.shop-bell.com/out.php?id=kibocase&category=ladies&url=https%3A%2F%2Fspo337.com/
https://richmonkey.biz/go/?https%3A%2F%2Fspo337.com
https://online-knigi.com/site/getlitresurl?url=http%3A%2F%2Fspo337.com
akvaforum.no/go.cfml?id=1040&uri=https%3A%2F%2Fspo337.com
www.kamphuisgroep.nl/r.php?cid=2314&site=https%3A%2F%2Fspo337.com
sns.51.ca/link.php?url=https%3A%2F%2Fspo337.com
http://newsrankey.com/view.html?url=https%3A%2F%2Fspo337.com/
www.minibuggy.net/forum/redirect-to/?redirect=https%3A%2F%2Fspo337.com
www.wagersmart.com/top/out.cgi?id=bet2gold&url=https%3A%2F%2Fspo337.com/
www.global-autonews.com/shop/bannerhit.php?bn_id=307&url=https%3A%2F%2Fspo337.com/
www.homuta.co.jp/link/?link=http%3A%2F%2Fspo337.com
klvr.link/redirect/venividivici/spotify?linkUrl=http%3A%2F%2Fspo337.com
www.pcstore.com.tw/adm/act.htm?src=vipad_click&store_type=SUP_TOP&big_exh=STOREAD-%A7%E950&reurl=http%3A%2F%2Fspo337.com
http://baantawanchandao.com/change_language.asp?language_id=th&MemberSite_session=site_47694_&link=https%3A%2F%2Fspo337.com/
grannyfuck.in/cgi-bin/atc/out.cgi?id=139&u=https%3A%2F%2Fspo337.com
www.joserodriguez.info/?wptouch_switch=desktop&redirect=https%3A%2F%2Fspo337.com
https://navigraph.com/redirect.ashx?url=https%3A%2F%2Fspo337.com
edu54.ru/bitrix/redirect.php?goto=https%3A%2F%2Fspo337.com
https://honolulufestival.com/ja/?wptouch_switch=desktop&redirect=http%3A%2F%2Fspo337.com
www.perinosboilingpot.com/site.php?pageID=1&bannerID=19&vmoment=1430132758&url=http%3A%2F%2Fspo337.com
www.hardcoreoffice.com/tp/out.php?link=txt&url=https%3A%2F%2Fspo337.com/
best.amateursecrets.net/cgi-bin/out.cgi?ses=onmfsqgs6c&id=318&url=https%3A%2F%2Fspo337.com/
elitesm.ru/bitrix/rk.php?id=102&site_id=ru&event1=banner&event2=click&event3=1+%2F+%5B102%5D+%5Bright_group_bot%5D+%DD%CA%CE+3%C4&goto=https%3A%2F%2Fspo337.com/
www.iasb.com/sso/login/?userToken=Token&returnURL=https%3A%2F%2Fspo337.com
prominentjobs.co.uk/jobclick/?RedirectURL=http%3A%2F%2Fspo337.com
http://workshopweekend.net/er?url=https%3A%2F%2Fspo337.com/
go.pnuna.com/go.php?url=https%3A%2F%2Fspo337.com
https://przyjazniseniorom.com/language/en/?returnUrl=http%3A%2F%2Fspo337.com
motorrad-stecki.de/trigger.php?r_link=http%3A%2F%2Fspo337.com
news.animravel.fr/retrolien.aspx?id_dest=1035193&id_envoi=463&url=spo337.com/
http://aldenfamilyonline.com/KathySite/MomsSite/MOM_SHARE_MEMORIES/msg_system/go.php?url=https%3A%2F%2Fspo337.com
www.sports-central.org/cgi-bin/axs/ax.pl?https%3A%2F%2Fspo337.com/
shop.mediaport.cz/redirect.php?action=url&goto=spo337.com
adlogic.ru/?goto=jump&url=https%3A%2F%2Fspo337.com/
www.waters.com/waters/downloadFile.htm?lid=134799103&id=134799102&fileName=Download&fileUrl=http%3A%2F%2Fspo337.com
www.bari91.com/tz.php?zone=Pacific/Niue&r=http%3A%2F%2Fspo337.com
www.xfdq123.com/url.aspx?url=https%3A%2F%2Fspo337.com
www.mirogled.com/banner-clicks/10?url=https%3A%2F%2Fspo337.com
youngpussy.ws/out.php?https%3A%2F%2Fspo337.com
www.hartje.name/go?r=1193&jumpto=https%3A%2F%2Fspo337.com
www.yplf.com/cgi-bin/a2/out.cgi?id=141&l=toplist&u=https%3A%2F%2Fspo337.com
www.joblinkapply.com/Joblink/5972/Account/ChangeLanguage?lang=es-MX&returnUrl=https%3A%2F%2Fspo337.com
smedia.ru/bitrix/rk.php?goto=https%3A%2F%2Fspo337.com
navitrinu.ru/redirect/?go=http%3A%2F%2Fspo337.com
https://hatboroalive.com/abnrs/countguideclicks.cfm?targeturl=http%3A%2F%2Fspo337.com&businessid=29277
www.irrigationnz.co.nz/ClickThru?mk=5120.0&Redir=https%3A%2F%2Fspo337.com
buecher-teneues.de/mlm/lm/lm.php?tk=CQkJRkRhdW1AdGVuZXVlcy5jb20JU3BlY2lhbCBPZmZlcnMgYmVpIHRlTmV1ZXMgCTM3CQkzNzQ1CWNsaWNrCXllcwlubw==&url=https%3A%2F%2Fspo337.com
karir.imslogistics.com/language/en?return=https%3A%2F%2Fspo337.com/
yun.smartlib.cn/widgets/fulltext/?url=https%3A%2F%2Fspo337.com
www.okhba.org/clicks.php?bannerid=51&url=http%3A%2F%2Fspo337.com
www.all1.co.il/goto.php?url=https%3A%2F%2Fspo337.com/
https://jipijapa.net/jobclick/?RedirectURL=http%3A%2F%2Fspo337.com&Domain=jipijapa.net&rgp_m=co3&et=4495
chaku.tv/i/rank/out.cgi?url=https%3A%2F%2Fspo337.com
www.mendocino.com/?id=4884&url=spo337.com
doc.enervent.com/op/op.SetLanguage.php?lang=de_DE&referer=http%3A%2F%2Fspo337.com
www.feg-jena.de/link/?link=https%3A%2F%2Fspo337.com
http://dstats.net/redir.php?url=https%3A%2F%2Fspo337.com/
crescent.netcetra.com/inventory/military/dfars/?saveme=MS51957-42*&redirect=https%3A%2F%2Fspo337.com/
r5.dir.bg/rem.php?word_id=0&place_id=9&ctype=mp&fromemail=&iid=3770&aid=4&cid=0&url=https%3A%2F%2Fspo337.com/
www.store-datacomp.eu/Home/ChangeLanguage?lang=en&returnUrl=http%3A%2F%2Fspo337.com
http://hotgrannyworld.com/cgi-bin/crtr/out.cgi?id=41&l=toplist&u=https%3A%2F%2Fspo337.com
t.wxb.com/order/sourceUrl/1894895?url=spo337.com/
shop.merchtable.com/users/authorize?return_url=https%3A%2F%2Fspo337.com/
elinks.qp.land.to/link.php?url=https%3A%2F%2Fspo337.com
www.powerflexweb.com/centers_redirect_log.php?idDivision=25&nameDivision=Homepage&idModule=m551&nameModule=myStrength&idElement=298&nameElement=Provider%20Search&url=https%3A%2F%2Fspo337.com/
rbs-crm.ru/?redirect_url=http%3A%2F%2Fspo337.com
www.theukhighstreet.com/perl/jump.cgi?ID=12&URL=https%3A%2F%2Fspo337.com
ekonomka.dn.ua/out.php?link=http://spo337.com/
digital.fijitimes.com/api/gateway.aspx?f=https%3A%2F%2Fspo337.com/
https://amateurdorado.com/wp-content/plugins/AND-AntiBounce/redirector.php?url=https%3A%2F%2Fspo337.com
www.medicumlaude.de/index.php/links/index.php?url=https%3A%2F%2Fspo337.com
www.figurama.eu/cz/redirect.php?path=https%3A%2F%2Fspo337.com
nagranitse.ru/url.php?q=https%3A%2F%2Fspo337.com/
www.wien-girls.at/out-link?url=https%3A%2F%2Fspo337.com/
www.blackpictures.net/jcet/tiov.cgi?cvns=1&s=65&u=https%3A%2F%2Fspo337.com
https://athleticforum.biz/redirect/?to=http%3A%2F%2Fspo337.com
www.telehaber.com/redir.asp?url=https%3A%2F%2Fspo337.com
www.elmore.ru/go.php?to=https%3A%2F%2Fspo337.com
www.tido.al/vazhdo.php?url=https%3A%2F%2Fspo337.com/
os-company.ru/bitrix/redirect.php?goto=https%3A%2F%2Fspo337.com
duma-slog.ru/bitrix/redirect.php?event1=file&event2=spo337.com/&event3=29.01.2015_312_rd.doc&goto=https%3A%2F%2Fspo337.com/
www.interempresas.net/estadisticas/r.asp?idsector=129&e=221083&c=195&d=https%3A%2F%2Fspo337.com
pravoslavieru.trckmg.com/app/click/30289/561552041/?goto_url=spo337.com
jump.fan-site.biz/rank.cgi?mode=link&id=342&url=https%3A%2F%2Fspo337.com/
http://anti-kapitalismus.org/sites/all/modules/pubdlcnt/pubdlcnt.php?file=https%3A%2F%2Fspo337.com&nid=435
rubyconnection.com.au/umbraco/newsletterstudio/tracking/trackclick.aspx?nid=207065033113056034011005043041220243180024215107&e=011204127253056232044128247253046214192002250116195220062107112232157159227010159247231011081075001197133136091194134170178051032155159001112047&url=https%3A%2F%2Fspo337.com/
video.childsheroes.com/Videos/SetCulture?culture=en-US&returnURL=http%3A%2F%2Fspo337.com
winehall.ru/bitrix/rk.php?goto=https%3A%2F%2Fspo337.com
efir-kazan.ru/bitrix/rk.php?id=367&site_id=s1&event1=banner&event2=click&event3=47+/367[Main_middle_1]%D0%90%D0%BA%D0%91%D0%B0%D1%80%D1%81+%D0%A1%D0%BF%D0%BE%D1%80%D1%82&goto=https%3A%2F%2Fspo337.com/
yiwu.0579.com/jump.asp?url=https%3A%2F%2Fspo337.com
t.goadservices.com/optout?url=https%3A%2F%2Fspo337.com
auth.philpapers.org/login?service=https%3A%2F%2Fspo337.com
forsto.ru/bitrix/redirect.php?goto=https%3A%2F%2Fspo337.com
www.slavenibas.lv/bancp/www/delivery/ck.php?ct=1&oaparams=2bannerid=82zoneid=2cb=008ea50396oadest=http%3A%2F%2Fspo337.com
neso.r.niwepa.com/ts/i5536875/tsc?tst=!&amc=con.blbn.490450.485278.164924&pid=6508&rmd=3&trg=https%3A%2F%2Fspo337.com/
italiantrip.it/information_about_cookie_read.php?url=https%3A%2F%2Fspo337.com
cgi1.bellacoola.com/adios.cgi/630?https%3A%2F%2Fspo337.com/
adengine.old.rt.ru/go.jsp?to=https%3A%2F%2Fspo337.com
www.simpleet.lu/Home/ChangeCulture?lang=de-DE&returnUrl=http%3A%2F%2Fspo337.com
enseignants.flammarion.com/Banners_Click.cfm?ID=86&URL=spo337.com/
content.sixflags.com/news/director.aspx?gid=0&iid=72&cid=3714&link=https%3A%2F%2Fspo337.com/
www.offendorf.fr/spip_cookie.php?url=https%3A%2F%2Fspo337.com/
suche6.ch/count.php?url=https%3A%2F%2Fspo337.com/
shiftlink.ca/AbpLocalization/ChangeCulture?cultureName=de&returnUrl=http%3A%2F%2Fspo337.com
www.canakkaleaynalipazar.com/advertising.php?r=3&l=https%3A%2F%2Fspo337.com
www.tascher-de-la-pagerie.org/fr/liens.php?l=https%3A%2F%2Fspo337.com/
i.s0580.cn/module/adsview/content/?action=click&bid=5&aid=163&url=http%3A%2F%2Fspo337.com&variable=&source=https%3A%2F%2Fcutepix.info%2Fsex%2Friley-reyes.php
jsv3.recruitics.com/redirect?rx_cid=506&rx_jobId=39569207&rx_url=https%3A%2F%2Fspo337.com/
https://jobinplanet.com/away?link=spo337.com
www.supermoto8.com/sidebanner/62?href=https%3A%2F%2Fspo337.com
presse.toyota.dk/login.aspx?returnurl=https%3A%2F%2Fspo337.com/
www.uktrademarkregistration.co.uk/JumpTo.aspx?url=https%3A%2F%2Fspo337.com/
www.mir-stalkera.ru/go?https%3A%2F%2Fspo337.com
duhocphap.edu.vn/?wptouch_switch=desktop&redirect=http%3A%2F%2Fspo337.com
www.millerovo161.ru/go?https%3A%2F%2Fspo337.com/
www.naturaltranssexuals.com/cgi-bin/a2/out.cgi?id=97&l=toplist&u=https%3A%2F%2Fspo337.com
https://amanaimages.com/lsgate/?lstid=pM6b0jdQgVM-Y9ibFgTe6Zv1N0oD2nYuMA&lsurl=https%3A%2F%2Fspo337.com
planszowkiap.pl/trigger.php?r_link=http%3A%2F%2Fspo337.com
http://covenantpeoplesministry.org/cpm/wp/sermons/?show&url=https%3A%2F%2Fspo337.com/
www.sporteasy.net/redirect/?url=https%3A%2F%2Fspo337.com
getwimax.jp/st-manager/click/track?id=3894&type=raw&url=https%3A%2F%2Fspo337.com/&source_url=https://getwimax.jp/&source_title=GetWiMAX.jp%EF%BD%9CWiMAX%EF%BC%88%E3%83%AF%E3%82%A4%E3%83%9E%E3%83%83%E3%82%AF%E3%82%B9%EF%BC%89%E6%AF%94%E8%BC%83%EF%BC%81%E3%81%8A%E3%81%99%E3%81%99%E3%82%81%E3%83%97%E3%83%AD%E3%83%90%E3%82%A4%E3%83%803%E9%81%B8
mobo.osport.ee/Home/SetLang?lang=cs&returnUrl=https%3A%2F%2Fspo337.com
materinstvo.ru/forward?link=http%3A%2F%2Fspo337.com
www.veloxbox.us/link/?h=http%3A%2F%2Fspo337.com
www.adult-plus.com/ys/rank.php?mode=link&id=592&url=https%3A%2F%2Fspo337.com/
mobilize.org.br/handlers/anuncioshandler.aspx?anuncio=55&canal=2&redirect=https%3A%2F%2Fspo337.com/
sparktime.justclick.ru/lms/api-login/?hash=MO18szcRUQdzpT%2FrstSCW5K8Gz6ts1NvTJLVa34vf1A%3D&authBhvr=1&email=videotrend24%40mail.ru&expire=1585462818&lms%5BrememberMe%5D=1&targetPath=http%3A%2F%2Fspo337.com
redirect.icurerrors.com/http/spo337.com
http://vcteens.com/cgi-bin/at3/out.cgi?trade=https%3A%2F%2Fspo337.com
www.bquest.org/Links/Redirect.aspx?ID=164&url=https%3A%2F%2Fspo337.com/
www.stipendije.info/phpAdsNew/adclick.php?bannerid=129&zoneid=1&source=&dest=https%3A%2F%2Fspo337.com/
today.od.ua/redirect.php?url=https%3A%2F%2Fspo337.com/
laskma.megastart-slot.ru/redirect/?g=https%3A%2F%2Fspo337.com
mightypeople.asia/link.php?id=M0ZGNHFISkd2bFh0RmlwSFU4bDN4QT09&destination=https%3A%2F%2Fspo337.com
www.chinaleatheroid.com/redirect.php?url=https%3A%2F%2Fspo337.com
http://computer-chess.org/lib/exe/fetch.php?media=https%3A%2F%2Fspo337.com
https://enchantedcottageshop.com/shop/trigger.php?r_link=http%3A%2F%2Fspo337.com
https://buist-keatch.org/sphider/include/click_counter.php?url=http%3A%2F%2Fspo337.com
www.mistress-and-slave.com/cgi-bin/out.cgi?id=123crush&url=https%3A%2F%2Fspo337.com/
rel.chubu-gu.ac.jp/soumokuji/cgi-bin/go.cgi?https%3A%2F%2Fspo337.com/
fallout3.ru/utils/ref.php?url=https%3A%2F%2Fspo337.com/
https://www.youtube.com/redirect?q=https%3A%2F%2Fspo337.com%2F
https://www.youtube.com/redirect?q=https%3A%2F%2Fspo337.com%2F&gl=DE
https://www.youtube.com/redirect?q=https%3A%2F%2Fspo337.com%2F&gl=IT
https://www.youtube.com/redirect?q=https%3A%2F%2Fspo337.com&gl=AR
https://www.youtube.at/redirect?q=https%3A%2F%2Fspo337.com%2F
https://www.youtube.ch/redirect?q=https%3A%2F%2Fspo337.com%2F
https://www.youtube.fr/redirect?q=https%3A%2F%2Fspo337.com%2F
https://www.youtube.es/redirect?q=https%3A%2F%2Fspo337.com%2F
https://www.youtube.jp/redirect?q=https%3A%2F%2Fspo337.com%2F
https://www.youtube.co.uk/redirect?q=https%3A%2F%2Fspo337.com%2F
https://www.youtube.ru/redirect?q=https%3A%2F%2Fspo337.com%2F
https://www.youtube.pl/redirect?q=https%3A%2F%2Fspo337.com%2F
https://www.youtube.gr/redirect?q=https%3A%2F%2Fspo337.com%2F
https://www.youtube.nl/redirect?q=https%3A%2F%2Fspo337.com%2F
https://www.youtube.ca/redirect?q=https%3A%2F%2Fspo337.com%2F
https://www.youtube.cz/redirect?q=https%3A%2F%2Fspo337.com%2F
https://www.youtube.com/redirect?q=https%3A%2F%2Fspo337.com%2F&gl=AU
https://www.youtube.com.tw/redirect?q=https%3A%2F%2Fspo337.com%2F
http://www.youtube.de/redirect?event=channel_description&q=https%3A%2F%2Fspo337.com%2F
http://www.youtube.com/redirect?event=channel_description&q=https%3A%2F%2Fspo337.com%2F
http://www.youtube.co/redirect?event=channel_description&q=https%3A%2F%2Fspo337.com%2F
http://www.youtube.es/redirect?event=channel_description&q=https%3A%2F%2Fspo337.com%2F
http://www.youtube.ca/redirect?event=channel_description&q=https%3A%2F%2Fspo337.com%2F
http://www.youtube.nl/redirect?event=channel_description&q=https%3A%2F%2Fspo337.com%2F
http://www.youtube.pl/redirect?event=channel_description&q=https%3A%2F%2Fspo337.com%2F
http://www.youtube.ch/redirect?event=channel_description&q=https%3A%2F%2Fspo337.com%2F
http://www.youtube.be/redirect?event=channel_description&q=https%3A%2F%2Fspo337.com%2F
http://www.youtube.se/redirect?event=channel_description&q=https%3A%2F%2Fspo337.com%2F
http://www.youtube.dk/redirect?event=channel_description&q=https%3A%2F%2Fspo337.com%2F
http://www.youtube.pt/redirect?event=channel_description&q=https%3A%2F%2Fspo337.com%2F
http://www.youtube.no/redirect?event=channel_description&q=https%3A%2F%2Fspo337.com%2F
http://www.youtube.gr/redirect?event=channel_description&q=https%3A%2F%2Fspo337.com%2F
http://www.youtube.cl/redirect?event=channel_description&q=https%3A%2F%2Fspo337.com%2F
http://www.youtube.at/redirect?event=channel_description&q=https%3A%2F%2Fspo337.com%2F
http://www.youtube.bg/redirect?event=channel_description&q=https%3A%2F%2Fspo337.com%2F
http://www.youtube.sk/redirect?event=channel_description&q=https%3A%2F%2Fspo337.com%2F
http://www.youtube.rs/redirect?event=channel_description&q=https%3A%2F%2Fspo337.com%2F
http://www.youtube.lt/redirect?event=channel_description&q=https%3A%2F%2Fspo337.com%2F
http://www.youtube.si/redirect?event=channel_description&q=https%3A%2F%2Fspo337.com%2F
http://www.youtube.hr/redirect?event=channel_description&q=https%3A%2F%2Fspo337.com%2F
http://www.youtube.ee/redirect?event=channel_description&q=https%3A%2F%2Fspo337.com%2F
http://www.youtube.lu/redirect?event=channel_description&q=https%3A%2F%2Fspo337.com%2F
http://www.youtube.tn/redirect?event=channel_description&q=https%3A%2F%2Fspo337.com%2F
http://www.youtube.co.ke/redirect?event=channel_description&q=https%3A%2F%2Fspo337.com%2F
http://www.youtube.co.cr/redirect?event=channel_description&q=https%3A%2F%2Fspo337.com%2F
http://www.youtube.kz/redirect?event=channel_description&q=https%3A%2F%2Fspo337.com%2F
http://www.youtube.cat/redirect?event=channel_description&q=https%3A%2F%2Fspo337.com%2F
http://www.youtube.ge/redirect?event=channel_description&q=https%3A%2F%2Fspo337.com%2F
https://www.youtube.com/redirect?event=channel_description&q=https%3A%2F%2Fspo337.com%2F&gl=ml
www.mytokachi.jp/index.php?type=click&mode=sbm&code=2981&url=https%3A%2F%2Fspo337.com/
apiv2.nextgenshopping.com/api/click?domainName=spo337.com&partnerTag4=portal&partnerTag2=coupon&clickId=A499UMEpK&partnerWebsiteKey=QatCO22&syncBack=true
www.wqketang.com/logout?goto=https%3A%2F%2Fspo337.com
access.bridges.com/externalRedirector.do?url=spo337.com/
www.cossa.ru/bitrix/redirect.php?event1=click&event2=&event3=&goto=https%3A%2F%2Fspo337.com
bot.buymeapie.com/recipe?url=https%3A%2F%2Fspo337.com/
www.frodida.org/BannerClick.php?BannerID=29&LocationURL=https%3A%2F%2Fspo337.com
extremaduraempresarial.juntaex.es/cs/c/document_library/find_file_entry?p_l_id=47702&noSuchEntryRedirect=http%3A%2F%2Fspo337.com
rtb-asiamax.tenmax.io/bid/click/1462922913409/e95f2c30-1706-11e6-a9b4-a9f6fe33c6df/3456/5332/?rUrl=https%3A%2F%2Fspo337.com
www.joeshouse.org/booking?link=http%3A%2F%2Fspo337.com&ID=1112
hanhphucgiadinh.vn/ext-click.php?url=https%3A%2F%2Fspo337.com/
www.nymfer.dk/atc/out.cgi?s=60&l=topgallery&c=1&u=https%3A%2F%2Fspo337.com/
swra.backagent.net/ext/rdr/?http%3A%2F%2Fspo337.com
www.hschina.net/ADClick.aspx?SiteID=206&ADID=1&URL=https%3A%2F%2Fspo337.com
cipresso.ru/bitrix/redirect.php?goto=https%3A%2F%2Fspo337.com
www.vacacionartravel.com/DTCSpot/public/banner_redirect.aspx?idca=286&ids=75665176&cp=167&idcli=0&ci=2&p=https%3A%2F%2Fspo337.com
www.mydosti.com/Advertisement/updateadvhits.aspx?adid=48&gourl=https%3A%2F%2Fspo337.com
www.malles-bertault.com/?change_langue=en&url=http%253a%252f%252fspo337.com
www.accesslocksmithatlantaga.com/?wptouch_switch=mobile&redirect=http%3A%2F%2Fspo337.com
www.beeicons.com/redirect.php?site=https%3A%2F%2Fspo337.com/
hirlevel.pte.hu/site/redirect?newsletter_id=UFV1UG5yZ3hOaWFyQVhvSUFoRmRQUT09&recipient=Y25zcm1ZaGxvR0xJMFNtNmhwdmpPNFlVSzlpS2c4ZnA1NzRPWjJKY3QrND0=&address=spo337.com
www.cccowe.org/lang.php?lang=en&url=https%3A%2F%2Fspo337.com
namiotle.pl/?wptouch_switch=mobile&redirect=https%3A%2F%2Fspo337.com/
gfb.gameflier.com/func/actionRewriter.aspx?pro=http&url=spo337.com
golfpark.jp/banner/counter.aspx?url=https%3A%2F%2Fspo337.com
www.mexicolore.co.uk/click.php?url=https%3A%2F%2Fspo337.com
www.tiersertal.com/clicks/uk_banner_click.php?url=https%3A%2F%2Fspo337.com/
www.invisalign-doctor.in/api/redirect?url=https%3A%2F%2Fspo337.com
www.isadatalab.com/redirect?clientId=ee5a64e1-3743-9b4c-d923-6e6d092ae409&appId=69&value=[EMV%20FIELD]EMAIL[EMV%20/FIELD]&cat=Techniques+culturales&url=https%3A%2F%2Fspo337.com/
www.immunallergo.ru/bitrix/redirect.php?goto=https%3A%2F%2Fspo337.com/
dort.brontosaurus.cz/forum/go.php?url=https%3A%2F%2Fspo337.com/
eatart.dk/Home/ChangeCulture?lang=da&returnUrl=http%3A%2F%2Fspo337.com
trackingapp4.embluejet.com/Mod_Campaigns/tracking.asp?idem=31069343&em=larauz@untref.edu.ar&ca=73143&ci=0&me=72706&of=581028&adirecta=0&url=https%3A%2F%2Fspo337.com/
smils.ru/bitrix/redirect.php?goto=https%3A%2F%2Fspo337.com
spb-medcom.ru/redirect.php?https%3A%2F%2Fspo337.com
forest.ru/links.php?go=http%3A%2F%2Fspo337.com
reefcentral.ru/bitrix/rk.php?goto=https%3A%2F%2Fspo337.com
bsau.ru/bitrix/redirect.php?event1=news_out&event2=2Fiblock9CB0%D1D0D0D0%B0BB87B0%D1D1D0D1%82B5%D1D0%B8B0%D0D1D0D1%81828C+2.pdf&goto=https%3A%2F%2Fspo337.com
https://trackdaytoday.com/redirect-out?url=http%3A%2F%2Fspo337.com
空の最安値.travel.jp/smart/pc.asp?url=https%3A%2F%2Fspo337.com/
https://bigjobslittlejobs.com/jobclick/?RedirectURL=http%3A%2F%2Fspo337.com&Domain=bigjobslittlejobs.com&rgpm=title23&et=4495
ekovjesnik.hr/ads/www/delivery/ck.php?ct=1&oaparams=2bannerid=4zoneid=4cb=68dbdae1d1oadest=http%3A%2F%2Fspo337.com
www.obertaeva.com/include/get.php?go=https%3A%2F%2Fspo337.com
https://studiohire.com/admin-web-tel-process.php?memberid=4638&indentifier=weburl&websitelinkhitnumber=7&telnumberhitnumber=0&websiteurl=https%3A%2F%2Fspo337.com
www.quanmama.com/t/goto.aspx?url=https%3A%2F%2Fspo337.com
quartiernetz-friesenberg.ch/links-go.php?to=https%3A%2F%2Fspo337.com
www.maultalk.com/url.php?to=https%3A%2F%2Fspo337.com
www.infohakodate.com/ps/ps_search.cgi?act=jump&url=https%3A%2F%2Fspo337.com
www.e-expo.net/category/click_url.html?url=https%3A%2F%2Fspo337.com
www.chitaitext.ru/bitrix/redirect.php?event1=utw&event2=utw1&event3=&goto=https%3A%2F%2Fspo337.com
www.realsubliminal.com/newsletter/t/c/11098198/c?dest=http%3A%2F%2Fspo337.com
http://getdatasheet.com/url.php?url=https%3A%2F%2Fspo337.com
https://kirei-style.info/st-manager/click/track?id=7643&type=raw&url=http%3A%2F%2Fspo337.com
www.aldolarcher.com/tools/esstat/esdown.asp?File=http%3A%2F%2Fspo337.com
www.wave24.net/cgi-bin/linkrank/out.cgi?id=106248&cg=1&url=spo337.com/
www.tssweb.co.jp/?wptouch_switch=mobile&redirect=http%253a%252f%252fspo337.com
e.ourger.com/?c=scene&a=link&id=47154371&url=https%3A%2F%2Fspo337.com/
zh-hk.guitarians.com/home/redirect/ubid/1015?r=http%3A%2F%2Fspo337.com
www.mastercleaningsupply.com/trigger.php?r_link=http%3A%2F%2Fspo337.com
www.cumshoter.com/cgi-bin/at3/out.cgi?id=98&tag=top&trade=https%3A%2F%2Fspo337.com
shp.hu/hpc_uj/click.php?ml=5&url=https%3A%2F%2Fspo337.com/
lrnews.ru/xgo.php?url=spo337.com
https://indonesianmma.com/modules/mod_jw_srfr/redir.php?url=https%3A%2F%2Fspo337.com
www.themza.com/redirect.php?r=spo337.com
lambda.ecommzone.com/lz/srr/00as0z/06e397d17325825ee6006c3c5ee495f922/actions/redirect.aspx?url=http://spo337.com
v.wcj.dns4.cn/?c=scene&a=link&id=8833621&url=https%3A%2F%2Fspo337.com/
embed.gabrielny.com/embedlink?key=f12cc3d5-e680-47b0-8914-a6ce19556f96&width=100%25&height=1200&division=bridal&no_chat=1&domain=http%3A%2F%2Fspo337.com
http://blackwhitepleasure.com/cgi-bin/atx/out.cgi?trade=https%3A%2F%2Fspo337.com
www.knet-web.net/m/pRedirect.php?uID=2&iID=259&iURL=http%3A%2F%2Fspo337.com
azlan.techdata.com/InTouch/GUIBnrT3/BnrTrackerPublic.aspx?CountryCode=18&BannerLangCulture=nl-nl&URL=https%3A%2F%2Fspo337.com/&Target=2&BannerId=41919&Zoneid=281&Parameters=&cos=Azlan
www.mintmail.biz/track/clicks/v2/?messageid=1427&cid=54657&url=https%3A%2F%2Fspo337.com
www.lzmfjj.com/Go.asp?URL=https%3A%2F%2Fspo337.com/
http://alexmovs.com/cgi-bin/atx/out.cgi?id=148&tag=topatx&trade=https%3A%2F%2Fspo337.com
marciatravessoni.com.br/revive/www/delivery/ck.php?ct=1&oaparams=2bannerid=40zoneid=16cb=fc1d72225coadest=https%3A%2F%2Fspo337.com/
https://sohodiffusion.com/mod/mod_langue.asp?action=francais&url=https%3A%2F%2Fspo337.com
www.mybunnies.net/te3/out.php?u=https%3A%2F%2Fspo337.com/
lubaczowskie.pl/rdir/?l=http%3A%2F%2Fspo337.com&lid=1315
www.kowaisite.com/bin/out.cgi?id=kyouhuna&url=https%3A%2F%2Fspo337.com/
www.ieslaasuncion.org/enlacesbuscar/clicsenlaces.asp?Idenlace=411&url=https%3A%2F%2Fspo337.com/
www.168web.com.tw/in/front/bin/adsclick.phtml?Nbr=114_02&URL=https%3A%2F%2Fspo337.com
asstomouth.guru/out.php?url=https%3A%2F%2Fspo337.com
advtest.exibart.com/adv/adv.php?id_banner=7201&link=http%3A%2F%2Fspo337.com
https://thairesidents.com/l.php?b=85&p=2,5&l=http%3A%2F%2Fspo337.com
www.latestnigeriannews.com/link_channel.php?channel=http%3A%2F%2Fspo337.com
www.haogaoyao.com/proad/default.aspx?url=spo337.com/
globalmedia51.ru/bitrix/redirect.php?goto=https%3A%2F%2Fspo337.com
citysafari.nl/Home/setCulture?language=en&returnUrl=http%3A%2F%2Fspo337.com
es.catholic.net/ligas/ligasframe.phtml?liga=https%3A%2F%2Fspo337.com
https://fishki.net/click?https%3A%2F%2Fspo337.com
catalog.flexcom.ru/go?z=36047&i=55&u=https%3A%2F%2Fspo337.com
www.wellvit.nl/response/forward/c1e41491e30c5af3c20f80a2af44e440.php?link=0&target=http%3A%2F%2Fspo337.com
go.flx1.com/click?id=1&m=11&pl=113&dmcm=16782&euid=16603484876&out=https%3A%2F%2Fspo337.com
moba-hgh.de/link2http.php?href=spo337.com/
https://gazetablic.com/ads/www/delivery/ck.php?ct=1&oaparams=2bannerid=34zoneid=26cb=0e0dfef92boadest=http%3A%2F%2Fspo337.com
watchvideo.co/go.php?url=https%3A%2F%2Fspo337.com
www.todoku.info/gpt/rank.cgi?mode=link&id=29649&url=https%3A%2F%2Fspo337.com
www.fotochki.com/redirect.php?go=spo337.com/
bayerwald.tips/plugins/bannerverwaltung/bannerredirect.php?bannerid=1&url=http%3A%2F%2Fspo337.com
www.ident.de/adserver/www/delivery/ck.php?ct=1&oaparams=2bannerid=76zoneid=2cb=8a18c95a9eoadest=http%3A%2F%2Fspo337.com
shop-uk.fmworld.com/Queue/Index?url=https%3A%2F%2Fspo337.com
nieuws.rvent.nl/bitmailer/statistics/mailstatclick/42261?link=https%3A%2F%2Fspo337.com
https://jobanticipation.com/jobclick/?RedirectURL=http%3A%2F%2Fspo337.com&Domain=jobanticipation.com
www.xsbaseball.com/tracker/index.html?t=ad&pool_id=3&ad_id=5&url=https%3A%2F%2Fspo337.com
eos.ru/bitrix/redirect.php?goto=https%3A%2F%2Fspo337.com
b.sm.su/click.php?bannerid=56&zoneid=10&source=&dest=https%3A%2F%2Fspo337.com/
https://paspn.net/default.asp?p=90&gmaction=40&linkid=52&linkurl=https%3A%2F%2Fspo337.com
www.dialogportal.com/Services/Forward.aspx?link=https%3A%2F%2Fspo337.com
www.poddebiczak.pl/?action=set-desktop&url=http%3A%2F%2Fspo337.com
ant53.ru/file/link.php?url=https%3A%2F%2Fspo337.com
www.docin.com/jsp_cn/mobile/tip/android_v1.jsp?forward=https%3A%2F%2Fspo337.com
old.magictower.ru/cgi-bin/redir/redir.pl?https%3A%2F%2Fspo337.com/
c.yam.com/srh/wsh/r.c?https%3A%2F%2Fspo337.com
www.jolletorget.no/J/l.php?l=http%3A%2F%2Fspo337.com
www.bobclubsau.com/cmshome/WebsiteAuditor/6744?url=spo337.com
www.moonbbs.com/dm/dmlink.php?dmurl=https%3A%2F%2Fspo337.com
www.sparetimeteaching.dk/forward.php?link=https%3A%2F%2Fspo337.com
www.bmwfanatics.ru/goto.php?l=https%3A%2F%2Fspo337.com
www.saabsportugal.com/forum/index.php?thememode=full;redirect=https%3A%2F%2Fspo337.com
www.interecm.com/interecm/tracker?op=click&id=5204.db2&url=https%3A%2F%2Fspo337.com/
cms.sive.it/Jump.aspx?gotourl=http%3A%2F%2Fspo337.com
largusladaclub.ru/go/url=https:/spo337.com
https://bethlehem-alive.com/abnrs/countguideclicks.cfm?targeturl=http%3A%2F%2Fspo337.com&businessid=29579
www.bookmark-favoriten.com/?goto=https%3A%2F%2Fspo337.com
shop.yuliyababich.eu/RU/ViewSwitcher/SwitchView?mobile=False&returnUrl=http%3A%2F%2Fspo337.com
https://urgankardesler.com/anasayfa/yonlen?link=http%3A%2F%2Fspo337.com
www.metalindex.ru/netcat/modules/redir/?&site=https%3A%2F%2Fspo337.com/
www.rprofi.ru/bitrix/redirect.php?goto=https%3A%2F%2Fspo337.com
www.postsabuy.com/autopost4/page/generate/?link=http%3A%2F%2Fspo337.com&list=PL9d7lAncfCDSkF4UPyhzO59Uh8cOoD-8q&fb_node=942812362464093&picture&name=%E0%B9%82%E0%B8%9B%E0%B8%A3%E0%B9%81%E0%B8%81%E0%B8%A3%E0%B8%A1%E0%B9%82%E0%B8%9E%E0%B8%AA%E0%B8%82%E0%B8%B2%E0%B8%A2%E0%B8%AA%E0%B8%B4%E0%B8%99%E0%B8%84%E0%B9%89%E0%B8%B2%E0%B8%AD%E0%B8%AD%E0%B8%99%E0%B9%84%E0%B8%A5%E0%B8%99%E0%B9%8C&caption=%E0%B9%80%E0%B8%A5%E0%B8%82%E0%B8%B2%E0%B8%AA%E0%B9%88%E0%B8%A7%E0%B8%99%E0%B8%95%E0%B8%B1%E0%B8%A7+%E0%B8%97%E0%B8%B5%E0%B8%84%E0%B8%B8%E0%B8%93%E0%B8%A5%E0%B8%B7%E0%B8%A1%E0%B9%84%E0%B8%A1%E0%B9%88%E0%B8%A5%E0%B8%87+Line+%40postsabuy&description=%E0%B8%A3%E0%B8%B2%E0%B8%84%E0%B8%B2%E0%B8%96%E0%B8%B9%E0%B8%81%E0%B8%97%E0%B8%B5%E0%B9%88%E0%B8%AA%E0%B8%B8%E0%B8%94%E0%B9%83%E0%B8%99+3+%E0%B9%82%E0%B8%A5%E0%B8%81+%E0%B8%AD%E0%B8%B4%E0%B8%AD%E0%B8%B4
www.perimeter.org/track.pdf?url=http%3A%2F%2Fspo337.com
forum.darievna.ru/go.php?https%3A%2F%2Fspo337.com
techlab.rarus.ru/bitrix/rk.php?goto=https%3A%2F%2Fspo337.com
www.spiritualforums.com/vb/redir.php?link=https%3A%2F%2Fspo337.com
www.review-mag.com/cdn/www/delivery/view.php?ct=1&oaparams=2bannerid=268zoneid=1cb=8c1317f219oadest=http%3A%2F%2Fspo337.com
belantara.or.id/lang/s/ID?url=https%3A%2F%2Fspo337.com/
https://jobsflagger.com/jobclick/?RedirectURL=http%3A%2F%2Fspo337.com
https://gpoltava.com/away/?go=spo337.com
services.nfpa.org/Authentication/GetSSOSession.aspx?return=https%3A%2F%2Fspo337.com/
http://spaceup.org/?wptouch_switch=mobile&redirect=https%3A%2F%2Fspo337.com
yarko-zhivi.ru/redirect?url=https%3A%2F%2Fspo337.com
https://kekeeimpex.com/Home/ChangeCurrency?urls=http%3A%2F%2Fspo337.com&cCode=GBP&cRate=77.86247
mycounter.com.ua/go.php?https%3A%2F%2Fspo337.com
l2base.su/go?https%3A%2F%2Fspo337.com
www.duomodicagliari.it/reg_link.php?link_ext=http%3A%2F%2Fspo337.com&prov=1
assine.hostnet.com.br/cadastro/?rep=17&url=https%3A%2F%2Fspo337.com/
www.dvnlp.de/profile/gruppe/redirect/5?url=spo337.com
www.smkn5pontianak.sch.id/redirect/?alamat=https%3A%2F%2Fspo337.com
www.cheapdealuk.co.uk/go.php?url=https%3A%2F%2Fspo337.com/
bilometro.brksedu.com.br/tracking?url=spo337.com&zorigem=hotsite-blackfriday
tramplintk.ru/bitrix/redirect.php?goto=https%3A%2F%2Fspo337.com
cnc.extranet.gencat.cat/treball_cnc/AppJava/FileDownload.do?pdf=http%3A%2F%2Fspo337.com&codi_cnv=9998045
www.gamecollections.co.uk/search/redirect.php?retailer=127&deeplink=https%3A%2F%2Fspo337.com
bearcong.no1.sexy/hobby-delicious/rank.cgi?mode=link&id=19&url=https%3A%2F%2Fspo337.com/
www.omschweiz.ch/select-your-country?publicUrl=http%3A%2F%2Fspo337.com
https://anacolle.net/?wptouch_switch=desktop&redirect=http%3A%2F%2Fspo337.com
www.cubamusic.com/Home/ChangeLanguage?lang=es-ES&returnUrl=http%3A%2F%2Fspo337.com
expoclub.ru/bitrix/redirect.php?goto=https%3A%2F%2Fspo337.com
365sekretov.ru/redirect.php?action=url&goto=spo337.com%20
www.sgdrivingtest.com/redirect.php?page=spo337.com/
https://slashwrestling.com/cgi-bin/redirect.cgi?https%3A%2F%2Fspo337.com
lorena-kuhni.kz/redirect?link=spo337.com/
www.webshoptrustmark.fr/Change/en?returnUrl=http%3A%2F%2Fspo337.com
www.widgetinfo.net/read.php?sym=FRA_LM&url=http%3A%2F%2Fspo337.com
affiliates.kanojotoys.com/affiliate/scripts/click.php?a_aid=widdi77&desturl=http%3A%2F%2Fspo337.com
www.my-sms.ru/ViewSwitcher/SwitchView?mobile=False&returnUrl=http%3A%2F%2Fspo337.com&rel=external
www.genderpsychology.com/http/spo337.com/
http://chtbl.com/track/118167/spo337.com/
www.360wichita.com/amp-banner-tracking?adid=192059&url=http%3A%2F%2Fspo337.com
www.cheek.co.jp/location/location.php?id=keibaseminar&url=https%3A%2F%2Fspo337.com
www.quantixtickets3.com/php-bin-8/kill_session_and_redirect.php?redirect=https%3A%2F%2Fspo337.com/
www.photokonkurs.com/cgi-bin/out.cgi?url=https%3A%2F%2Fspo337.com/
www.pcreducator.com/Common/SSO.aspx?returnUrl=https%3A%2F%2Fspo337.com/
go.eniro.dk/lg/ni/cat-2611/http:/spo337.com
www.fisherly.com/redirect?type=website&ref=listing_detail&url=https%3A%2F%2Fspo337.com
www.modernipanelak.cz/?b=618282165&redirect=https%3A%2F%2Fspo337.com/
h5.hbifeng.com/index.php?c=scene&a=link&id=14240604&url=https%3A%2F%2Fspo337.com
www.bkdc.ru/bitrix/redirect.php?event1=news_out&event2=32reg.roszdravnadzor.ru/&event3=A0A0B5A09180D0%A09582A0BBA1A085%D0E2A084D0D1C2D0%A085+A0A0B5A182B0A0%C2D0D0D096+A1A0BBA0B180D0%A09795+A0A0B0A09582A1%D1D0D0D0A182B5+A0A091A08695A0%D1D0A6A185A0A085%D0D1D0D082A1A085%D0D0D1D0A095B1A0%C2D0D0D091&goto=https%3A%2F%2Fspo337.com/
d-click.artenaescola.org.br/u/3806/290/32826/1416_0/53052/?url=https%3A%2F%2Fspo337.com
www.morroccoaffiliate.com/aff.php?id=883&url=https%3A%2F%2Fspo337.com/
www.omegon.eu/de/?r=http%3A%2F%2Fspo337.com
www.flooble.com/cgi-bin/clicker.pl?id=grabbadl&url=https%3A%2F%2Fspo337.com/
gameshock.jeez.jp/rank.cgi?mode=link&id=307&url=https%3A%2F%2Fspo337.com/
https://tripyar.com/go.php?https%3A%2F%2Fspo337.com/
www.floridafilmofficeinc.com/?goto=https%3A%2F%2Fspo337.com/
tsvc1.teachiworld.com/bin/checker?mode=4&module=11&mailidx=19130&dmidx=0&emidx=0&service=0&cidx=&etime=20120328060000&seqidx=3&objidx=22&encoding=0&url=https%3A%2F%2Fspo337.com/
rechner.atikon.at/lbg.at/newsletter/linktracking?subscriber=&delivery=38116&url=http%3A%2F%2Fspo337.com
www.sportsbook.ag/ctr/acctmgt/pl/openLink.ctr?ctrPage=https%3A%2F%2Fspo337.com/
www.ra2d.com/directory/redirect.asp?id=596&url=https%3A%2F%2Fspo337.com/
www.anorexicnudes.net/cgi-bin/atc/out.cgi?u=https%3A%2F%2Fspo337.com
www.zjjiajiao.com.cn/ad/adredir.asp?url=https%3A%2F%2Fspo337.com/
https://hakobo.com/wp/?wptouch_switch=desktop&redirect=http%3A%2F%2Fspo337.com
t.agrantsem.com/tt.aspx?cus=216&eid=1&p=216-2-71016b553a1fa2c9.3b14d1d7ea8d5f86&d=https%3A%2F%2Fspo337.com/
www.tagirov.org/out.php?url=spo337.com/
startlist.club/MSF/Language/Set?languageIsoCode=en&returnUrl=http%3A%2F%2Fspo337.com
www.tetsumania.net/search/rank.cgi?mode=link&id=947&url=https%3A%2F%2Fspo337.com/
www.brainlanguage-sa.com/setcookie.php?lang=en&file=https%3A%2F%2Fspo337.com/
www.asensetranslations.com/modules/babel/redirect.php?newlang=en_US&newurl=https%3A%2F%2Fspo337.com/
https://www.nbda.org/?URL=spo337.com/
https://evoautoshop.com/?wptouch_switch=mobile&redirect=http%3A%2F%2Fspo337.com
http://beautycottageshop.com/change.php?lang=cn&url=https%3A%2F%2Fspo337.com/
http://1000love.net/lovelove/link.php?url=https%3A%2F%2Fspo337.com/
https://www.contactlenshouse.com/currency.asp?c=CAD&r=http%3A%2F%2Fspo337.com
https://www.bartaz.lt/wp-content/plugins/clikstats/ck.php?Ck_id=70&Ck_lnk=https%3A%2F%2Fspo337.com
https://sogo.i2i.jp/link_go.php?url=https%3A%2F%2Fspo337.com/
https://malehealth.ie/redirect/?age=40&part=waist&illness=obesity&refer=https%3A%2F%2Fspo337.com/
https://magicode.me/affiliate/go?url=https%3A%2F%2Fspo337.com/
forum.marillion.com/forum/index.php?thememode=full;redirect=https%3A%2F%2Fspo337.com
https://jobregistry.net/jobclick/?RedirectURL=http%3A%2F%2Fspo337.com&Domain=jobregistry.net&rgp_m=title13&et=4495
https://www.goatzz.com/adredirect.aspx?adType=SiteAd&ItemID=9595&ReturnURL=https%3A%2F%2Fspo337.com/
https://www.thislife.net/cgi-bin/webcams/out.cgi?id=playgirl&url=https%3A%2F%2Fspo337.com/
https://www.dans-web.nu/klick.php?url=https%3A%2F%2Fspo337.com/
https://voobrajulya.ru/bitrix/redirect.php?goto=https%3A%2F%2Fspo337.com
www.resnichka.ru/partner/go.php?https%3A%2F%2Fspo337.com
www.nafta-him.com/bitrix/redirect.php?goto=https%3A%2F%2Fspo337.com/
www.kyoto-osaka.com/search/rank.cgi?mode=link&id=9143&url=https%3A%2F%2Fspo337.com/
www.findingfarm.com/redir?url=https%3A%2F%2Fspo337.com/
www.fairpoint.net/~jensen1242/gbook/go.php?url=https%3A%2F%2Fspo337.com/
www.cnainterpreta.it/redirect.asp?url=https%3A%2F%2Fspo337.com
https://edcommunity.ru/bitrix/rk.php?goto=https%3A%2F%2Fspo337.com/
particularcareers.co.uk/jobclick/?RedirectURL=http%3A%2F%2Fspo337.com
https://bsaonline.com/MunicipalDirectory/SelectUnit?unitId=411&returnUrl=http%3A%2F%2Fspo337.com&sitetransition=true
www.elit-apartament.ru/go?https%3A%2F%2Fspo337.com
sendai.japansf.net/rank.cgi?mode=link&id=1216&url=http%3A%2F%2Fspo337.com
www.dresscircle-net.com/psr/rank.cgi?mode=link&id=14&url=https%3A%2F%2Fspo337.com
www.counterwelt.com/charts/click.php?user=14137&link=https%3A%2F%2Fspo337.com/
fms.csonlineschool.com.au/changecurrency/1?returnurl=https%3A%2F%2Fspo337.com
www.jagat.co.jp/analysis/analysis.php?url=http%3A%2F%2Fspo337.com
https://vse-doski.com/redirect/?go=https%3A%2F%2Fspo337.com
www.karatetournaments.net/link7.asp?LRURL=https%3A%2F%2Fspo337.com/&LRTYP=O
simracing.su/go/?https%3A%2F%2Fspo337.com
click.cheshi.com/go.php?proid=218&clickid=1393306648&url=https%3A%2F%2Fspo337.com/
fdeam.finanzen-partnerprogramm.de/tracking/?as_id=9257&c_id=595&url=https%3A%2F%2Fspo337.com
www.horsesmouth.com/LinkTrack.aspx?u=https%3A%2F%2Fspo337.com
d-click.fiemg.com.br/u/18081/131/75411/137_0/82cb7/?url=https%3A%2F%2Fspo337.com/
www.winxuan.com/page/cps/eqifacookieinterface.jsp?from=yiqifa&wid=8&url=https%3A%2F%2Fspo337.com
www.packmage.net/uc/goto/?url=https%3A%2F%2Fspo337.com
my.9991.com/login_for_index_0327.php?action=logout&forward=https%3A%2F%2Fspo337.com/
www.sdchamber.biz/admin/mod_newsletter/redirect.aspx?message_id=986&redirect=https%3A%2F%2Fspo337.com
www.naturum.co.jp/ad/linkshare/?siteID=p_L785d6UQY-V4Fh4Rxs7wNzOPgtzv95Tg&lsurl=http%3A%2F%2Fspo337.com
www.d-e-a.eu/newsletter/redirect.php?link=https%3A%2F%2Fspo337.com
www.bom.ai/goweburl?go=http%3A%2F%2Fspo337.com
enews2.sfera.net/newsletter/redirect.php?id=sabricattani@gmail.com_0000006566_144&link=https%3A%2F%2Fspo337.com/
underwater.com.au/redirect_url/id/7509/?redirect=https%3A%2F%2Fspo337.com
https://eqsoftwares.com/languages/setlanguage?languagesign=en&redirect=https%3A%2F%2Fspo337.com
www.jagdambasarees.com/Home/ChangeCurrency?urls=http%3A%2F%2Fspo337.com&cCode=MYR&cRate=14.554
www.priegeltje.nl/gastenboek/go.php?url=https%3A%2F%2Fspo337.com
www.serie-a.ru/bitrix/redirect.php?goto=https%3A%2F%2Fspo337.com
www.rz114.cn/url.html?url=https%3A%2F%2Fspo337.com/
www.greatdealsindia.com/redirects/infibeam.aspx?url=https%3A%2F%2Fspo337.com
rs.345kei.net/rank.php?id=37&mode=link&url=https%3A%2F%2Fspo337.com/
webapp.jgz.la/?c=scene&a=link&id=8665466&url=https%3A%2F%2Fspo337.com
scribe.mmonline.io/click?evt_nm=Clicked+Registration+Completion&evt_typ=clickEmail&app_id=m4marry&eml_sub=Registration+Successful&usr_did=4348702&cpg_sc=NA&cpg_md=email&cpg_nm=&cpg_cnt=&cpg_tm=NA&link_txt=Live+Chat&em_type=Notification&url=https%3A%2F%2Fspo337.com
polo-v1.feathr.co/v1/analytics/crumb?flvr=email_link_click&rdr=https%3A%2F%2Fspo337.com/
sintesi.provincia.mantova.it/portale/LinkClick.aspx?link=https%3A%2F%2Fspo337.com
www.surinenglish.com/backend/conectar.php?url=https%3A%2F%2Fspo337.com
www.reference-cannabis.com/interface/sortie.php?adresse=https%3A%2F%2Fspo337.com/
login.0×69416d.co.uk/sso/logout?tenantId=tnl&gotoUrl=http%3A%2F%2Fspo337.com&domain=0×69416d.co.uk
blog.link-usa.jp/emi?wptouch_switch=mobile&redirect=https%3A%2F%2Fspo337.com/
http://damki.net/go/?https%3A%2F%2Fspo337.com
opensesame.wellymulia.zaxaa.com/b/66851136?s=1&redir=https%3A%2F%2Fspo337.com
ism3.infinityprosports.com/ismdata/2009100601/std-sitebuilder/sites/200901/www/en/tracker/index.html?t=ad&pool_id=1&ad_id=112&url=https%3A%2F%2Fspo337.com
apps.cancaonova.com/ads/www/delivery/ck.php?ct=1&oaparams=2bannerid=149zoneid=20cb=87d2c6208doadest=http%3A%2F%2Fspo337.com
www.lespritjardin.be/?advp_click_bimage_id=19&url=http%253a%252f%252fspo337.com&shortcode_id=10
https://careerchivy.com/jobclick/?RedirectURL=http%3A%2F%2Fspo337.com
shinsekai.type.org/?wptouch_switch=desktop&redirect=https%3A%2F%2Fspo337.com/
www.etaigou.com/turn2.php?ad_id=276&link=https%3A%2F%2Fspo337.com
moscowdesignmuseum.ru/bitrix/rk.php?goto=https%3A%2F%2Fspo337.com
https://clients1.google.es/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.co.uk/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.co.jp/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.fr/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.it/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.br/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.co.in/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.ca/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.ru/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.hk/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.au/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.co.id/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.nl/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.tw/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.pl/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.be/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.co.th/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.at/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.cz/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.se/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.mx/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.ch/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.vn/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.pt/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.ua/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.tr/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.ro/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.my/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.gr/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.dk/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.hu/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.ar/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.fi/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.co.il/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.co.nz/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.co.za/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.cl/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.co/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.sg/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.ie/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.sk/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.co.kr/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.ph/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.no/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.lt/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.bg/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.sa/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.hr/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.pe/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.ae/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.co.ve/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.ee/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.pk/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.rs/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.eg/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.si/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.ec/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.qa/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.pr/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.mu/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.li/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.lv/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.mn/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.gt/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.co.cr/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.uy/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.lu/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.ba/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.is/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.dz/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.kg/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.co.ke/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.az/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.ng/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.np/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.mt/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.bi/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.by/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.bd/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.as/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.do/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.kz/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.co.ma/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.jo/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.lk/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.cu/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.ai/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.gi/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.cf/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.ni/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.md/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.mg/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.la/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.jm/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.vc/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.tj/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.cy/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.sv/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.rw/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.om/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.ps/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.bo/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.tk/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.co.mz/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.bs/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.mk/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.co.bw/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.al/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.sm/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.co.zw/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.tm/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.bh/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.af/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.fj/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.kh/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.cg/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.ki/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.mw/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.kw/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.bf/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.lb/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.co.ls/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.ms/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.ci/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.dm/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.sb/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.co.vi/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.so/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.nu/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.dj/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.hn/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.nr/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.co.tz/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.mv/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.tn/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.sc/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.py/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.sn/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.am/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.ad/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.gh/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.bz/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.iq/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.to/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.bn/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.cat/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.sh/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.cm/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.gg/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.co.ug/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.ly/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.co.uz/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.co.zm/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.na/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.ag/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.me/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.cd/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.fm/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.co.ck/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.et/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.pa/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.je/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.gl/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.ge/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.ht/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.im/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.gy/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.sl/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.bj/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.ml/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.cv/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.tl/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.mm/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.bt/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.ac/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.st/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.td/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.com.pg/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.co.ao/url?q=https%3A%2F%2Fspo337.com/
https://clients1.google.gp/url?q=https%3A%2F%2Fspo337.com/
https://www.gudarjavalambre.com/sections/miscelany/link.php?url=https%3A%2F%2Fspo337.com
www.interracialhall.com/cgi-bin/atx/out.cgi?trade=https%3A%2F%2Fspo337.com
https://www.tumimusic.com/link.php?url=https%3A%2F%2Fspo337.com/
www.glorioustronics.com/redirect.php?link=https%3A%2F%2Fspo337.com/
mail.resen.gov.mk/redir.hsp?url=https%3A%2F%2Fspo337.com/
https://www.pompengids.net/followlink.php?id=495&link=https%3A%2F%2Fspo337.com
https://www.hirforras.net/scripts/redir.php?url=https%3A%2F%2Fspo337.com/
http://3dcreature.com/cgi-bin/at3/out.cgi?id=187&trade=https%3A%2F%2Fspo337.com/
https://nowlifestyle.com/redir.php?k=9a4e080456dabe5eebc8863cde7b1b48&url=https%3A%2F%2Fspo337.com
http://slipknot1.info/go.php?url=https%3A%2F%2Fspo337.com
www.acutenet.co.jp/cgi-bin/lcount/lcounter.cgi?link=https%3A%2F%2Fspo337.com
http://news-matome.com/method.php?method=1&url=https%3A%2F%2Fspo337.com/
https://lifecollection.top/site/gourl?url=https%3A%2F%2Fspo337.com
speakrus.ru/links.php?go=https%3A%2F%2Fspo337.com/
https://lens-club.ru/link?go=https%3A%2F%2Fspo337.com/
iphoneapp.impact.co.th/i/r.php?u=https%3A%2F%2Fspo337.com
www.rencai8.com/web/jump_to_ad_url.php?id=642&url=https%3A%2F%2Fspo337.com/
http://crackstv.com/redirect.php?bnn=CabeceraHyundai&url=https%3A%2F%2Fspo337.com
https://www.kichink.com/home/issafari?uri=https%3A%2F%2Fspo337.com/
https://blogranking.fc2.com/out.php?id=1032500&url=https%3A%2F%2Fspo337.com/
5cfxm.hxrs6.servertrust.com/v/affiliate/setCookie.asp?catId=1180&return=https%3A%2F%2Fspo337.com/
https://www.morhipo.com/shared/partnercookie?k=gort&url=https%3A%2F%2Fspo337.com
https://www.luckyplants.com/cgi-bin/toplist/out.cgi?id=rmontero&url=https%3A%2F%2Fspo337.com
https://webankety.cz/dalsi.aspx?site=https%3A%2F%2Fspo337.com
https://www.rexart.com/cgi-rexart/al/affiliates.cgi?aid=872&redirect=https%3A%2F%2Fspo337.com
https://ulfishing.ru/forum/go.php?https%3A%2F%2Fspo337.com
https://www.ab-search.com/rank.cgi?mode=link&id=107&url=https%3A%2F%2Fspo337.com/
https://www.studyrama.be/tracking.php?origine=ficheform5683&lien=http://spo337.com/
https://miyagi.lawyer-search.tv/details/linkchk.aspx?type=o&url=https%3A%2F%2Fspo337.com/
https://www.snwebcastcenter.com/event/page/count_download_time.php?url=https%3A%2F%2Fspo337.com/
https://runkeeper.com/apps/authorize?redirect_uri=https%3A%2F%2Fspo337.com
https://www.emiratesvoice.com/footer/comment_like_dislike_ajax/?code=like&commentid=127&redirect=https%3A%2F%2Fspo337.com/
https://envios.uces.edu.ar/control/click.mod.php?id_envio=8147&email=gramariani@gmail.com&url=http://spo337.com/
https://www.weather.net/cgi-bin/redir?https%3A%2F%2Fspo337.com
https://gaggedtop.com/cgi-bin/top/out.cgi?ses=sBZFnVYGjF&id=206&url=https%3A%2F%2Fspo337.com/
https://e-bike-test.net/wp-content/plugins/AND-AntiBounce/redirector.php?url=https%3A%2F%2Fspo337.com/
https://www.cheerunion.org/tracker/index.html?t=ad&pool_id=2&ad_id=5&url=https%3A%2F%2Fspo337.com/
https://area51.to/go/out.php?s=100&l=site&u=https%3A%2F%2Fspo337.com/
https://games4ever.3dn.ru/go?https%3A%2F%2Fspo337.com/
https://de.reasonable.shop/SetCurrency.aspx?currency=CNY&returnurl=https%3A%2F%2Fspo337.com/
https://www.podcastone.com/site/rd?satype=40&said=4&aaid=email&camid=-4999600036534929178&url=https%3A%2F%2Fspo337.com/
https://monarchbeachmembers.play18.com/ViewSwitcher/SwitchView?mobile=False&returnUrl=https%3A%2F%2Fspo337.com/
https://d.agkn.com/pixel/2389/?che=2979434297&col=22204979,1565515,238211572,435508400,111277757&l1=https%3A%2F%2Fspo337.com/
https://www.pcreducator.com/Common/SSO.aspx?returnUrl=https%3A%2F%2Fspo337.com/
https://horsesmouth.com/LinkTrack.aspx?u=https%3A%2F%2Fspo337.com/
https://soom.cz/projects/get2mail/redir.php?id=c2e52da9ad&url=https%3A%2F%2Fspo337.com/
https://www.iaai.com/VehicleInspection/InspectionProvidersUrl?name=AA%20Transit%20Pros%20Inspection%20Service&url=https%3A%2F%2Fspo337.com/
https://shop.merchtable.com/users/authorize?return_url=https%3A%2F%2Fspo337.com/
https://nudewwedivas.forumcommunity.net/m/ext.php?url=https%3A%2F%2Fspo337.com/
https://cztt.ru/redir.php?url=https%3A%2F%2Fspo337.com/
https://ageoutloud.gms.sg/visit.php?item=54&uri=https%3A%2F%2Fspo337.com/
https://multimedia.inrap.fr/redirect.php?li=287&R=https%3A%2F%2Fspo337.com/
https://mobo.osport.ee/Home/SetLang?lang=cs&returnUrl=https%3A%2F%2Fspo337.com/
https://www.shop-bell.com/out.php?id=kibocase&category=ladies&url=https%3A%2F%2Fspo337.com/
https://m.addthis.com/live/redirect/?url=https%3A%2F%2Fspo337.com/
https://mobilize.org.br/handlers/anuncioshandler.aspx?anuncio=55&canal=2&redirect=https%3A%2F%2Fspo337.com/
https://bemidji.bigdealsmedia.net/include/sort.php?return_url=https%3A%2F%2Fspo337.com/&sort=a:3:{s:9:%E2%80%9Ddirection%E2%80%9D;s:3:%E2%80%9DASC%E2%80%9D;s:5:%E2%80%9Dfield%E2%80%9D;s:15:%E2%80%9DItems.PriceList%E2%80%9D;s:5:%E2%80%9Dlabel%E2%80%9D;s:9:%E2%80%9Dvalue_asc%E2%80%9D;}
https://hslda.org/content/a/LinkTracker.aspx?id=4015475&appeal=385&package=36&uri=https%3A%2F%2Fspo337.com/
https://www.sign-in-china.com/newsletter/statistics.php?type=mail2url&bs=88&i=114854&url=https%3A%2F%2Fspo337.com/
https://panarmenian.net/eng/tofv?tourl=https%3A%2F%2Fspo337.com/
http://mientaynet.com/advclick.php?o=textlink&u=15&l=https%3A%2F%2Fspo337.com/
https://atlanticleague.com/tracker/index.html?t=ad&pool_id=11&ad_id=5&url=https%3A%2F%2Fspo337.com/
https://www.castellodivezio.it/lingua.php?lingua=EN&url=https%3A%2F%2Fspo337.com/
https://www.kvinfo.dk/visit.php?linkType=2&linkValue=https%3A%2F%2Fspo337.com/
https://www.dodeley.com/?action=show_ad&url=https%3A%2F%2Fspo337.com/
http://chtbl.com/track/118167/https%3A%2F%2Fspo337.com/
https://www.tremblant.ca/Shared/LanguageSwitcher/ChangeCulture?culture=en&url=https%3A%2F%2Fspo337.com/
https://www.winxuan.com/page/cps/eqifacookieinterface.jsp?from=yiqifa&wid=8&url=https%3A%2F%2Fspo337.com/
https://moscowdesignmuseum.ru/bitrix/rk.php?goto=https%3A%2F%2Fspo337.com/
https://igert2011.videohall.com/to_client?target=https%3A%2F%2Fspo337.com/
https://ovatu.com/e/c?url=https%3A%2F%2Fspo337.com/
https://primepartners.globalprime.com/afs/wcome.php?c=427|0|1&e=GP204519&url=https%3A%2F%2Fspo337.com/
https://ltp.org/home/setlocale?locale=en&returnUrl=https%3A%2F%2Fspo337.com/
https://www.morhipo.com/shared/partnercookie?k=gort&url=https%3A%2F%2Fspo337.com/
https://rs.businesscommunity.it/snap.php?u=https%3A%2F%2Fspo337.com/
https://www.aiac.world/pdf/October-December2015Issue?pdf_url=https%3A%2F%2Fspo337.com/
https://fdeam.finanzen-partnerprogramm.de/tracking/?as_id=9257&c_id=595&url=https%3A%2F%2Fspo337.com/
https://pdcn.co/e/https%3A%2F%2Fspo337.com/
https://www.swipeclock.com/sc/cookie.asp?sitealias=79419397&redirect=https%3A%2F%2Fspo337.com/
https://home.uceusa.com/Redirect.aspx?r=https%3A%2F%2Fspo337.com/
https://www.seankenney.com/include/jump.php?num=https%3A%2F%2Fspo337.com/
https://runningcheese.com/go?url=https%3A%2F%2Fspo337.com/
https://cingjing.fun-taiwan.com/AdRedirector.aspx?padid=303&target=https%3A%2F%2Fspo337.com/
https://rtb-asiamax.tenmax.io/bid/click/1462922913409/e95f2c30-1706-11e6-a9b4-a9f6fe33c6df/3456/5332/?rUrl=https%3A%2F%2Fspo337.com/
https://api.webconnex.com/v1/postmaster/track/click/4f8036d14ee545798599c8921fbfcd22/db005310dba511e89fb606f49a4ee876?url=https%3A%2F%2Fspo337.com/
https://www.lecake.com/stat/goto.php?url=https%3A%2F%2Fspo337.com/
http://www.youtube.de/redirect?event=channel_description&q=https://sites.google.com/view/spo337
http://www.youtube.com/redirect?event=channel_description&q=https://sites.google.com/view/spo337
http://www.youtube.co/redirect?event=channel_description&q=https://sites.google.com/view/spo337
http://www.youtube.es/redirect?event=channel_description&q=https://sites.google.com/view/spo337
http://www.youtube.ca/redirect?event=channel_description&q=https://sites.google.com/view/spo337
http://www.youtube.nl/redirect?event=channel_description&q=https://sites.google.com/view/spo337
http://www.youtube.pl/redirect?event=channel_description&q=https://sites.google.com/view/spo337
http://www.youtube.ch/redirect?event=channel_description&q=https://sites.google.com/view/spo337
http://www.youtube.be/redirect?event=channel_description&q=https://sites.google.com/view/spo337
http://www.youtube.se/redirect?event=channel_description&q=https://sites.google.com/view/spo337
http://www.youtube.dk/redirect?event=channel_description&q=https://sites.google.com/view/spo337
http://www.youtube.pt/redirect?event=channel_description&q=https://sites.google.com/view/spo337
http://www.youtube.no/redirect?event=channel_description&q=https://sites.google.com/view/spo337
http://www.youtube.gr/redirect?event=channel_description&q=https://sites.google.com/view/spo337
http://www.youtube.cl/redirect?event=channel_description&q=https://sites.google.com/view/spo337
http://www.youtube.at/redirect?event=channel_description&q=https://sites.google.com/view/spo337
http://www.youtube.bg/redirect?event=channel_description&q=https://sites.google.com/view/spo337
http://www.youtube.sk/redirect?event=channel_description&q=https://sites.google.com/view/spo337
http://www.youtube.rs/redirect?event=channel_description&q=https://sites.google.com/view/spo337
http://www.youtube.lt/redirect?event=channel_description&q=https://sites.google.com/view/spo337
http://www.youtube.si/redirect?event=channel_description&q=https://sites.google.com/view/spo337
http://www.youtube.hr/redirect?event=channel_description&q=https://sites.google.com/view/spo337
http://www.youtube.ee/redirect?event=channel_description&q=https://sites.google.com/view/spo337
http://www.youtube.lu/redirect?event=channel_description&q=https://sites.google.com/view/spo337
http://www.youtube.tn/redirect?event=channel_description&q=https://sites.google.com/view/spo337
http://www.youtube.co.ke/redirect?event=channel_description&q=https://sites.google.com/view/spo337
http://www.youtube.co.cr/redirect?event=channel_description&q=https://sites.google.com/view/spo337
http://www.youtube.kz/redirect?event=channel_description&q=https://sites.google.com/view/spo337
http://www.youtube.cat/redirect?event=channel_description&q=https://sites.google.com/view/spo337
http://www.youtube.ge/redirect?event=channel_description&q=https://sites.google.com/view/spo337
https://www.youtube.com/redirect?event=channeldescription&q=https://sites.google.com/view/spo337&gl=ml
https://www.youtube.com/redirect?q=https://sites.google.com/view/spo337
https://www.youtube.com/redirect?q=https://sites.google.com/view/spo337&gl=DE
https://www.youtube.com/redirect?q=https://sites.google.com/view/spo337&gl=IT
https://www.youtube.com/redirect?q=https%3A%2F%2Ftinyurl.com/yc2ypppz&gl=AR
https://www.youtube.at/redirect?q=https://sites.google.com/view/spo337
https://www.youtube.ch/redirect?q=https://sites.google.com/view/spo337
https://www.youtube.fr/redirect?q=https://sites.google.com/view/spo337
https://www.youtube.es/redirect?q=https://sites.google.com/view/spo337
https://www.youtube.jp/redirect?q=https://sites.google.com/view/spo337
https://www.youtube.co.uk/redirect?q=https://sites.google.com/view/spo337
https://www.youtube.ru/redirect?q=https://sites.google.com/view/spo337
https://www.youtube.pl/redirect?q=https://sites.google.com/view/spo337
https://www.youtube.gr/redirect?q=https://sites.google.com/view/spo337
https://www.youtube.nl/redirect?q=https://sites.google.com/view/spo337
https://www.youtube.ca/redirect?q=https://sites.google.com/view/spo337
https://www.youtube.cz/redirect?q=https://sites.google.com/view/spo337
https://www.youtube.com/redirect?q=https://sites.google.com/view/spo337&gl=AU
https://www.youtube.com.tw/redirect?q=https://sites.google.com/view/spo337
http://toolbarqueries.google.com.uy/url?sa=t&url=https://sites.google.com/view/spo337
http://toolbarqueries.google.com.tw/url?sa=t&url=https://sites.google.com/view/spo337
http://toolbarqueries.google.com.tr/url?sa=t&url=https://sites.google.com/view/spo337
http://toolbarqueries.google.com.sa/url?sa=t&url=https://sites.google.com/view/spo337
http://toolbarqueries.google.com.py/url?sa=t&url=https://sites.google.com/view/spo337
http://toolbarqueries.google.com.pr/url?sa=t&url=https://sites.google.com/view/spo337
http://toolbarqueries.google.com.pk/url?sa=t&url=https://sites.google.com/view/spo337
http://toolbarqueries.google.com.pe/url?sa=t&url=https://sites.google.com/view/spo337
http://toolbarqueries.google.com.my/url?sa=t&url=https://sites.google.com/view/spo337
http://toolbarqueries.google.com.hk/url?sa=t&url=https://sites.google.com/view/spo337
http://toolbarqueries.google.com.gt/url?sa=t&url=https://sites.google.com/view/spo337
http://toolbarqueries.google.com.gh/url?sa=t&url=https://sites.google.com/view/spo337
https://clients1.google.com.ar/url?sa=t&url=https://sites.google.com/view/spo337
https://clients1.google.com.ag/url?sa=t&url=https://sites.google.com/view/spo337
https://clients1.google.co.zm/url?sa=t&url=https://sites.google.com/view/spo337
https://clients1.google.co.za/url?sa=t&url=https://sites.google.com/view/spo337
https://clients1.google.co.ve/url?sa=t&url=https://sites.google.com/view/spo337
https://clients1.google.co.uz/url?sa=t&url=https://sites.google.com/view/spo337
https://clients1.google.co.ug/url?sa=t&url=https://sites.google.com/view/spo337
https://clients1.google.co.th/url?sa=t&url=https://sites.google.com/view/spo337
https://clients1.google.co.nz/url?sa=t&url=https://sites.google.com/view/spo337
https://clients1.google.co.kr/url?sa=t&url=https://sites.google.com/view/spo337
https://clients1.google.co.ke/url?sa=t&url=https://sites.google.com/view/spo337
https://clients1.google.co.il/url?sa=t&url=https://sites.google.com/view/spo337
https://clients1.google.co.id/url?sa=t&url=https://sites.google.com/view/spo337
https://clients1.google.co.cr/url?sa=t&url=https://sites.google.com/view/spo337
https://clients1.google.co.ck/url?sa=t&url=https://sites.google.com/view/spo337
https://cse.google.co.ck/url?sa=t&url=https://sites.google.com/view/spo337
https://cse.google.co.bw/url?sa=t&url=https://sites.google.com/view/spo337
https://cse.google.cm/url?sa=t&url=https://sites.google.com/view/spo337
https://cse.google.cl/url?sa=t&url=https://sites.google.com/view/spo337
https://cse.google.ci/url?sa=t&url=https://sites.google.com/view/spo337
https://cse.google.ch/url?sa=t&url=https://sites.google.com/view/spo337
https://cse.google.ch/url?sa=i&url=https://sites.google.com/view/spo337
https://cse.google.cg/url?sa=t&url=https://sites.google.com/view/spo337
https://cse.google.cd/url?sa=t&url=https://sites.google.com/view/spo337
https://cse.google.by/url?sa=t&url=https://sites.google.com/view/spo337
https://cse.google.bs/url?sa=t&url=https://sites.google.com/view/spo337
https://cse.google.bi/url?sa=t&url=https://sites.google.com/view/spo337
https://cse.google.bg/url?sa=t&url=https://sites.google.com/view/spo337
https://cse.google.be/url?sa=t&url=https://sites.google.com/view/spo337
https://cse.google.be/url?sa=i&url=https://sites.google.com/view/spo337
https://cse.google.ba/url?sa=t&url=https://sites.google.com/view/spo337
https://cse.google.az/url?sa=t&url=https://sites.google.com/view/spo337
https://cse.google.at/url?sa=t&url=https://sites.google.com/view/spo337
https://images.google.ca/url?sa=t&url=https://sites.google.com/view/spo337
https://images.google.by/url?sa=t&url=https://sites.google.com/view/spo337
https://images.google.bs/url?sa=t&url=https://sites.google.com/view/spo337
https://images.google.bi/url?sa=t&url=https://sites.google.com/view/spo337
https://images.google.bg/url?sa=t&url=https://sites.google.com/view/spo337
https://images.google.bf/url?sa=t&url=https://sites.google.com/view/spo337
https://images.google.be/url?sa=t&url=https://sites.google.com/view/spo337
https://images.google.ba/url?sa=t&url=https://sites.google.com/view/spo337
https://images.google.ba/url?q=https://sites.google.com/view/spo337
https://images.google.az/url?sa=t&url=https://sites.google.com/view/spo337
https://images.google.at/url?sa=t&url=https://sites.google.com/view/spo337
https://images.google.as/url?sa=t&url=https://sites.google.com/view/spo337
https://images.google.am/url?sa=t&url=https://sites.google.com/view/spo337
https://images.google.al/url?sa=t&url=https://sites.google.com/view/spo337
https://images.google.ae/url?sa=t&url=https://sites.google.com/view/spo337
https://images.google.ae/url?q=https://sites.google.com/view/spo337
https://images.google.ad/url?sa=t&url=https://sites.google.com/view/spo337
https://maps.google.co.ke/url?sa=t&url=https://sites.google.com/view/spo337
https://maps.google.co.jp/url?sa=t&url=https://sites.google.com/view/spo337
https://maps.google.co.in/url?sa=t&url=https://sites.google.com/view/spo337
https://maps.google.co.il/url?sa=t&url=https://sites.google.com/view/spo337
https://maps.google.co.id/url?sa=t&url=https://sites.google.com/view/spo337
https://maps.google.co.cr/url?sa=t&url=https://sites.google.com/view/spo337
https://maps.google.co.bw/url?sa=t&url=https://sites.google.com/view/spo337
https://maps.google.cm/url?sa=t&url=https://sites.google.com/view/spo337
https://maps.google.cl/url?sa=t&url=https://sites.google.com/view/spo337
https://maps.google.ci/url?sa=t&url=https://sites.google.com/view/spo337
https://maps.google.ch/url?sa=t&url=https://sites.google.com/view/spo337
https://maps.google.cd/url?sa=t&url=https://sites.google.com/view/spo337
https://maps.google.cat/url?sa=t&url=https://sites.google.com/view/spo337
https://maps.google.ca/url?sa=t&url=https://sites.google.com/view/spo337
https://www.google.co.tz/url?sa=t&url=https://sites.google.com/view/spo337
https://www.google.co.th/url?sa=t&url=https://sites.google.com/view/spo337
https://www.google.co.nz/url?sa=t&url=https://sites.google.com/view/spo337
https://www.google.co.ma/url?sa=t&url=https://sites.google.com/view/spo337
https://www.google.co.ls/url?sa=t&url=https://sites.google.com/view/spo337
https://www.google.co.kr/url?sa=t&url=https://sites.google.com/view/spo337
https://www.google.co.ke/url?sa=t&url=https://sites.google.com/view/spo337
https://www.google.co.jp/url?sa=t&url=https://sites.google.com/view/spo337
https://www.google.co.in/url?sa=t&url=https://sites.google.com/view/spo337
https://www.google.co.il/url?sa=t&url=https://sites.google.com/view/spo337
https://www.google.co.id/url?sa=t&url=https://sites.google.com/view/spo337
https://www.google.co.cr/url?sa=t&url=https://sites.google.com/view/spo337
https://www.google.co.bw/url?sa=t&url=https://sites.google.com/view/spo337
https://www.google.cm/url?sa=t&url=https://sites.google.com/view/spo337
https://www.google.cl/url?sa=t&url=https://sites.google.com/view/spo337
https://www.google.ci/url?sa=t&url=https://sites.google.com/view/spo337
https://www.google.ch/url?sa=t&url=https://sites.google.com/view/spo337
https://www.google.cd/url?sa=t&url=https://sites.google.com/view/spo337
https://www.google.cat/url?sa=t&url=https://sites.google.com/view/spo337
https://www.google.ca/url?sa=t&url=https://sites.google.com/view/spo337
https://betop24.com
http://bit.ly/m/betop24
파라오카지노 https://betop24.com/pharaoh-casino/
쿨카지노 https://betop24.com/cool-casino/
뉴헤븐카지노 https://betop24.com/nhcasino/
솔카지노 https://betop24.com/solcasino/
펀카지노 https://betop24.com/fun-casino/
헤라카지노 https://betop24.com/hera-casino/
안전 카지노사이트 https://betop24.com/
온라인카지노 추천 https://betop24.com/
바카라사이트 추천 https://betop24.com/
https://wp.me/peP4kL-37
https://bit.ly/3rlMEof
https://adamscottybest.wordpress.com/2023/08/16/%ec%98%a8%eb%9d%bc%ec%9d%b8%ec%b9%b4%ec%a7%80%eb%85%b8-%ec%b6%94%ec%b2%9c/
https://wordpress.com/read/feeds/142272460/posts/4852882316
https://adamscottybest.wordpress.com/2023/07/09/%eb%b0%94%ec%b9%b4%eb%9d%bc%ec%82%ac%ec%9d%b4%ed%8a%b8-%ec%b6%94%ec%b2%9c/
https://wordpress.com/read/feeds/142272460/posts/4795680942
https://wordpress.com/read/feeds/142272460
https://adamscottybest.wordpress.com/2023/07/16/%ec%95%88%ec%a0%84-%ec%b9%b4%ec%a7%80%eb%85%b8%ec%82%ac%ec%9d%b4%ed%8a%b8/
https://wordpress.com/read/feeds/142272460/posts/4806574227
https://wp.me/peP4kL-3u
https://rb.gy/elfmp
https://ln.run/1_Agy
http://bit.ly/3mSEOju
https://cutt.ly/E4furTX
https://rb.gy/mixjhy
http://bit.ly/m/betop24
http://bit.ly/3r8oE7T
http://bit.ly/3XzwzHs
http://bit.ly/3NJAVZR
http://bit.ly/3HM4DcO
https://sites.google.com/view/betop24baccarat/%ED%99%88/
https://sites.google.com/view/betop24casino/%ED%99%88/
https://sites.google.com/view/betop24online/%ED%99%88/
https://sites.google.com/view/betop24/%ED%99%88
https://sites.google.com/view/funcasino1/%ED%99%88
https://sites.google.com/view/pharaohcasino1/%ED%99%88
https://sites.google.com/view/solcasino1/%ED%99%88
https://sites.google.com/view/nhcasino/%ED%99%88
https://sites.google.com/view/coolcasino/%ED%99%88
https://sites.google.com/view/heracasino1/%ED%99%88
apps.cancaonova.com/ads/www/delivery/ck.php?ct=1&oaparams=2__bannerid%3D149__zoneid%3D20__cb%3D87d2c6208d__oadest%3Dhttps://betop24.com%2F
apps.cancaonova.com/ads/www/delivery/ck.php?ct=1&oaparams=2__bannerid%3D149__zoneid%3D20__cb%3D87d2c6208d__oadest%3Dhttp%3A%2F%2Fwww.betop24.com/
apps.cancaonova.com/ads/www/delivery/ck.php?ct=1&oaparams=2__bannerid%3D149__zoneid%3D20__cb%3D87d2c6208d__oadest%3Dhttp://www.betop24.com%2F
apps.cancaonova.com/ads/www/delivery/ck.php?ct=1&oaparams=2__bannerid%3D318__zoneid%3D4__cb%3Db3a8c7b256__oadest%3Dhttp://www.betop24.com/
radio.cancaonova.com/iframe-loader/?ra&t=Dating%20Single%3A%20No%20Longer%20a%20Mystery%20-%20R%C3%A1dio&url=http://www.betop24.com%2F
coub.com/away?promoted_coub=3904&to=http://www.betop24.com%2F
www.info-realty.ru/bitrix/rk.php?goto=http://www.betop24.com%2F
www.hentainiches.com/index.php?id=derris&tour=http://www.betop24.com%2F
www.fudbal91.com/tz.php?r=http://www.betop24.com%2F&zone=America%2FIqaluit
www.flyingsamaritans.net/Startup/SetupSite.asp?RestartPage=http://www.betop24.com%2F
www.russianrobotics.ru/bitrix/rk.php?goto=http://www.betop24.com%2F
uniline.co.nz/Document/Url/?url=http://www.betop24.com%2F
unicom.ru/links.php?go=http://www.betop24.com%2F
underwood.ru/away.html?url=http://www.betop24.com%2F
www.odmp.org/link?url=http://www.betop24.com%2F
www.indianjournals.com/redirectToAD.aspx?adAddress=http://www.betop24.com%2F&id=MQA1ADQAMQA0AA%3D%3D
http://www.indianjournals.com/redirectToAD.aspx?id=MQA1ADQAMQA0AA%3D%3D&adAddress=http://www.betop24.com%2F
https://underwood.ru/away.html?url=http://www.betop24.com%2F
https://unicom.ru/links.php?go=http://www.betop24.com%2F
https://uniline.co.nz/Document/Url/?url=http://www.betop24.com%2F
https://www.russianrobotics.ru/bitrix/rk.php?goto=http://www.betop24.com%2F
https://www.flyingsamaritans.net/Startup/SetupSite.asp?RestartPage=http://www.betop24.com%2F
https://www.hentainiches.com/index.php?id=derris&tour=http://www.betop24.com%2F
https://www.info-realty.ru/bitrix/rk.php?goto=http://www.betop24.com%2F
https://fdeam.finanzen-partnerprogramm.de/tracking/?as_id=9257&c_id=595&url=http://www.betop24.com%2F
https://pdcn.co/e/http://www.betop24.com%2F
https://api.webconnex.com/v1/postmaster/track/click/4f8036d14ee545798599c8921fbfcd22/db005310dba511e89fb606f49a4ee876?url=http://www.betop24.com%2F
https://www.lecake.com/stat/goto.php?url=http://www.betop24.com%2F
https://www.im-harz.com/counter/counter.php?url=http://www.betop24.com%2F
https://pzz.to/click?uid=8571&target_url=http://www.betop24.com%2F
https://www.ricacorp.com/Ricapih09/redirect.aspx?link=http://www.betop24.com%2F
https://www.tremblant.ca/Shared/LanguageSwitcher/ChangeCulture?culture=en&url=http://www.betop24.com%2F
https://www.winxuan.com/page/cps/eqifacookieinterface.jsp?from=yiqifa&wid=8&url=http://www.betop24.com%2F
https://moscowdesignmuseum.ru/bitrix/rk.php?goto=http://www.betop24.com%2F
https://igert2011.videohall.com/to_client?target=http://www.betop24.com%2F
http://micimpact.com/bannerhit.php?bn_id=31&url=http://www.betop24.com%2F
https://ovatu.com/e/c?url=http://www.betop24.com%2F
https://primepartners.globalprime.com/afs/wcome.php?c=427|0|1&e=GP204519&url=http://www.betop24.com%2F
https://ltp.org/home/setlocale?locale=en&returnUrl=http://www.betop24.com%2F
https://www.morhipo.com/shared/partnercookie?k=gort&url=http://www.betop24.com%2F
https://cingjing.fun-taiwan.com/AdRedirector.aspx?padid=303&target=http://www.betop24.com%2F
https://rtb-asiamax.tenmax.io/bid/click/1462922913409/e95f2c30-1706-11e6-a9b4-a9f6fe33c6df/3456/5332/?rUrl=http://www.betop24.com%2F
https://needsfs.clientcommunity.com.au/?EXT_URL=http://www.betop24.com%2F&MID=92738
https://campaign.unitwise.com/click?emid=31452&emsid=ee720b9f-a315-47ce-9552-fd5ee4c1c5fa&url=http://www.betop24.com%2F
https://www.stuhleck.com/io/out.asp?url=http://www.betop24.com%2F
https://www.swipeclock.com/sc/cookie.asp?sitealias=79419397&redirect=http://www.betop24.com%2F
https://www.gutscheinaffe.de/wp-content/plugins/AND-AntiBounce/redirector.php?url=http://www.betop24.com%2F
https://home.uceusa.com/Redirect.aspx?r=http://www.betop24.com%2F
https://www.prodesigns.com/redirect?url=http://www.betop24.com%2F
https://www.seankenney.com/include/jump.php?num=http://www.betop24.com%2F
https://runningcheese.com/go?url=http://www.betop24.com%2F
http://chtbl.com/track/118167/http://www.betop24.com%2F
https://ads.glassonline.com/ads.php?id_banner=370&id_campaign=291&link=http://www.betop24.com%2F
https://m.17ll.com/apply/tourl/?url=http://www.betop24.com%2F
https://crewroom.alpa.org/SAFETY/LinkClick.aspx?link=http://www.betop24.com%2F&mid=12872
https://www.sponsorship.com/Marketplace/redir.axd?ciid=514&cachename=advertising&PageGroupId=14&url=http://www.betop24.com%2F
https://rs.businesscommunity.it/snap.php?u=http://www.betop24.com%2F
http://dstats.net/redir.php?url=http://www.betop24.com%2F
https://www.aiac.world/pdf/October-December2015Issue?pdf_url=http://www.betop24.com%2F
https://click.alamode.com/?adcode=CPEMAQM0913_1&url=http://www.betop24.com%2F%20
https://m.caijing.com.cn/member/logout?referer=http://www.betop24.com%2F
https://jamesattorney.agilecrm.com/click?u=http://www.betop24.com%2F
https://wikimapia.org/external_link?url=http://www.betop24.com%2F
https://www.expoon.com/link?url=http://www.betop24.com%2F
https://rsv.nta.co.jp/affiliate/set/af100101.aspx?site_id=66108024&redi_url=http://www.betop24.com%2F
https://adengine.old.rt.ru/go.jsp?to=http://www.betop24.com%2F
https://thediplomat.com/ads/books/ad.php?i=4&r=http://www.betop24.com%2F
https://mitsui-shopping-park.com/lalaport/iwata/redirect.html?url=http://www.betop24.com%2F
https://m.addthis.com/live/redirect/?url=http://www.betop24.com%2F
https://za.zalo.me/v3/verifyv2/pc?token=OcNsmjfpL0XY2F3BtHzNRs4A-hhQ5q5sPXtbk3O&continue=http://www.betop24.com%2F%20
https://scanmail.trustwave.com/?c=8510&d=4qa02KqxZJadHuhFUvy7ZCUfI_2L10yeH0EeBz7FGQ&u=http://www.betop24.com%2F
https://animal.doctorsfile.jp/redirect/?ct=doctor&id=178583&url=http://www.betop24.com%2F
https://hr.pecom.ru/bitrix/rk.php?goto=http://www.betop24.com%2F
https://jump2.bdimg.com/mo/q/checkurl?url=http://www.betop24.com%2F
https://severeweather.wmo.int/cgi-bin/goto?where=http://www.betop24.com%2F
https://beam.jpn.org/rank.cgi?mode=link&url=http://www.betop24.com%2F
https://jump2.bdimg.com/mo/q/checkurl?url=http://www.betop24.com%2F
https://gar86.tmweb.ru/bitrix/rk.php?goto=http://www.betop24.com%2F
https://blogranking.fc2.com/out.php?id=414788&url=www.betop24.com%2F
https://wtk.db.com/777554543598768/optout?redirect=http://www.betop24.com%2F
https://community.rsa.com/t5/custom/page/page-id/ExternalRedirect?url=http://www.betop24.com
https://click.alamode.com/?adcode=CPEMAQM0913_1&url=http://www.betop24.com%2F
http://teenstgp.us/cgi-bin/out.cgi?u=http://www.betop24.com%2F
http://congovibes.com/index.php?thememode=full;redirect=http://www.betop24.com%2F
http://www.humaniplex.com/jscs.html?hj=y&ru=http://www.betop24.com%2F
http://www.qlt-online.de/cgi-bin/click/clicknlog.pl?link=http://www.betop24.com%2F
http://ww.sdam-snimu.ru/redirect.php?url=http://www.betop24.com%2F
http://www.uktrademarkregistration.co.uk/JumpTo.aspx?url=http://www.betop24.com%2F
http://landbidz.com/redirect.asp?url=http://www.betop24.com%2F
http://www.macro.ua/out.php?link=http://www.betop24.com%2F
http://www.aurki.com/jarioa/redirect?id_feed=510&url=http://www.betop24.com%2F
http://m.ee17.com/go.php?url=http://www.betop24.com%2F
http://shop-navi.com/link.php?mode=link&id=192&url=http://www.betop24.com%2F
http://www.cnainterpreta.it/redirect.asp?url=http://www.betop24.com%2F
http://www.site-navi.net/sponavi/rank.cgi?mode=link&id=890&url=http://www.betop24.com%2F
https://www.mattias.nu/cgi-bin/redirect.cgi?http://www.betop24.com%2F
http://distributors.hrsprings.com/?URL=spo337.com
http://dorf-v8.de/url?q=http://www.betop24.com%2F
http://doverwx.com/template/pages/station/redirect.php?url=http://www.betop24.com%2F
http://dr-guitar.de/quit.php?url=http://www.betop24.com%2F
http://drdrum.biz/quit.php?url=http://www.betop24.com%2F
http://ds-media.info/url?q=http://www.betop24.com%2F
http://excitingperformances.com/?URL=spo337.com
http://familie-huettler.de/link.php?link=spo337.com
http://forum.vizslancs.hu/lnks.php?uid=net&url=http://www.betop24.com%2F
http://forum.wonaruto.com/redirection.php?redirection=http://www.betop24.com%2F
http://fosteringsuccessmichigan.com/?URL=spo337.com
http://fotos24.org/url?q=http://www.betop24.com%2F
http://fouillez-tout.com/cgi-bin/redirurl.cgi?spo337.com
http://frag-den-doc.de/index.php?s=verlassen&url=http://www.betop24.com%2F
http://freethailand.com/goto.php?url=http://www.betop24.com%2F
http://freshcannedfrozen.ca/?URL=spo337.com
http://funkhouse.de/url?q=http://www.betop24.com%2F
http://ga.naaar.nl/link/?url=http://www.betop24.com%2F
http://gb.poetzelsberger.org/show.php?c453c4=spo337.com
http://gdnswebapppro.cloudapp.net/gooutside?url=http://www.betop24.com%2F
http://getmethecd.com/?URL=spo337.com
http://go.netatlantic.com/subscribe/subscribe.tml?list=michaelmoore&confirm=none&url=http://www.betop24.com%2F&email=hmabykq%40outlook.com
http://goldankauf-oberberg.de/out.php?link=http://www.betop24.com%2F
https://www.fairsandfestivals.net/?URL=http://www.betop24.com%2F
http://crewe.de/url?q=http://www.betop24.com%2F
http://cwa4100.org/uebimiau/redir.php?http://www.betop24.com%2F
http://d-quintet.com/i/index.cgi?id=1&mode=redirect&no=494&ref_eid=33&url=http://www.betop24.com%2F
http://data.allie.dbcls.jp/fct/rdfdesc/usage.vsp?g=http://www.betop24.com%2F
http://data.linkedevents.org/describe/?url=http://www.betop24.com%2F
http://datos.infolobby.cl/describe/?url=http://www.betop24.com%2F
http://congressrb.info/bitrix/rk.php?goto=http://www.betop24.com%2F
http://conny-grote.de/url?q=http://www.betop24.com%2F
http://crazyfrag91.free.fr/?URL=www.betop24.com%2F
http://dayviews.com/externalLinkRedirect.php?url=http://www.betop24.com%2F
http://db.cbservices.org/cbs.nsf/forward?openform&http://www.betop24.com%2F
http://chatx2.whocares.jp/redir.jsp?u=http://www.betop24.com%2F
http://chuanroi.com/Ajax/dl.aspx?u=http://www.betop24.com%2F
http://club.dcrjs.com/link.php?url=http://www.betop24.com%2F
http://www.paladiny.ru/go.php?url=http://www.betop24.com%2F
http://tido.al/vazhdo.php?url=http://www.betop24.com%2F
http://smile.wjp.am/link-free/link3.cgi?mode=cnt&no=8&hpurl=http://www.betop24.com%2F
https://www.usjournal.com/go.php?campusID=190&url=http://www.betop24.com%2F
https://analytics.bluekai.com/site/16231?phint=event=click&phint=campaign=BRAND-TAB&phint=platform=search&done=http://www.betop24.com%2F
https://temptationsaga.com/buy.php?url=http://www.betop24.com%2F
https://www.cheerunion.org/tracker/index.html?t=ad&pool_id=2&ad_id=5&url=http://www.betop24.com%2F
https://www.moneydj.com/ads/adredir.aspx?bannerid=39863&url=http://www.betop24.com%2F
https://uk.kindofbook.com/redirect.php/?red=http://www.betop24.com%2F
http://m.17ll.com/apply/tourl/?url=http://www.betop24.com%2F
https://track.effiliation.com/servlet/effi.redir?id_compteur=13215059&url=http://www.betop24.com%2F
https://www.shtrih-m.ru/bitrix/redirect.php?goto=http://www.betop24.com%2F
http://simvol-veri.ru/xp/?goto=http://www.betop24.com%2F
http://www.skladcom.ru/(S(qdiwhk55jkcyok45u4ti0a55))/banners.aspx?url=http://www.betop24.com%2F
https://stroim100.ru/redirect?url=http://www.betop24.com%2F
https://kakaku-navi.net/items/detail.php?url=http://www.betop24.com%2F
https://meguro.keizai.biz/banner.php?type=image_banner&position=right&id=13&uri=http://www.betop24.com%2F
http://ads.cars.cz/adclick.php?bannerid=333&zoneid=237&source=&dest=http://www.betop24.com%2F
https://spb-medcom.ru/redirect.php?http://www.betop24.com%2F
https://www.info-realty.ru/bitrix/rk.php?goto=http://www.betop24.com%2F
http://www.sermemole.com/public/serbook/redirect.php?url=http://www.betop24.com%2F
https://www.hradycz.cz/redir.php?b=445&t=http://www.betop24.com%2F
http://prasat.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://meri.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://prabu.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://easehipranaam.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://nidatyi.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://krodyit.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://naine.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://breajwasi.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://beithe.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://tumari.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://hariomm.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://lejano.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://jotumko.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://auyttrv.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://bartos.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://cllfather.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://littlejohnny.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://semesmemos.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://faultypirations.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://mmurugesamnfo.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://esujkamien.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://ujkaeltnx.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://ausnangck2809.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://supermu.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://buyfcyclingteam.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://allthingnbeyondblog.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://usafunworldt.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://financialallorner.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://mjghouthernmatron.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://katihfmaxtron.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://ikkemandar.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://googlejfgdlenewstoday.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://bhrecadominicana.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://anupam-bestprice89.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://bhrepublica.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://allinspirations.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://bestandroid.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://discoverable.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://puriagatratt.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://azizlemon.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://chandancomputers.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://bingshopping.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://ptritam.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://tech-universes.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://littleboy.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://correctinspiration.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://weallfreieds.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://shanmegurad.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://manualmfuctional.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://verybeayurifull.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://dilip.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://avinasg.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://virat.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://hsadyttk.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://ashu-quality.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://tinko.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://konkaruna.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://udavhav.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://matura.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://kripa.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://charanraj.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://pream.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://sang.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://pasas.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://kanchan.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://ptrfsiitan.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://boardgame-breking.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://flowwergulab.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://bingcreater-uk.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://skinnyskin.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://amil.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://mohan.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://patana.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://bopal.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://gwl.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://delhi.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://gopi.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://boogiewoogie.com/?URL=spo337.com
http://bsumzug.de/url?q=http://www.betop24.com%2F
http://business.eatonton.com/list?globaltemplate=http://www.betop24.com%2F
http://cdiabetes.com/redirects/offer.php?URL=http://www.betop24.com%2F
https://padletcdn.com/cgi/fetch?disposition=attachment&url=http://www.betop24.com%2F
http://cdn.iframe.ly/api/iframe?url=http://www.betop24.com%2F
http://centuryofaction.org/?URL=spo337.com
http://alexanderroth.de/url?q=http://www.betop24.com%2F
http://alt.baunetzwissen.de/rd/nl-track/?obj=bnw&cg1=redirect&cg2=wissen-newsletter:beton&cg3=partner&datum=2017-01&titel=partnerklick-beton&firma=informationszentrumbeton&link=http://www.betop24.com%2F
http://andreasgraef.de/url?q=http://www.betop24.com%2F
http://app.espace.cool/ClientApi/SubscribeToCalendar/1039?url=http://www.betop24.com%2F
http://archive.paulrucker.com/?URL=http://www.betop24.com%2F
http://archives.midweek.com/?URL=spo337.com
http://bernhardbabel.com/url?q=http://www.betop24.com%2F
http://bigbarganz.com/g/?http://www.betop24.com%2F
http://bioinformatics.cenicafe.org/?URL=spo337.com
http://Somewh.a.T.dfqw@www.newsdiffs.org/article-history/www.findabeautyschool.com/map.aspx?url=http://www.betop24.com%2F
http://a-jansen.de/url?q=http://www.betop24.com%2F
http://actontv.org/?URL=spo337.com
http://ajman.dubaicityguide.com/main/advertise.asp?oldurl=http://www.betop24.com%2F
http://albertaaerialapplicators.com/?URL=spo337.com
http://blackberryvietnam.net/proxy.php?link=http://www.betop24.com%2F
http://baseballpodcasts.net/Feed2JS/feed2js.php?src=http://www.betop24.com%2F
http://bbs.mottoki.com/index?bbs=hpsenden&act=link&page=94&linkk=http://www.betop24.com%2F
http://armdrag.com/mb/get.php?url=http://www.betop24.com%2F
http://asai-kota.com/acc/acc.cgi?REDIRECT=http://www.betop24.com%2F
http://ass-media.de/wbb2/redir.php?url=http://www.betop24.com%2F
http://assadaaka.nl/?URL=spo337.com
http://atchs.jp/jump?u=http://www.betop24.com%2F
http://away3d.com/?URL=spo337.com
http://community.nxp.com/t5/custom/page/page-id/ExternalRedirect?url=http://www.betop24.com%2F
http://ceskapozice.lidovky.cz/redir.aspx?url=http://www.betop24.com%2F
http://mitsui-shopping-park.com/lalaport/ebina/redirect.html?url=http://www.betop24.com%2F
http://kupiauto.zr.ru/bitrix/rk.php?goto=http://www.betop24.com%2F
http://ipx.bcove.me/?url=http://www.betop24.com%2F
http://client.paltalk.com/client/webapp/client/External.wmt?url=http://www.betop24.com%2F
http://www.interempresas.net/estadisticas/r.asp?idsector=129&e=221083&c=195&d=http://www.betop24.com%2F
http://fishbiz.seagrant.uaf.edu/click-thru.html?url=http://www.betop24.com%2F
http://foro.infojardin.com/proxy.php?link=http://www.betop24.com%2F
http://www.t.me/iv?url=http://www.betop24.com%2F
http://es.catholic.net/ligas/ligasframe.phtml?liga=http://www.betop24.com%2F
http://forum.solidworks.com/external-link.jspa?url=http://www.betop24.com%2F
http://www.guru-pon.jp/search/rank.cgi?mode=link&id=107&url=http://www.betop24.com%2F
http://www.nickl-architects.com/url?q=http://www.betop24.com%2F
https://www.ocbin.com/out.php?url=http://www.betop24.com%2F
http://www.lobenhausen.de/url?q=http://www.betop24.com%2F
http://redirect.me/?http://www.betop24.com%2F
http://kank.o.oo7.jp/cgi-bin/ys4/rank.cgi?mode=link&id=569&url=http://www.betop24.com%2F
http://ruslog.com/forum/noreg.php?http://www.betop24.com%2F
http://log.vt.open8.com/v1/click?campaign_id=644&flight_id=5105&ad_id=3185&flight_ad_id=8884&size=BIGINFEED&media=BI_MODELPRESS&area=BI_ALL&advertiser_id=149&redirect_url=http://www.betop24.com%2F
http://www.inkwell.ru/redirect/?url=http://www.betop24.com%2F
http://www.delnoy.com/url?q=http://www.betop24.com%2F
http://click-navi.jp/cgi/service-search/rank.cgi?mode=link&id=121&url=http://www.betop24.com%2F
https://n1653.funny.ge/redirect.php?url=http://www.betop24.com%2F
https://www.livecmc.com/?lang=fr&id=Ld9efT&url=http://www.betop24.com%2F
http://deai-ranking.org/search/rank.cgi?mode=link&id=28&url=http://www.betop24.com%2F
https://izispicy.com/go.php?url=http://www.betop24.com%2F
http://demo.vieclamcantho.vn/baohiemthatnghiep/Redirect.aspx?sms=90bb20bb20tbb20thc3%B4ng&link=http://www.betop24.com%2F
http://www.city-fs.de/url?q=http://www.betop24.com%2F
http://p.profmagic.com/urllink.php?url=http://www.betop24.com%2F
http://maxnetworks.org/searchlink/rank.cgi?mode=link&id=321&url=http://www.betop24.com%2F
http://go.e-frontier.co.jp/rd2.php?uri=http://www.betop24.com%2F
http://adchem.net/Click.aspx?url=http://www.betop24.com%2F
http://www.reddotmedia.de/url?q=http://www.betop24.com%2F
https://10ways.com/fbredir.php?orig=http://www.betop24.com%2F
https://emailtrackerapi.leadforensics.com/api/URLOpen?EmailSentRecordID=17006&URL=http://www.betop24.com%2F
http://search.haga-f.net/rank.cgi?mode=link&url=http://www.betop24.com%2F
http://7ba.ru/out.php?url=http://www.betop24.com%2F
http://www.51queqiao.net/link.php?url=http://www.betop24.com%2F
https://rsyosetsu.bookmarks.jp/ys4/rank.cgi?mode=link&id=3519&url=http://www.betop24.com%2F
http://forum.ahigh.ru/away.htm?link=http://www.betop24.com%2F
http://www.wildner-medien.de/url?q=http://www.betop24.com%2F
http://www.tifosy.de/url?q=http://www.betop24.com%2F
http://ads.icorp.ro/others/STS/?t=CeNortjKxUjK0NDFVsgZcMBADAm4-&g=http://www.betop24.com%2F
http://t.me/iv?url=http://www.betop24.com%2F
http://www.air-dive.com/au/mt4i.cgi?mode=redirect&ref_eid=697&url=http://www.betop24.com%2F
http://okozukai.j-web.jp/j-web/okozukai/ys4/rank.cgi?mode=link&id=6817&url=http://www.betop24.com%2F
https://sfmission.com/rss/feed2js.php?src=http://www.betop24.com%2F
https://www.watersportstaff.co.uk/extern.aspx?src=http://www.betop24.com%2F&cu=60096&page=1&t=1&s=42"
https://partner.jpc.de/go.cgi?pid=125&wmid=cc&cpid=1&subid=release&target=http://www.betop24.com%2F
http://siamcafe.net/board/go/go.php?http://www.betop24.com%2F
http://www.whitening-navi.info/cgi/search-smartphone/rank.cgi?mode=link&id=1431&url=http://www.betop24.com%2F
http://www.youtube.com/redirect?q=http://www.betop24.com%2F
url?sa=j&source=web&rct=j&url=http://www.betop24.com%2F
https://home.guanzhuang.org/link.php?url=http://www.betop24.com%2F
http://dvd24online.de/url?q=http://www.betop24.com%2F
http://twindish-electronics.de/url?q=http://www.betop24.com%2F
http://www.beigebraunapartment.de/url?q=http://www.betop24.com%2F
http://www.mix-choice.com/yomi/rank.cgi?mode=link&id=391&url=http://www.betop24.com%2F
https://bbs.hgyouxi.com/kf.php?u=http://www.betop24.com%2F
http://www.1soft-tennis.com/search/rank.cgi?mode=link&id=17&url=http://www.betop24.com%2F
http://big-data-fr.com/linkedin.php?lien=http://www.betop24.com%2F
http://reg.kost.ru/cgi-bin/go?http://www.betop24.com%2F
http://www.kirstenulrich.de/url?q=http://www.betop24.com%2F
https://www.kenzai-navi.com/location/location_topbanner.php?bs=238&m=899&href=http://www.betop24.com%2F
https://www.anybeats.jp/jump/?http://www.betop24.com%2F
http://url-collector.appspot.com/positiveVotes?topic=Hate%20speech&url=http://www.betop24.com%2F
https://top.hange.jp/linkdispatch/dispatch?targetUrl=http://www.betop24.com%2F
http://www.link.gokinjyo-eikaiwa.com/rank.cgi?mode=link&id=5&url=http://www.betop24.com%2F
http://members.asoa.org/sso/logout.aspx?returnurl=http://www.betop24.com%2F
http://login.mediafort.ru/autologin/mail/?code=14844×02ef859015x290299&url=http://www.betop24.com%2F
https://www.sports-central.org/cgi-bin/axs/ax.pl?http://www.betop24.com%2F
http://dot.boss.sk/o/rs.php?ssid=1&szid=4&dsid=12&dzid=32&url=www.betop24.com%2F
http://sistema.sendmailing.com.ar/includes/php/emailer.track.php?vinculo=http://www.betop24.com%2F
http://www.americanstylefridgefreezer.co.uk/go.php?url=http://www.betop24.com%2F
https://www.newzealandgirls.co.nz/auckland/escorts/clicks.php?click_id=NavBar_AF_AdultDirectory&target=http://www.betop24.com%2F
https://wdesk.ru/go?http://www.betop24.com%2F
http://1000love.net/lovelove/link.php?url=http://www.betop24.com%2F
https://ulfishing.ru/forum/go.php?http://www.betop24.com%2F
https://underwood.ru/away.html?url=http://www.betop24.com%2F
https://unicom.ru/links.php?go=http://www.betop24.com%2F
http://www.unifin.ru/bitrix/redirect.php?goto=http://www.betop24.com%2F
https://uogorod.ru/feed/520?redirect=http://www.betop24.com%2F
http://uvbnb.ru/go?http://www.betop24.com%2F
https://skibaza.ru/bitrix/rk.php?goto=http://www.betop24.com%2F
https://smartservices.ru/bitrix/rk.php?goto=http://www.betop24.com%2F
http://www.topkam.ru/gtu/?url=http://www.betop24.com%2F
https://torggrad.ru/bitrix/rk.php?goto=http://www.betop24.com%2F
http://torgi-rybinsk.ru/bitrix/rk.php?goto=http://www.betop24.com%2F
http://tpprt.ru/bitrix/rk.php?goto=http://www.betop24.com%2F
https://www.snek.ai/redirect?url=http://www.betop24.com%2F
https://www.auburnapartmentguide.com/MobileDefault.aspx?reff=http://www.betop24.com%2F
http://gals.graphis.ne.jp/mkr/out.cgi?id=01019&go=http://www.betop24.com%2F
http://www.capitalbikepark.se/bok/go.php?url=http://www.betop24.com%2F
http://an.to/?go=http://www.betop24.com%2F
http://cooltgp.org/tgp/click.php?id=370646&u=http://www.betop24.com%2F
https://joomlinks.org/?url=http://www.betop24.com%2F
https://managementportal.de/modules/mod_jw_srfr/redir.php?url=http://www.betop24.com%2F
http://www.green-cross.pro/bitrix/redirect.php?goto=http://www.betop24.com%2F
http://m.adlf.jp/jump.php?l=http://www.betop24.com%2F
https://www.pilot.bank/out.php?url=http://www.betop24.com%2F
https://www.moonbbs.com/dm/dmlink.php?dmurl=http://www.betop24.com%2F
http://www.mac52ipod.cn/urlredirect.php?go=http://www.betop24.com%2F
http://salinc.ru/redirect.php?url=http://www.betop24.com%2F
http://minlove.biz/out.html?id=nhmode&go=http://www.betop24.com%2F
http://www.diwaxx.ru/win/redir.php?redir=http://www.betop24.com%2F
http://request-response.com/blog/ct.ashx?url=http://www.betop24.com%2F
https://turbazar.ru/url/index?url=http://www.betop24.com%2F
https://www.vicsport.com.au/analytics/outbound?url=http://www.betop24.com%2F
http://www.cyprus-net.com/banner_click.php?banid=4&link=http://www.betop24.com%2F
http://a.gongkong.com/db/adredir.asp?id=16757&url=http://www.betop24.com%2F
http://gfaq.ru/go?http://www.betop24.com%2F
http://edcommunity.ru/bitrix/rk.php?goto=http://www.betop24.com%2F
http://page.yicha.cn/tp/j?url=http://www.betop24.com%2F
http://www.kollabora.com/external?url=http://www.betop24.com%2F
http://www.internettrafficreport.com/cgi-bin/cgirdir.exe?http://www.betop24.com%2F
http://www.interfacelift.com/goto.php?url=http://www.betop24.com%2F
https://passport.sfacg.com/LoginOut.aspx?Returnurl=http://www.betop24.com%2F
https://offers.sidex.ru/stat_ym_new.php?redir=http://www.betop24.com%2F&hash=1577762
https://globalmedia51.ru/bitrix/redirect.php?goto=http://www.betop24.com%2F
https://ramset.com.au/document/url/?url=http://www.betop24.com%2F
http://gbi-12.ru/links.php?go=http://www.betop24.com%2F
http://www.global-flat.com/mobile/mobile_switch.php?dir=tofull&url=http://www.betop24.com%2F
https://golden-resort.ru/out.php?out=http://www.betop24.com%2F
http://avalon.gondor.ru/away.php?link=http://www.betop24.com%2F
http://www.laosubenben.com/home/link.php?url=http://www.betop24.com%2F
http://www.lifeshow.com.tw/show.php?ty5_id=1599&url=http://www.betop24.com%2F
http://www.humanbrainmapping.org/i4a/etrack/track.cfm?rType=2&campaignID=3572&contactID=4524&origURL=http://www.betop24.com%2F
https://gcup.ru/go?http://www.betop24.com%2F
http://www.gearguide.ru/phpbb/go.php?http://www.betop24.com%2F
https://es.catholic.net/ligas/ligasframe.phtml?liga=http://www.betop24.com%2F
https://good-surf.ru/r.php?g=http://www.betop24.com%2F
http://mbrf.ae/knowledgeaward/language/ar/?redirect_url=http://www.betop24.com%2F
https://gektor-nsk.ru/bitrix/redirect.php?goto=http://www.betop24.com%2F
https://lekoufa.ru/banner/go?banner_id=4&link=http://www.betop24.com%2F
https://forsto.ru/bitrix/redirect.php?goto=http://www.betop24.com%2F
https://www.cossa.ru/bitrix/redirect.php?event1=click&event2=&event3=&goto=http://www.betop24.com%2F
http://www.gigatran.ru/go?url=http://www.betop24.com%2F
http://www.gigaalert.com/view.php?h=&s=http://www.betop24.com%2F
http://www.jkes.tyc.edu.tw/dyna/webs/gotourl.php?id=357&url=http://www.betop24.com%2F
http://www.laopinpai.com/gourl.asp?url=/gourl.asp?url=http://www.betop24.com%2F
http://bw.irr.by/knock.php?bid=252583&link=http://www.betop24.com%2F
http://www.beeicons.com/redirect.php?site=http://www.betop24.com%2F
http://www.diwaxx.ru/hak/redir.php?redir=http://www.betop24.com%2F
http://www.actuaries.ru/bitrix/rk.php?goto=http://www.betop24.com%2F
http://www.isadatalab.com/redirect?clientId=ee5a64e1-3743-9b4c-d923-6e6d092ae409&appId=69&value=[EMVFIELD]EMAIL[EMV/FIELD]&cat=Techniques+culturales&url=www.betop24.com%2F
https://www.jukujo.gs/bin/out.cgi?id=kimiomof&url=http://www.betop24.com%2F
http://hansolav.net/blog/ct.ashx?id=0af6301b-e71f-44be-838f-905709eee792&url=http://www.betop24.com%2F
http://bitrix.adlr.ru/bitrix/rk.php?goto=http://www.betop24.com%2F
https://m.snek.ai/redirect?url=http://www.betop24.com%2F
http://www.officeservice.com.br/trackMKT/trackerFolha.aspx?DESCRIPTION=HotSite_EcrmContabil&FROM=Workshop&DESTINATION=www.betop24.com%2F
https://www.arbsport.ru/gotourl.php?url=http://www.betop24.com%2F
http://earnupdates.com/goto.php?url=http://www.betop24.com%2F
https://automall.md/ru?url=www.betop24.com%2F
http://www.strana.co.il/finance/redir.aspx?site=www.betop24.com%2F
https://www.tourplanisrael.com/redir/?url=http://www.betop24.com%2F
http://www.mutualgravity.com/archangel/webcontact/d_signinsimple.php?action=signup&CID=240&EID=&S=default.css&return=http://www.betop24.com%2F
http://buildingreputation.com/lib/exe/fetch.php?media=http://www.betop24.com%2F
http://gals.graphis.ne.jp/mkr/out.cgi?id=04489&go=http://www.betop24.com%2F
http://mardigrasparadeschedule.com/phpads/adclick.php?bannerid=18&zoneid=2&source=&dest=http://www.betop24.com%2F
http://dir.tetsumania.net/search/rank.cgi?mode=link&id=3267&url=http://www.betop24.com%2F
http://www.project24.info/mmview.php?dest=www.betop24.com%2F
http://suek.com/bitrix/rk.php?goto=http://www.betop24.com%2F
http://tfads.testfunda.com/TFServeAds.aspx?strTFAdVars=4a086196-2c64-4dd1-bff7-aa0c7823a393,TFvar,00319d4f-d81c-4818-81b1-a8413dc614e6,TFvar,GYDH-Y363-YCFJ-DFGH-5R6H,TFvar,http://www.betop24.com%2F
http://lilholes.com/out.php?http://www.betop24.com%2F
http://www.request-response.com/blog/ct.ashx?id=d827b163-39dd-48f3-b767-002147c94e05&url=http://www.betop24.com%2F
http://www.cooltgp.org/tgp/click.php?id=370646&u=http://www.betop24.com%2F
https://www.bandb.ru/redirect.php?URL=http://www.betop24.com%2F
http://sdchamber.biz/admin/mod_newsletter/redirect.aspx?message_id=785&redirect=http://www.betop24.com%2F
http://www.kyoto-osaka.com/search/rank.cgi?mode=link&url=http://www.betop24.com%2F
http://metalist.co.il/redirect.asp?url=http://www.betop24.com%2F
https://www.voxlocalis.net/enlazar/?url=http://www.betop24.com%2F
http://no-smok.net/nsmk/InterWiki?action=goto&oe=EUC-KR&url=www.betop24.com%2F
http://www.stalker-modi.ru/go?http://www.betop24.com%2F
http://www.p1-uranai.com/rank.cgi?mode=link&id=538&url=http://www.betop24.com%2F
http://www.kitchencabinetsdirectory.com/redirect.asp?url=http://www.betop24.com%2F
http://cityprague.ru/go.php?go=http://www.betop24.com%2F
https://www.hachimantaishi.com/click3/click3.cgi?cnt=c5&url=http://www.betop24.com%2F
http://www.careerbuilder.com.cn/cn/tallyredirector.aspx?task=Ushi&objName=SnsShare&redirectUrl=www.betop24.com%2F&methodName=SetSnsShareLink
http://banners.knollenstein.com/adnetwork/servlet/advertBeans.TrackingServlet?seid=27709655&t=100&redirectTo=spo337.com&cid=DECONL&vid=986809&externalsource=jobbsquare_ppc
https://meyeucon.org/ext-click.php?url=www.betop24.com%2F
http://www.cnmhe.fr/spip.php?action=cookie&url=www.betop24.com%2F
https://perezvoni.com/blog/away?url=http://www.betop24.com%2F
http://www.littlearmenia.com/redirect.asp?url=http://www.betop24.com%2F
https://www.ciymca.org/af/register-redirect/71104?url=www.betop24.com%2F
https://whizpr.nl/tracker.php?u=http://www.betop24.com%2F
http://games.cheapdealuk.co.uk/go.php?url=http://www.betop24.com%2F
https://www.netwalkerstore.com/redirect.asp?accid=18244&adcampaignid=2991&adcampaigntype=2&affduration=30&url=http://www.betop24.com%2F
http://tsugarubrand.jp/blog/?wptouch_switch=mobile&redirect=www.betop24.com%2F
http://blackhistorydaily.com/black_history_links/link.asp?link_id=5&URL=http://www.betop24.com%2F
http://i-marine.eu/pages/goto.aspx?link=http://www.betop24.com%2F
http://www.teenlove.biz/cgibin/atc/out.cgi?id=24&u=http://www.betop24.com%2F
http://www.altoona.com/newsletter/click.php?volume=52&url=http://www.betop24.com%2F
http://www.katjushik.ru/link.php?to=http://www.betop24.com%2F
http://gondor.ru/go.php?url=http://www.betop24.com%2F
http://proekt-gaz.ru/go?http://www.betop24.com%2F
https://www.tricitiesapartmentguide.com/MobileDefault.aspx?reff=http://www.betop24.com%2F
http://www.vietnamsingle.com/vn/banners/redirect.asp?url=http://www.betop24.com%2F
http://archive.cym.org/conference/gotoads.asp?url=http://www.betop24.com%2F
http://cdp.thegoldwater.com/click.php?id=101&url=http://www.betop24.com%2F
http://www.appenninobianco.it/ads/adclick.php?bannerid=159&zoneid=8&source=&dest=http://www.betop24.com%2F
https://www.eduplus.hk/special/emailalert/goURL.jsp?clickURL=http://www.betop24.com%2F
http://www.japan.road.jp/navi/navi.cgi?jump=226&url=www.betop24.com%2F
http://earthsciencescanada.com/modules/babel/redirect.php?newlang=en_us&newurl=http://www.betop24.com%2F
http://psykodynamiskt.nu/?wptouch_switch=desktop&redirect=www.betop24.com%2F
http://www.pcbheaven.com/forum/index.php?thememode=full;redirect=http://www.betop24.com%2F
https://www.oltv.cz/redirect.php?url=http://www.betop24.com%2F
http://www.bigblackmamas.com/cgi-bin/sites/out.cgi?url=www.betop24.com%2F
https://www.yeaah.com/disco/DiscoGo.asp?ID=3435&Site=www.betop24.com%2F
https://primorye.ru/go.php?id=19&url=http://www.betop24.com%2F
https://www.123gomme.it/it/ViewSwitcher/SwitchView?mobile=True&returnUrl=www.betop24.com%2F
http://youmydaddy.com/cgi-bin/at3/out.cgi?id=88&trade=www.betop24.com%2F
https://naviking.localking.com.tw/about/redirect.aspx?mid=7&url=http://www.betop24.com%2F
http://sms-muzeji.si/Home/ChangeCulture?lang=sl&returnUrl=www.betop24.com%2F
http://r.emeraldexpoinfo.com/s.ashx?ms=EXI2:125462_115115&e=krubin723@aol.com&eId=440186886&c=h&url=www.betop24.com%2F
https://apps.firstrain.com/service/openurl.jsp?action=titleclick&src=rss&u=http://www.betop24.com%2F
https://delphic.games/bitrix/redirect.php?goto=http://www.betop24.com%2F
http://www.castellodivezio.it/lingua.php?lingua=EN&url=www.betop24.com%2F
http://moscowdesignmuseum.ru/bitrix/rk.php?goto=http://www.betop24.com%2F
http://www.fairpoint.net/~jensen1242/gbook/go.php?url=http://www.betop24.com%2F
https://lirasport.com.ar/bloques/bannerclick.php?id=7&url=http://www.betop24.com%2F
http://www.knabstrupper.se/guestbook/go.php?url=http://www.betop24.com%2F
https://www.pba.ph/redirect?url=www.betop24.com%2F&id=3&type=tab
https://bondage-guru.net/bitrix/rk.php?goto=http://www.betop24.com%2F
http://www.westbloomfieldlibrary.org/includes/statistics.php?StatType=Link&StatID=Facebook&weblink=www.betop24.com%2F
https://tenisnews.com.br/clickTracker/clickTracker.php?u=http://www.betop24.com%2F
http://www.stevestechspot.com/ct.ashx?id=9fdcf4a4-6e09-4807-bc31-ac1adf836f6c&url=http://www.betop24.com%2F
http://rcoi71.ru/bitrix/redirect.php?goto=http://www.betop24.com%2F
http://www.laselection.net/redir.php3?cat=int&url=spo337.com
http://www.masai-mara.com/cgi-bin/link2.pl?grp=mm&link=www.betop24.com%2F
http://evenemangskalender.se/redirect/?id=15723&lank=http://www.betop24.com%2F
http://anifre.com/out.html?go=www.betop24.com%2F
http://www.restavracije-gostilne.si/banner.php?id=44&url=http://www.betop24.com%2F
http://hobbyplastic.co.uk/trigger.php?r_link=www.betop24.com%2F
https://www.actualitesdroitbelge.be/click_newsletter.php?url=http://www.betop24.com%2F
http://ads.pukpik.com/myads/click.php?banner_id=316&banner_url=http://www.betop24.com%2F
https://www.adminer.org/redirect/?sa=t&url=https%3A%2F%2Fspo337.com%2F
http://www.bigblackbootywatchers.com/cgi-bin/sites/out.cgi?url=www.betop24.com%2F
http://barykin.com/go.php?spo337.com
https://seocodereview.com/redirect.php?url=www.betop24.com%2F
http://miningusa.com/adredir.asp?url=http://www.betop24.com%2F
http://www.arch.iped.pl/artykuly.php?id=1&cookie=1&url=http://www.betop24.com%2F
http://hirlevelcenter.eu/click.php?hirlevel_id=13145441085508&url=http://www.betop24.com%2F
https://birds.cz/avif/redirect.php?from=avif.obs.php&url=http://www.betop24.com%2F
http://old.kob.su/url.php?url=www.betop24.com%2F
http://www.is.kyusan-u.ac.jp/htmllint/htmllint.cgi?ViewSource=on;URL=http://www.betop24.com%2F
https://forum.everleap.com/proxy.php?link=http://www.betop24.com%2F
http://www.mosig-online.de/url?q=http://www.betop24.com%2F
http://www.hccincorporated.com/?URL=http://www.betop24.com%2F
http://fatnews.com/?URL=http://www.betop24.com%2F
http://www.dominasalento.it/?URL=http://www.betop24.com%2F
https://csirealty.com/?URL=http://www.betop24.com%2F
http://asadi.de/url?q=http://www.betop24.com%2F
http://treblin.de/url?q=http://www.betop24.com%2F
https://kentbroom.com/?URL=http://www.betop24.com%2F
http://2cool2.be/url?q=http://www.betop24.com%2F
http://4coma.net/cgi/mt4/mt4i.cgi?cat=12&mode=redirect&ref_eid=3231&url=http://www.betop24.com%2F
http://4travel.jp/dynamic/redirect.php?mode=dm_tour&url=http://www.betop24.com%2F
http://imaginingourselves.globalfundforwomen.org/pb/External.aspx?url=http://www.betop24.com%2F
https://s-p.me/template/pages/station/redirect.php?url=http://www.betop24.com%2F
http://ww.thesteelbrothers.com/buy.php?store=iBooks&url=http://www.betop24.com%2F
http://thdt.vn/convert/convert.php?link=http://www.betop24.com%2F
http://www.noimai.com/modules/thienan/news.php?id=http://www.betop24.com%2F
https://www.weerstationgeel.be/template/pages/station/redirect.php?url=http://www.betop24.com%2F
http://www.doitweb365.de/scripts/doitweb.exe/rasklickzaehler2?http://www.betop24.com%2F
https://www.guadamur.eu/template/pages/station/redirect.php?url=http://www.betop24.com%2F
https://weather.cube.com.gr/pages/station/redirect.php?url=http://www.betop24.com%2F
http://www.fimmgviterbo.org/mobfimmgviterbo/index.php?nametm=counter&idbanner=4&dir_link=http://www.betop24.com%2F
https://wuqishi.com/wp-content/themes/begin/inc/go.php?url=http://www.betop24.com%2F
http://www.mckinneyfarm.com/template/plugins/stationExtremes/redirect.php?url=http://www.betop24.com%2F
http://search.pointcom.com/k.php?ai=&url=http://www.betop24.com%2F
http://www.cercasostituto.it/index.php?name=GestBanner&file=counter&idbanner=40&dir_link=http://www.betop24.com%2F
http://vsvejr.dk/mt/plugins/stationExtremes/redirect.php?url=http://www.betop24.com%2F
http://www.meteo-leran.fr/meteotemplate/template/plugins/deviations/redirect.php?url=http://www.betop24.com%2F
https://www.eurobichons.com/fda%20alerts.php?url=http://www.betop24.com%2F
https://mydojo.at/de_AT/karate/weiterleitung?redirect=http://www.betop24.com%2F
http://click.phosphodiesterase4.com/k.php?ai=&url=http://www.betop24.com%2F
http://www.mariahownersclub.com/forum/redirect-to/?redirect=http://www.betop24.com%2F
http://www.wildromance.com/buy.php?url=http://www.betop24.com%2F&store=iBooks&book=omk-ibooks-us
http://mapleriverweather.com/mobile/pages/station/redirect.php?url=http://www.betop24.com%2F
http://www.archijob.co.il/index/comp_website.asp?companyId=1469&website=http://www.betop24.com%2F
https://student-helpr.rminds.dev/redirect?redirectTo=http://www.betop24.com%2F
http://www.kalinna.de/url?q=http://www.betop24.com%2F
http://www.hartmanngmbh.de/url?q=http://www.betop24.com%2F
https://www.the-mainboard.com/proxy.php?link=http://www.betop24.com%2F
https://www.betamachinery.com/?URL=http://www.betop24.com%2F
http://nishiyama-takeshi.com/mobile2/mt4i.cgi?id=3&mode=redirect&no=67&ref_eid=671&url=http://www.betop24.com%2F
http://www.sprang.net/url?q=http://www.betop24.com%2F
http://local.rongbachkim.com/rdr.php?url=http://www.betop24.com%2F
http://bachecauniversitaria.it/link/frm_top.php?url=http://www.betop24.com%2F
https://www.stcwdirect.com/redirect.php?url=http://www.betop24.com%2F
http://forum.vcoderz.com/externalredirect.php?url=http://www.betop24.com%2F
https://www.momentumstudio.com/?URL=http://www.betop24.com%2F
http://kuzu-kuzu.com/l.cgi?http://www.betop24.com%2F
http://sintez-oka.ru/bitrix/redirect.php?goto=http://www.betop24.com%2F
https://utmagazine.ru/r?url=http://www.betop24.com%2F
http://www.evrika41.ru/redirect?url=http://www.betop24.com%2F
http://expomodel.ru/bitrix/redirect.php?goto=http://www.betop24.com%2F
https://facto.ru/bitrix/rk.php?goto=http://www.betop24.com%2F
http://fallout3.ru/utils/ref.php?url=http://www.betop24.com%2F
https://fc-zenit.ru/bitrix/redirect.php?goto=http://www.betop24.com%2F
http://forum-region.ru/forum/away.php?s=http://www.betop24.com%2F
http://forumdate.ru/redirect-to/?redirect=http://www.betop24.com%2F
http://shckp.ru/ext_link?url=http://www.betop24.com%2F
https://shinglas.ru/bitrix/redirect.php?goto=http://www.betop24.com%2F
https://sibran.ru/bitrix/redirect.php?goto=http://www.betop24.com%2F
http://karkom.de/url?q=http://www.betop24.com%2F
http://kens.de/url?q=http://www.betop24.com%2F
http://kinderundjugendpsychotherapie.de/url?q=http://www.betop24.com%2F
http://kinhtexaydung.net/redirect/?url=http://www.betop24.com%2F
https://www.hudsonvalleytraveler.com/Redirect?redirect_url=http://www.betop24.com%2F
http://neoromance.info/link/rank.cgi?mode=link&id=26&url=http://www.betop24.com%2F
http://www.hainberg-gymnasium.com/url?q=http://www.betop24.com%2F
https://befonts.com/checkout/redirect?url=http://www.betop24.com%2F
https://ref.webhostinghub.com/scripts/click.php?ref_id=Eduarea&desturl=http://www.betop24.com%2F
https://www.usap.gov/externalsite.cfm?http://www.betop24.com%2F
https://app.espace.cool/clientapi/subscribetocalendar/974?url=http://www.betop24.com%2F
http://111056.net/yomisearch/rank.cgi?mode=link&id=6205&url=http://www.betop24.com%2F
http://stanko.tw1.ru/redirect.php?url=http://www.betop24.com%2F
http://www.sozialemoderne.de/url?q=http://www.betop24.com%2F
http://www.showb.com/search/ranking.cgi?mode=link&id=7083&url=http://www.betop24.com%2F
https://telepesquisa.com/redirect?page=redirect&site=http://www.betop24.com%2F
http://imagelibrary.asprey.com/?URL=spo337.com
http://ime.nu/http://www.betop24.com%2F
http://interflex.biz/url?q=http://www.betop24.com%2F
http://ivvb.de/url?q=http://www.betop24.com%2F
http://j.lix7.net/?http://www.betop24.com%2F
http://jacobberger.com/?URL=spo337.com
http://jahn.eu/url?q=http://www.betop24.com%2F
http://jamesvelvet.com/?URL=spo337.com
http://jla.drmuller.net/r.php?url=http://www.betop24.com%2F
http://jump.pagecs.net/http://www.betop24.com%2F
http://kagarin.net/cgi/mt/mt4i.cgi?id=2&mode=redirect&no=330&ref_eid=103&url=http://www.betop24.com%2F
http://kancler-k.com.ua/bitrix/redirect.php?event1=&event2=&event3=&goto=http://www.betop24.com%2F
http://kancler-k.com/bitrix/redirect.php?event1=&event2=&event3=&goto=http://www.betop24.com%2F
https://seoandme.ru/bitrix/redirect.php?goto=http://www.betop24.com%2F
https://s-online.ru/bitrix/redirect.php?goto=http://www.betop24.com%2F
https://rssfeeds.wtsp.com/~/t/0/0/wtsp/home/~spo337.com
https://rostovmama.ru/redirect?url=http://www.betop24.com%2F
https://rev1.reversion.jp/redirect?url=http://www.betop24.com%2F
https://supplier-portal-uat.daimler.com/external-link.jspa?url=http://www.betop24.com%2F
https://strelmag.ru/bitrix/rk.php?goto=http://www.betop24.com%2F
https://spb90.ru/bitrix/redirect.php?goto=http://www.betop24.com%2F
https://spartak.ru/bitrix/redirect.php?goto=http://www.betop24.com%2F
https://sovaisova.ru/bitrix/redirect.php?event1=2009_mainpage&event2=go_www&event3=&goto=http://www.betop24.com%2F
https://socport.ru/redirect?url=http://www.betop24.com%2F
https://slashwrestling.com/cgi-bin/redirect.cgi?http://www.betop24.com%2F
http://mosprogulka.ru/go?http://www.betop24.com%2F
https://uniline.co.nz/Document/Url/?url=http://www.betop24.com%2F
https://tracker.onrecruit.net/api/v1/redirect/?redirect_to=http://www.betop24.com%2F
http://slipknot1.info/go.php?url=http://www.betop24.com%2F
https://www.samovar-forum.ru/go?http://www.betop24.com%2F
http://staldver.ru/go.php?go=http://www.betop24.com%2F
https://www.star174.ru/redir.php?url=http://www.betop24.com%2F
https://staten.ru/bitrix/rk.php?goto=http://www.betop24.com%2F
https://stav-geo.ru/go?http://www.betop24.com%2F
http://stopcran.ru/go?http://www.betop24.com%2F
http://studioad.ru/go?http://www.betop24.com%2F
http://swepub.kb.se/setattribute?language=en&redirect=http://www.betop24.com%2F
https://www.licnioglasi.org/index.php?thememode=full;redirect=http://www.betop24.com%2F
http://www.rexart.com/cgi-rexart/al/affiliates.cgi?aid=872&redirect=http://www.betop24.com%2F
http://www2.smartmail.com.ar/tl.php?p=hqf/f94/rs/1fp/4c0/rs//http://www.betop24.com%2F
http://old.roofnet.org/external.php?link=http://www.betop24.com%2F
http://www.bucatareasa.ro/link.php?url=http://www.betop24.com%2F
https://forum.solidworks.com/external-link.jspa?url=http://www.betop24.com%2F
https://foro.infojardin.com/proxy.php?link=http://www.betop24.com%2F
https://de.flavii.de/index.php?flavii=linker&link=http://www.betop24.com%2F
https://dakke.co/redirect/?url=http://www.betop24.com%2F
https://creativecommons.org/choose/results-one?q_1=2&q_1=1&field_attribute_to_url=http://www.betop24.com%2F
https://www.flyingsamaritans.net/Startup/SetupSite.asp?RestartPage=http://www.betop24.com%2F
https://www.ewind.cz/index.php?page=home/redirect&url=http://www.betop24.com%2F
https://www.eas-racing.se/gbook/go.php?url=http://www.betop24.com%2F
https://www.direkt-einkauf.de/includes/refer.php?id=170&url=http://www.betop24.com%2F
https://www.dialogportal.com/Services/Forward.aspx?link=http://www.betop24.com%2F
https://www.curseforge.com/linkout?remoteUrl=http://www.betop24.com%2F
https://www.counterwelt.com/charts/click.php?user=14137&link=http://www.betop24.com%2F
https://www.bing.com/news/apiclick.aspx?ref=FexRss&aid=&tid=9BB77FDA801248A5AD23FDBDD5922800&url=http://www.betop24.com%2F
https://tvtropes.org/pmwiki/no_outbounds.php?o=http://www.betop24.com%2F
https://trello.com/add-card?source=mode=popup&name=click+here&desc=http://www.betop24.com%2F
https://transtats.bts.gov/exit.asp?url=http://www.betop24.com%2F
https://sutd.ru/links.php?go=http://www.betop24.com%2F
https://abiznes.com.ua/bitrix/redirect.php?event1=&event2=&event3=&goto=http://www.betop24.com%2F
http://www.sv-mama.ru/shared/go.php?url=http://www.betop24.com%2F
http://www.sofion.ru/banner.php?r1=41&r2=2234&goto=http://www.betop24.com%2F
http://www.rss.geodles.com/fwd.php?url=http://www.betop24.com%2F
http://www.imsnet.at/LangChange.aspx?uri=http://www.betop24.com%2F
http://www.hon-cafe.net/cgi-bin/re.cgi?lid=hmw&url=http://www.betop24.com%2F
http://www.glorioustronics.com/redirect.php?link=http://www.betop24.com%2F
http://rostovklad.ru/go.php?http://www.betop24.com%2F
http://portalnp.snauka.ru/bitrix/redirect.php?goto=http://www.betop24.com%2F
http://markiza.me/bitrix/rk.php?goto=http://www.betop24.com%2F
http://jump.5ch.net/?http://www.betop24.com%2F
http://imperialoptical.com/news-redirect.aspx?url=http://www.betop24.com%2F
http://hellothai.com/wwwlink/wwwredirect.asp?hp_id=1242&url=http://www.betop24.com%2F
http://guru.sanook.com/?URL=http://www.betop24.com%2F
http://fr.knubic.com/redirect_to?url=http://www.betop24.com%2F
http://branch.app.link/?$deeplink_path=article/jan/123&$fallback_url=http://www.betop24.com%2F
http://biz-tech.org/bitrix/rk.php?goto=http://www.betop24.com%2F
https://www.hottystop.com/cgi-bin/at3/out.cgi?id=12&trade=http://www.betop24.com%2F
https://www.hobowars.com/game/linker.php?url=http://www.betop24.com%2F
https://www.hentainiches.com/index.php?id=derris&tour=http://www.betop24.com%2F
https://www.gudarjavalambre.com/sections/miscelany/link.php?url=http://www.betop24.com%2F
https://www.greencom.ru/catalog/irrigation_systems.html?jump_site=2008&url=http://www.betop24.com%2F
https://www.girlznation.com/cgi-bin/atc/out.cgi?id=50&l=side&u=http://www.betop24.com%2F
https://www.funeralunion.org/delete-company?nid=39&element=http://www.betop24.com%2F
https://www.fudbal91.com/tz.php?zone=America/Iqaluit&r=http://www.betop24.com%2F
https://www.frodida.org/BannerClick.php?BannerID=29&LocationURL=http://www.betop24.com%2F
https://www.autopartskart.com/buyfromamzon.php?url=http://www.betop24.com%2F
https://www.autoandrv.com/linkout.aspx?websiteurl=http://www.betop24.com%2F
https://www.art-prizes.com/AdRedirector.aspx?ad=MelbPrizeSculpture_2017&target=http://www.betop24.com%2F
https://www.adminer.org/redirect/?url=http://www.betop24.com%2F
https://wirelessestimator.com/advertise/newsletter_redirect.php?url=http://www.betop24.com%2F
https://wasitviewed.com/index.php?href=http://www.betop24.com%2F
https://naruto.su/link.ext.php?url=http://www.betop24.com%2F
https://justpaste.it/redirect/172fy/http://www.betop24.com%2F
https://jt-pr-dot-yamm-track.appspot.com/Redirect?ukey=1iXi3dF8AuzUIsgifbcVnqqx-anF4B8R-9PC3UGhHO3E-1131306248&key=YAMMID-79725512&link=http://www.betop24.com%2F
https://community.rsa.com/external-link.jspa?url=http://www.betop24.com%2F
https://community.nxp.com/external-link.jspa?url=http://www.betop24.com%2F
https://community.esri.com/external-link.jspa?url=http://www.betop24.com%2F
https://community.cypress.com/external-link.jspa?url=http://www.betop24.com%2F
https://club.panasonic.jp/member/terms/?siteId=B1&returnURL=http://www.betop24.com%2F
https://cdn.iframe.ly/api/iframe?url=http://www.betop24.com%2F
https://bukkit.org/proxy.php?link=http://www.betop24.com%2F
https://branch.app.link/?$deeplink_path=article%2Fjan%2F123&$fallback_url=https%3A%2F%2Fspo337.com&channel=facebook&feature=affiliate
https://boowiki.info/go.php?go=http://www.betop24.com%2F
https://blaze.su/bitrix/redirect.php?event1=&event2=&event3=&goto=http://www.betop24.com%2F
https://bbs.pku.edu.cn/v2/jump-to.php?url=http://www.betop24.com%2F
https://bares.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F
http://www.nuttenzone.at/jump.php?url=http://www.betop24.com%2F
http://www.myhottiewife.com/cgi-bin/arpro/out.cgi?id=Jojo&url=http://www.betop24.com%2F
http://www.mistress-and-slave.com/cgi-bin/out.cgi?id=123crush&url=http://www.betop24.com%2F
http://www.johnvorhees.com/gbook/go.php?url=http://www.betop24.com%2F
http://www.etis.ford.com/externalURL.do?url=http://www.betop24.com%2F
http://www.erotikplatz.at/redirect.php?id=939&mode=fuhrer&url=http://www.betop24.com%2F
http://www.chungshingelectronic.com/redirect.asp?url=http://www.betop24.com%2F
http://www.bdsmandfetish.com/cgi-bin/sites/out.cgi?id=mandymon&url=http://www.betop24.com%2F
http://visits.seogaa.ru/redirect/?g=http://www.betop24.com%2F
http://tharp.me/?url_to_shorten=http://www.betop24.com%2F
http://stroysoyuz.ru/bitrix/rk.php?goto=http://www.betop24.com%2F
http://speakrus.ru/links.php?go=http://www.betop24.com%2F
http://spbstroy.ru/bitrix/redirect.php?goto=http://www.betop24.com%2F
http://solo-center.ru/links.php?go=http://www.betop24.com%2F
http://sennheiserstore.ru/bitrix/rk.php?goto=http://www.betop24.com%2F
http://school364.spb.ru/bitrix/rk.php?goto=http://www.betop24.com%2F
http://sc.sie.gov.hk/TuniS/spo337.com
http://rzngmu.ru/go?http://www.betop24.com%2F
http://2010.russianinternetweek.ru/bitrix/rk.php?goto=http://www.betop24.com%2F
https://xat.com/web_gear/chat/linkvalidator.php?link=http://www.betop24.com%2F
https://www.yeaah.com/disco/DiscoGo.asp?ID=3435&Site=http://www.betop24.com%2F
https://www.woodlist.us/delete-company?nid=13964&element=http://www.betop24.com%2F
https://www.viecngay.vn/go?to=http://www.betop24.com%2F
https://www.uts.edu.co/portal/externo.php?id=http://www.betop24.com%2F
https://www.talgov.com/Main/exit.aspx?url=http://www.betop24.com%2F
https://www.skoberne.si/knjiga/go.php?url=http://www.betop24.com%2F
https://www.serie-a.ru/bitrix/redirect.php?goto=http://www.betop24.com%2F
https://www.russianrobotics.ru/bitrix/rk.php?goto=http://www.betop24.com%2F
https://www.ruchnoi.ru/ext_link?url=http://www.betop24.com%2F
https://www.rprofi.ru/bitrix/rk.php?goto=http://www.betop24.com%2F
https://www.roccotube.com/cgi-bin/at3/out.cgi?id=27&tag=toplist&trade=http://www.betop24.com%2F
https://www.nyl0ns.com/cgi-bin/a2/out.cgi?id=43&l=btop&u=http://www.betop24.com%2F
https://www.meetme.com/apps/redirect/?url=http://www.betop24.com%2F
https://www.kath-kirche-kaernten.at/pfarren/pfarre/C3014?URL=http://www.betop24.com%2F
https://www.interpals.net/urlredirect.php?href=http://www.betop24.com%2F
https://www.interecm.com/interecm/tracker?op=click&id=5204.db2&url=http://www.betop24.com%2F
https://www.fca.gov/?URL=http://www.betop24.com%2F
https://savvylion.com/?bmDomain=spo337.com
https://www.soyyooestacaido.com/spo337.com
https://www.gta.ru/redirect/spo337.com
https://directx10.org/go?http://www.betop24.com%2F
https://mejeriet.dk/link.php?id=spo337.com
https://ezdihan.do.am/go?http://www.betop24.com%2F
https://icook.ucoz.ru/go?http://www.betop24.com%2F
https://megalodon.jp/?url=http://www.betop24.com%2F
https://www.pasco.k12.fl.us/?URL=spo337.com
https://anolink.com/?link=http://www.betop24.com%2F
https://www.questsociety.ca/?URL=spo337.com
https://www.disl.edu/?URL=http://www.betop24.com%2F
https://holidaykitchens.com/?URL=spo337.com
https://www.mbcarolinas.org/?URL=spo337.com
https://sepoa.fr/wp/go.php?http://www.betop24.com%2F
https://world-source.ru/go?http://www.betop24.com%2F
https://mail2.mclink.it/SRedirect/spo337.com
https://www.swleague.ru/go?http://www.betop24.com%2F
https://nazgull.ucoz.ru/go?http://www.betop24.com%2F
https://www.rosbooks.ru/go?http://www.betop24.com%2F
https://pavon.kz/proxy?url=http://www.betop24.com%2F
https://beskuda.ucoz.ru/go?http://www.betop24.com%2F
https://richmonkey.biz/go/?http://www.betop24.com%2F
https://vlpacific.ru/?goto=http://www.betop24.com%2F
https://www.google.co.il/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.rs/url?sa=t&url=http://www.betop24.com%2F/
https://maps.google.com.ni/url?sa=t&url=http://www.betop24.com%2F/
https://ref.gamer.com.tw/redir.php?url=http://www.betop24.com%2F/
https://www.google.lt/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.ae/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.si/url?sa=t&url=http://www.betop24.com%2F/
https://images.google.com.co/url?sa=t&url=http://www.betop24.com%2F/
http://www.google.fi/url?sa=t&url=http://www.betop24.com%2F/
https://cse.google.fi/url?q=http://www.betop24.com%2F/
http://www.google.com.sg/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.hr/url?sa=t&url=http://www.betop24.com%2F/
http://images.google.co.nz/url?sa=t&url=http://www.betop24.com%2F/
https://qatar.vcu.edu/?URL=http://www.betop24.com%2F/
https://images.google.com.pe/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.ee/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.lv/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.com.pk/url?sa=t&url=http://www.betop24.com%2F/
https://maps.google.com.np/url?sa=t&url=http://www.betop24.com%2F/
https://maps.google.co.ve/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.lk/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.com.bd/url?sa=t&url=http://www.betop24.com%2F/
https://images.google.com.ec/url?sa=t&url=http://www.betop24.com%2F/
https://images.google.by/url?sa=t&url=http://www.betop24.com%2F/
http://maps.google.cz/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.com.ng/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.lu/url?sa=t&url=http://www.betop24.com%2F/
https://images.google.com.uy/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.co.cr/url?sa=t&url=http://www.betop24.com%2F/
https://images.google.tn/url?sa=t&url=http://www.betop24.com%2F/
http://www.london.umb.edu/?URL=http://www.betop24.com%2F/
https://www.google.com.do/url?sa=t&url=http://www.betop24.com%2F/
https://images.google.com.pr/url?sa=t&url=http://www.betop24.com%2F/
https://ceskapozice.lidovky.cz/redir.aspx?url=http://www.betop24.com%2F/
https://www.google.ba/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.mu/url?sa=t&url=http://www.betop24.com%2F/
https://images.google.co.ke/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.is/url?sa=t&url=http://www.betop24.com%2F/
https://images.google.com.lb/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.com.gt/url?sa=t&url=http://www.betop24.com%2F/
https://maps.google.com.py/url?sa=t&url=http://www.betop24.com%2F/
https://maps.google.dz/url?sa=t&url=http://www.betop24.com%2F/
https://images.google.cat/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.hn/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.com.bo/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.com.mt/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.kz/url?sa=t&url=http://www.betop24.com%2F/
https://images.google.com.kh/url?sa=t&url=http://www.betop24.com%2F/
https://images.google.cm/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.com.sv/url?sa=t&url=http://www.betop24.com%2F/
https://maps.google.ci/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.jo/url?sa=t&url=http://www.betop24.com%2F/
https://maps.google.com.bh/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.com.pa/url?sa=t&url=http://www.betop24.com%2F/
https://images.google.co.bw/url?sa=t&url=http://www.betop24.com%2F/
https://images.google.am/url?sa=t&url=http://www.betop24.com%2F/
https://images.google.az/url?sa=t&url=http://www.betop24.com%2F/
https://maps.google.ge/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.com.cu/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.com.kw/url?sa=t&url=http://www.betop24.com%2F/
https://images.google.co.ma/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.com.gh/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.mk/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.co.ug/url?sa=t&url=http://www.betop24.com%2F/
https://maps.google.as/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.ad/url?sa=t&url=http://www.betop24.com%2F/
http://www.astro.wisc.edu/?URL=http://www.betop24.com%2F/
http://www.astro.wisc.edu/?URL=/http://www.betop24.com%2F/
https://maps.google.cd/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.com.cy/url?sa=t&url=http://www.betop24.com%2F/
https://images.google.bs/url?sa=t&url=http://www.betop24.com%2F/
http://www.google.com.mx/url?sa=t&url=http://www.betop24.com%2F/
http://images.google.hu/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.li/url?sa=t&url=http://www.betop24.com%2F/
https://images.google.com.bz/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.com.af/url?sa=t&url=http://www.betop24.com%2F/
https://images.google.com.ag/url?sa=t&url=http://www.betop24.com%2F/
https://images.google.bi/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.mn/url?sa=t&url=http://www.betop24.com%2F/
https://maps.google.tt/url?sa=t&url=http://www.betop24.com%2F/
https://images.google.com.na/url?sa=t&url=http://www.betop24.com%2F/
https://images.google.com.qa/url?sa=t&url=http://www.betop24.com%2F/
https://images.google.sn/url?sa=t&url=http://www.betop24.com%2F/
https://images.google.al/url?sa=t&url=http://www.betop24.com%2F/
https://images.google.fm/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.iq/url?sa=t&url=http://www.betop24.com%2F/
https://images.google.com.gi/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.je/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.mg/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.com.om/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.dj/url?sa=t&url=http://www.betop24.com%2F/
https://images.google.com.ly/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.com.jm/url?sa=t&url=http://www.betop24.com%2F/
https://images.google.com.et/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.md/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.sh/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.co.tz/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.me/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.kg/url?sa=t&url=http://www.betop24.com%2F/
http://www.google.mw/url?q=http://www.betop24.com%2F/
https://www.google.mw/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.ht/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.rw/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.ms/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.ps/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.com.bn/url?sa=t&url=http://www.betop24.com%2F/
https://cse.google.pt/url?q=http://www.betop24.com%2F/
http://images.google.pt/url?sa=t&url=http://www.betop24.com%2F/
http://www.google.co.id/url?sa=t&url=http://www.betop24.com%2F/
https://images.google.co.ls/url?sa=t&url=http://www.betop24.com%2F/
https://images.google.bf/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.la/url?sa=t&url=http://www.betop24.com%2F/
https://maps.google.com.fj/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.mv/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.gg/url?sa=t&url=http://www.betop24.com%2F/
https://www.triathlon.org/?URL=/http://www.betop24.com%2F/
http://www.google.com.ar/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.co.mz/url?sa=t&url=http://www.betop24.com%2F/
http://www.google.co.th/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.gm/url?sa=t&url=http://www.betop24.com%2F/
http://www.google.no/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.gl/url?sa=t&url=http://www.betop24.com%2F/
http://maps.google.co.za/url?q=http://www.betop24.com%2F/
http://images.google.ro/url?sa=t&url=http://www.betop24.com%2F/
https://cse.google.com.vn/url?q=http://www.betop24.com%2F/
http://cse.google.com.ph/url?q=http://www.betop24.com%2F/
https://wikimapia.org/external_link?url=http://http://www.betop24.com%2F/
http://images.google.com.pk/url?q=http://www.betop24.com%2F/
http://maps.google.gr/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.gp/url?sa=t&url=http://www.betop24.com%2F/
http://www.google.ie/url?q=http://www.betop24.com%2F/
https://images.google.bg/url?q=http://www.betop24.com%2F/
https://images.google.com/url?q=http://www.betop24.com%2F/
http://www.cssdrive.com/?URL=/http://www.betop24.com%2F/
http://www.google.sk/url?q=http://www.betop24.com%2F/
http://www.google.co.il/url?q=http://www.betop24.com%2F/
http://cse.google.rs/url?q=http://www.betop24.com%2F/
http://www.earth-policy.org/?URL=http://www.betop24.com%2F/
http://maps.google.lt/url?q=http://www.betop24.com%2F/
http://maps.google.ae/url?q=http://www.betop24.com%2F/
http://www.google.com.co/url?q=http://www.betop24.com%2F/
http://maps.google.hr/url?q=http://www.betop24.com%2F/
https://community.cypress.com/external-link.jspa?url=http://http://www.betop24.com%2F/
https://community.rsa.com/external-link.jspa?url=http://www.betop24.com%2F/
http://www.pasco.k12.fl.us/?URL=http://www.betop24.com%2F/
https://cse.google.com/url?q=http://www.betop24.com%2F/
https://maps.google.ee/url?q=http://www.betop24.com%2F/
http://maps.google.lv/url?q=http://www.betop24.com%2F/
http://www.google.com.np/url?q=http://www.betop24.com%2F/
http://www.bookmerken.de/?url=http://www.betop24.com%2F/
http://maps.google.co.ve/url?q=http://www.betop24.com%2F/
http://maps.google.com.ec/url?q=http://www.betop24.com%2F/
http://cse.google.com.bd/url?q=http://www.betop24.com%2F/
http://maps.google.by/url?q=http://www.betop24.com%2F/
http://maps.google.lu/url?q=http://www.betop24.com%2F/
http://images.google.com.uy/url?q=http://www.betop24.com%2F/
http://www.google.co.cr/url?q=http://www.betop24.com%2F/
http://cse.google.tn/url?q=http://www.betop24.com%2F/
http://www.google.mu/url?q=http://www.betop24.com%2F/
https://www.fuzokubk.com/cgi-bin/LinkO.cgi?u=http://www.betop24.com%2F/
http://images.google.com.pr/url?q=http://www.betop24.com%2F/
https://legacy.aom.org/verifymember.asp?nextpage=http://http://www.betop24.com%2F/
http://www.novalogic.com/remote.asp?NLink=http://www.betop24.com%2F/
http://www.orthodoxytoday.org/?URL=http://www.betop24.com%2F/
https://bukkit.org/proxy.php?link=http://www.betop24.com%2F/
http://www.searchdaimon.com/?URL=http://www.betop24.com%2F/
http://icecap.us/?URL=http://www.betop24.com%2F/
https://www.adminer.org/redirect/?url=http://www.betop24.com%2F/
http://www.arakhne.org/redirect.php?url=http://www.betop24.com%2F/
https://www.raincoast.com/?URL=http://www.betop24.com%2F/
http://4vn.eu/forum/vcheckvirus.php?url=http://www.betop24.com%2F/
http://holidaykitchens.com/?URL=http://www.betop24.com%2F/
http://www.adhub.com/cgi-bin/webdata_pro.pl?cgifunction=clickthru&url=http://www.betop24.com%2F/
https://client.paltalk.com/client/webapp/client/External.wmt?url=http://www.betop24.com%2F/
http://www.virtual-egypt.com/framed/framed.cgi?url==http://www.betop24.com%2F/
http://www.webclap.com/php/jump.php?url=http://www.betop24.com%2F/
http://urlxray.com/display.php?url=http://www.betop24.com%2F/
http://daidai.gamedb.info/wiki/?cmd=jumpto&r=http://www.betop24.com%2F/
https://www.hobowars.com/game/linker.php?url=http://www.betop24.com%2F/
http://www.clevelandbay.com/?URL=http://www.betop24.com%2F/
https://www.wheretoskiandsnowboard.com/?URL=http://www.betop24.com%2F/
http://archive.paulrucker.com/?URL=http://www.betop24.com%2F/
http://openroadbicycles.com/?URL=http://www.betop24.com%2F/
http://www.onesky.ca/?URL=http://www.betop24.com%2F/
https://www.sjpcommunications.org/?URL=http://www.betop24.com%2F/
https://www.stmarysbournest.com/?URL=http://www.betop24.com%2F/
http://jc-log.jmirus.de/?URL=http://www.betop24.com%2F/
http://www.shamelesstraveler.com/?URL=http://www.betop24.com%2F/
https://ilns.ranepa.ru/bitrix/rk.php?goto=http://www.betop24.com%2F/
https://inva.gov.kz/ru/redirect?url=http://www.betop24.com%2F/
https://m.fishki.net/go/?url=http://www.betop24.com%2F/
https://www.rospotrebnadzor.ru/bitrix/redirect.php?event1=file&event2=download&event3=prilozheniya-k-prikazu-1018.doc&goto=http://www.betop24.com%2F/
http://76-rus.ru/bitrix/redirect.php?event1=catalog_out&event2=http2FEEECEBEEE3%EEEE-E5F1FBEDF0&goto=http://www.betop24.com%2F/
http://cenproxy.mnpals.net/login?url=http://www.betop24.com%2F/
http://oca.ucsc.edu/login?url=http://www.betop24.com%2F/
http://www.hyiphistory.com/visit.php?url=http://www.betop24.com%2F/
http://www.liveinternet.ru/journal_proc.php?action=redirect&url=http://www.betop24.com%2F/
https://old.fishki.net/go/?url=http://www.betop24.com%2F/
https://www.banki.ru/away/?url=http://www.betop24.com%2F/
https://www.institutoquinquelamartin.edu.ar/Administracion/top-10-cuadros-mas-famosos6-1/?unapproved=10807http://www.betop24.com%2F/
https://mcpedl.com/leaving/?url=https%3A%2F%2Fwww.statusvideosongs.in%2F&cookie_check=1http://www.betop24.com%2F/
http://gelendzhik.org/bitrix/redirect.php?event1=&event2=&event3=&goto=http://www.betop24.com%2F/
http://sasisa.ru/forum/out.php?link=%3F&yes=1http://www.betop24.com%2F/
http://usolie.info/bitrix/redirect.php?event1=&event2=&event3=&goto=http://www.betop24.com%2F/
http://vologda-portal.ru/bitrix/redirect.php?event1=news_out&event2=farsicontent.blogspot.com&event3=C1CEAB8CEBE5F1AAFFF2EAA0F8E0C2A5F120EAAEFBEAF1B1E2F0E8A4FF29&goto=http://www.betop24.com%2F/
http://vstu.ru/bitrix/rk.php?goto=http://www.betop24.com%2F/
http://www.bsaa.edu.ru/bitrix/rk.php?goto=http://www.betop24.com%2F/
http://www.geogr.msu.ru/bitrix/rk.php?goto=http://www.betop24.com%2F/
https://academy.1c-bitrix.ru/bitrix/redirect.php?event1=acsdemy&event2=usable&event3=&goto=http://www.betop24.com%2F/
https://adamb-bartos.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
https://adamburda.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
https://adammikulasek.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
https://adamvanek.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
https://adamvasina.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
https://agalarov.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
https://alenapekarova.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
https://alenapitrova.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
https://alesbeseda.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
https://alesmerta.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
https://alexova.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
https://andreanovotna1.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
https://maps.google.com/url?sa=t&url=http://www.betop24.com%2F/
http://www.google.com/url?q=http://www.betop24.com%2F/
https://www.google.co.uk/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.fr/url?sa=t&url=http://www.betop24.com%2F/
https://maps.google.es/url?sa=t&url=http://www.betop24.com%2F/
https://maps.google.ca/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.nl/url?sa=t&url=http://www.betop24.com%2F/
https://www.marcellusmatters.psu.edu/?URL=http://www.betop24.com%2F/
https://www.google.com.au/url?sa=t&url=http://www.betop24.com%2F/
https://v1.addthis.com/live/redirect/?url=http://www.betop24.com%2F/
http://images.google.de/url?sa=t&url=http://www.betop24.com%2F/
https://maps.google.be/url?sa=t&url=http://www.betop24.com%2F/
https://maps.google.ru/url?sa=t&url=http://www.betop24.com%2F/
http://www.google.se/url?sa=t&source=web&cd=1&ved=0CBcQFjAA&url=http://www.betop24.com%2F/
https://www.google.se/url?sa=t&url=http://www.betop24.com%2F/
https://maps.google.com.tr/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.dk/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.com.hk/url?sa=t&url=http://www.betop24.com%2F/
https://images.google.com.mx/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.hu/url?sa=t&url=http://www.betop24.com%2F/
https://images.google.com.au/url?q=http://www.betop24.com%2F/
https://maps.google.pt/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.co.nz/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202766&url=http://http://www.betop24.com%2F/
https://www.google.com.ar/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.co.th/url?sa=i&rct=j&q=&esrc=s&source=images&cd=&cad=rja&uact=8&ved=0ahUKEwjB4_-A3tnWAhWHOY8KHTcgDxMQjRwIBw&url=http://www.betop24.com%2F/
https://www.google.co.th/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.com.ua/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.no/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.co.za/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.ro/url?sa=t&url=http://www.betop24.com%2F/
https://images.google.com.vn/url?sa=t&url=http://www.betop24.com%2F/
https://guru.sanook.com/?URL=http://www.betop24.com%2F/
https://images.google.com.ph/url?sa=t&url=http://www.betop24.com%2F/
http://wikimapia.org/external_link?url=http://www.betop24.com%2F/
https://maps.google.cl/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.ie/url?sa=t&url=http://www.betop24.com%2F/
https://fjb.kaskus.co.id/redirect?url=http://www.betop24.com%2F/
https://maps.google.com.my/url?sa=t&url=http://www.betop24.com%2F/
https://www.google.sk/url?sa=t&url=http://www.betop24.com%2F/
https://images.google.com/url?sa=t&url=http://www.betop24.com%2F/
http://www.google.com.tw/url?sa=t&url=http://www.betop24.com%2F/
http://www.morrowind.ru/redirect/grillages-wunschel.fr/?URL=www.betop24.com%2Ffeft/ref/xiswi/
https://smmry.com/xn-herzrhythmusstrungen-hbc.biz/goto.php?site=www.betop24.com%2F
http://www.ut2.ru/redirect/slrc.org/?URL=www.betop24.com%2F
https://jump.5ch.net/?blingguard.com/?URL=www.betop24.com%2F
https://jump.5ch.net/?ponsonbyacupunctureclinic.co.nz/?URL=www.betop24.com%2F
http://bios.edu/?URL=puttyandpaint.com/?URL=www.betop24.com%2F
https://cwcab.com/?URL=eaglesgymnastics.com/?URL=www.betop24.com%2F
http://www.ut2.ru/redirect/weburg.net/redirect?url=www.betop24.com%2F
http://2ch.io/assertivenorthwest.com/?URL=www.betop24.com%2F
http://www.morrowind.ru/redirect/slighdesign.com/?URL=www.betop24.com%2F
http://ime.nu/gumexslovakia.sk/?URL=www.betop24.com%2F
http://www.morrowind.ru/redirect/roserealty.com.au/?URL=www.betop24.com%2F
http://www.allods.net/redirect/magenta-mm.com/?URL=www.betop24.com%2F
http://bios.edu/?URL=shavermfg.com/?URL=www.betop24.com%2F
http://www.allods.net/redirect/healthyeatingatschool.ca/?URL=www.betop24.com%2Ffeft/ref/xiswi/
http://www.gta.ru/redirect/dcfossils.org/?URL=www.betop24.com%2F
https://jump.5ch.net/?pro-net.se/?URL=www.betop24.com%2F
http://bios.edu/?URL=civicvoice.org.uk/?URL=www.betop24.com%2F
https://jump.5ch.net/?arbor-tech.be/?URL=www.betop24.com%2F
http://www.allods.net/redirect/theaustonian.com/?URL=www.betop24.com%2F
http://ime.nu/basebusiness.com.au/?URL=www.betop24.com%2F
https://cwcab.com/?URL=hfw1970.de/redirect.php?url=www.betop24.com%2F
http://www.ut2.ru/redirect/firma.hr/?URL=www.betop24.com%2F
http://www.nwnights.ru/redirect/giruna.hu/redirect.php?url=www.betop24.com%2F
http://bios.edu/?URL=weburg.net/redirect?url=www.betop24.com%2F
http://www.gta.ru/redirect/emotional.ro/?URL=www.betop24.com%2F
http://www.gta.ru/redirect/couchsrvnation.com/?URL=www.betop24.com%2Ffeft/ref/xiswi/
http://www.allods.net/redirect/turbo-x.hr/?URL=www.betop24.com%2F
http://www.gta.ru/redirect/albins.com.au/?URL=www.betop24.com%2F
http://www.ut2.ru/redirect/emophilips.com/?URL=www.betop24.com%2F
http://bios.edu/?URL=pcrnv.com.au/?URL=www.betop24.com%2F
http://2ch.io/md-technical.com/?URL=www.betop24.com%2Ffeft/ref/xiswi/
http://www.ut2.ru/redirect/stcroixblades.com/?URL=www.betop24.com%2F
https://jump.5ch.net/?sassyj.net/?URL=www.betop24.com%2F
http://www.nwnights.ru/redirect/applicationadvantage.com/?URL=www.betop24.com%2F
http://www.ut2.ru/redirect/hotyoga.co.nz/?URL=www.betop24.com%2F
http://bios.edu/?URL=boc-ks.com/speedbump.asp?link=www.betop24.com%2F
http://www.morrowind.ru/redirect/accord.ie/?URL=www.betop24.com%2F
http://ime.nu/s79457.gridserver.com/?URL=www.betop24.com%2F
http://2ch.io/morrowind.ru/redirect/www.betop24.com%2Ffeft/ref/xiswi/
https://smmry.com/centre.org.au/?URL=www.betop24.com%2F
http://ime.nu/pulaskiticketsandtours.com/?URL=www.betop24.com%2F
https://smmry.com/promoincendie.com/?URL=www.betop24.com%2Ffeft/ref/xiswi/
http://www.nwnights.ru/redirect/hs-events.nl/?URL=www.betop24.com%2F
http://www.ut2.ru/redirect/chivemediagroup.com/?URL=www.betop24.com%2Ffeft/ref/xiswi/
http://www.nwnights.ru/redirect/horizon-environ.com/?URL=www.betop24.com%2F
http://www.morrowind.ru/redirect/yesfest.com/?URL=www.betop24.com%2F
http://bios.edu/?URL=cssanz.org/?URL=www.betop24.com%2F
http://bios.edu/?URL=roserealty.com.au/?URL=www.betop24.com%2F
http://www.morrowind.ru/redirect/t.me/iv?url=www.betop24.com%2F
http://www.allods.net/redirect/boosterblog.com/vote-815901-624021.html?adresse=www.betop24.com%2F
http://www.ut2.ru/redirect/client.paltalk.com/client/webapp/client/External.wmt?url=www.betop24.com%2F
http://www.morrowind.ru/redirect/minecraft-galaxy.ru/redirect/?url=www.betop24.com%2F
http://www.nwnights.ru/redirect/rescuetheanimals.org/?URL=www.betop24.com%2F
http://ime.nu/foosball.com/?URL=www.betop24.com%2F
http://www.ut2.ru/redirect/supertramp.com/?URL=www.betop24.com%2F
http://www.allods.net/redirect/gmmdl.com/?URL=www.betop24.com%2Ffeft/ref/xiswi/
http://2ch.io/icecap.us/?URL=www.betop24.com%2Ffeft/ref/xiswi/
http://www.nwnights.ru/redirect/labassets.com/?URL=www.betop24.com%2F
https://cwcab.com/?URL=acceleweb.com/register?aw_site_id=www.betop24.com%2F
http://www.allods.net/redirect/cim.bg/?URL=www.betop24.com%2F
http://ime.nu/rawseafoods.com/?URL=www.betop24.com%2F
https://cwcab.com/?URL=judiisrael.com/?URL=www.betop24.com%2F
http://bios.edu/?URL=roserealty.com.au/?URL=www.betop24.com%2F
http://www.morrowind.ru/redirect/wdvstudios.be/?URL=www.betop24.com%2Ffeft/ref/xiswi/
https://jump.5ch.net/?frienddo.com/out.php?url=www.betop24.com%2F
https://jump.5ch.net/?ctlimo.com/?URL=www.betop24.com%2Ffeft/ref/xiswi/
http://www.ut2.ru/redirect/bvilpcc.com/?URL=www.betop24.com%2F
https://cwcab.com/?URL=reedring.com/?URL=www.betop24.com%2F
http://www.ut2.ru/redirect/lbaproperties.com/?URL=www.betop24.com%2Ffeft/ref/xiswi/
http://ime.nu/kingswelliesnursery.com/?URL=www.betop24.com%2F
https://smmry.com/miloc.hr/?URL=www.betop24.com%2F
https://smmry.com/nslgames.com/?URL=www.betop24.com%2F
http://bios.edu/?URL=thebigmo.nl/?URL=www.betop24.com%2F
http://www.nwnights.ru/redirect/steamcommunity.com/linkfilter/?url=www.betop24.com%2F
http://2ch.io/barrypopik.com/index.php?URL=www.betop24.com%2F
https://cwcab.com/?URL=batterybusiness.com.au/?URL=www.betop24.com%2F
https://smmry.com/blingguard.com/?URL=www.betop24.com%2Ffeft/ref/xiswi/
https://cwcab.com/?URL=professor-murmann.info/?URL=www.betop24.com%2F
http://www.allods.net/redirect/burkecounty-ga.gov/?URL=www.betop24.com%2F
http://2ch.io/2ch.io/www.betop24.com%2F
http://www.nwnights.ru/redirect/mikropul.com/?URL=www.betop24.com%2F
https://cwcab.com/?URL=bytecheck.com/results?resource=www.betop24.com%2F
http://www.nwnights.ru/redirect/youtube.com/redirect?q=www.betop24.com%2F
http://www.nwnights.ru/redirect/salonfranchise.com.au/?URL=www.betop24.com%2F
https://cwcab.com/?URL=couchsrvnation.com/?URL=www.betop24.com%2F
http://ime.nu/usich.gov/?URL=www.betop24.com%2F
http://www.nwnights.ru/redirect/sostrategic.com.au/?URL=www.betop24.com%2F
http://www.allods.net/redirect/boosterblog.net/vote-146-144.html?adresse=www.betop24.com%2F
http://www.allods.net/redirect/wilsonlearning.com/?URL=www.betop24.com%2F
http://ime.nu/nerida-oasis.com/?URL=www.betop24.com%2Ffeft/ref/xiswi/
http://2ch.io/aldonauto.com/?URL=www.betop24.com%2F
http://www.allods.net/redirect/spot-car.com/?URL=www.betop24.com%2F
http://ime.nu/hornbeckoffshore.com/?URL=www.betop24.com%2F
http://bios.edu/?URL=anzela.edu.au/?URL=www.betop24.com%2F
http://bios.edu/?URL=lbast.ru/zhg_img.php?url=www.betop24.com%2F
https://cwcab.com/?URL=dentalcommunity.com.au/?URL=www.betop24.com%2F
http://www.morrowind.ru/redirect/peter.murmann.name/?URL=www.betop24.com%2F
http://www.morrowind.ru/redirect/firma.hr/?URL=www.betop24.com%2F
https://jump.5ch.net/?labassets.com/?URL=www.betop24.com%2Ffeft/ref/xiswi/
http://ime.nu/progressprinciple.com/?URL=www.betop24.com%2F
http://www.nwnights.ru/redirect/mbcarolinas.org/?URL=www.betop24.com%2F
https://jump.5ch.net/?fotka.com/link.php?u=www.betop24.com%2F
http://bios.edu/?URL=restaurant-zahnacker.fr/?URL=www.betop24.com%2F
https://cwcab.com/?URL=accord.ie/?URL=www.betop24.com%2F
http://www.nwnights.ru/redirect/morrisparks.net/?URL=www.betop24.com%2F
https://cwcab.com/?URL=fishidy.com/go?url=www.betop24.com%2F
http://www.nwnights.ru/redirect/blingguard.com/?URL=www.betop24.com%2Ffeft/ref/xiswi/
http://www.ut2.ru/redirect/ria-mar.com/?URL=www.betop24.com%2F
https://jump.5ch.net/?batterybusiness.com.au/?URL=www.betop24.com%2F
http://bios.edu/?URL=ocmdhotels.com/?URL=www.betop24.com%2F
http://www.gta.ru/redirect/healthyeatingatschool.ca/?URL=www.betop24.com%2Ffeft/ref/xiswi/
https://cwcab.com/?URL=ntltyres.com.au/?URL=www.betop24.com%2F
https://jump.5ch.net/?romanodonatosrl.com/?URL=www.betop24.com%2F
http://www.morrowind.ru/redirect/pcrnv.com.au/?URL=www.betop24.com%2Ffeft/ref/xiswi/
http://bios.edu/?URL=wup.pl/?URL=www.betop24.com%2F
https://jump.5ch.net/?bluewatergrillri.com/?URL=www.betop24.com%2F
http://2ch.io/washburnvalley.org/?URL=www.betop24.com%2F
https://smmry.com/crspublicity.com.au/?URL=www.betop24.com%2F
http://www.allods.net/redirect/vectechnologies.com/?URL=www.betop24.com%2F
https://smmry.com/troxellwebdesign.com/?URL=www.betop24.com%2F
https://jump.5ch.net/?jacobberger.com/?URL=www.betop24.com%2Ffeft/ref/xiswi/
http://www.ut2.ru/redirect/oncreativity.tv/?URL=www.betop24.com%2F
http://www.allods.net/redirect/ww2.torahlab.org/?URL=www.betop24.com%2F
http://2ch.io/essencemusicagency.com/?URL=www.betop24.com%2F
http://www.ut2.ru/redirect/livingtrustplus.com/?URL=www.betop24.com%2F
https://cwcab.com/?URL=giruna.hu/redirect.php?url=www.betop24.com%2F
http://www.nwnights.ru/redirect/cssdrive.com/?URL=www.betop24.com%2Ffeft/ref/xiswi/
http://bios.edu/?URL=basebusiness.com.au/?URL=www.betop24.com%2F
http://www.allods.net/redirect/2ch.io/www.betop24.com%2F http://2ch.io/slrc.org/?URL=www.betop24.com%2F
http://www.nwnights.ru/redirect/life-church.com.au/?URL=www.betop24.com%2F
http://bios.edu/?URL=precisioncomponents.com.au/?URL=www.betop24.com%2F
http://www.nwnights.ru/redirect/okellymoylan.ie/?URL=www.betop24.com%2F
http://ime.nu/sc.hkexnews.hk/TuniS/www.betop24.com%2F
http://www.allods.net/redirect/davidcouperconsulting.com/?URL=www.betop24.com%2F
https://jump.5ch.net/?morrowind.ru/redirect/www.betop24.com%2F
http://www.morrowind.ru/redirect/ucrca.org/?URL=www.betop24.com%2F
https://jump.5ch.net/?wagyu.org/?URL=www.betop24.com%2F
https://jump.5ch.net/?unbridledbooks.com/?URL=www.betop24.com%2Ffeft/ref/xiswi/
http://www.allods.net/redirect/jongeriuslab.com/?URL=www.betop24.com%2F
https://smmry.com/woodforestcharitablefoundation.org/?URL=www.betop24.com%2F
www.waters.com/waters/downloadFile.htm?lid=134799103&id=134799102&fileName=Download&fileUrl=http%3A%2F%2Fwww.betop24.com%2F
www.bari91.com/tz.php?zone=Pacific/Niue&r=http%3A%2F%2Fwww.betop24.com%2F
www.xfdq123.com/url.aspx?url=http://www.betop24.com%2F
www.ignicaodigital.com.br/affiliate/?idev_id=270&u=http://www.betop24.com%2F
www.mirogled.com/banner-clicks/10?url=http://www.betop24.com%2F
www.haveanice.com/refer/D3dKFDIUbYk66eqlL1163PlAW3BXqx/jpg?hvAn_url=http://www.betop24.com%2F
www.avilas-style.com/shop/affiche.php?ad_id=132&from=&uri=www.betop24.com%2F
www.hentaicrack.com/cgi-bin/atx/out.cgi?s=95&u=http://www.betop24.com%2F/
www.voxlocalis.net/enlazar/?url=http://www.betop24.com%2F/
https://christchurchcitylibraries.com/Databases/GVRL/jumpto.asp?url=http%3A%2F%2Fwww.betop24.com%2F
www.ferrosystems.com/setLocale.jsp?language=en&url=http://www.betop24.com%2F
test.healinghealth.com/?wptouch_switch=desktop&redirect=http://www.betop24.com%2F/
www.lastdates.com/l/?www.betop24.com%2F
kaimono-navi.jp/rd?u=http%3A%2F%2Fwww.betop24.com%2F
www.shop-bell.com/out.php?id=kibocase&category=ladies&url=http://www.betop24.com%2F/
https://richmonkey.biz/go/?http://www.betop24.com%2F
https://online-knigi.com/site/getlitresurl?url=http%3A%2F%2Fwww.betop24.com%2F
akvaforum.no/go.cfml?id=1040&uri=http://www.betop24.com%2F
www.kamphuisgroep.nl/r.php?cid=2314&site=http://www.betop24.com%2F
sns.51.ca/link.php?url=http://www.betop24.com%2F
http://newsrankey.com/view.html?url=http://www.betop24.com%2F/
www.minibuggy.net/forum/redirect-to/?redirect=http://www.betop24.com%2F
www.wagersmart.com/top/out.cgi?id=bet2gold&url=http://www.betop24.com%2F/
www.global-autonews.com/shop/bannerhit.php?bn_id=307&url=http://www.betop24.com%2F/
pnevmopodveska-club.ru/index.php?app=core&module=system&controller=redirect&do=redirect&url=http://www.betop24.com%2F
www.darussalamciamis.or.id/redirect/?alamat=http%3A%2F%2Fwww.betop24.com%2F
agama.su/go.php?url=http://www.betop24.com%2F
bas-ip.ru/bitrix/rk.php?goto=http://www.betop24.com%2F
college.captainu.com/college_teams/1851/campaigns/51473/tracking/click?contact_id=1154110&email_id=1215036&url=http://www.betop24.com%2F/
www.oldfold.com/g?u=http://www.betop24.com%2F
www.anibox.org/go?http://www.betop24.com%2F
https://acejobs.net/jobclick/?RedirectURL=http%3A%2F%2Fwww.betop24.com%2F&Domain=acejobs.net
www.m.mobilegempak.com/wap_api/get_msisdn.php?URL=https://www.www.betop24.com%2F
sitesdeapostas.co.mz/track/odd?url-id=11&game-id=1334172&odd-type=draw&redirect=http://www.betop24.com%2F
www.autaabouracky.cz/plugins/guestbook/go.php?url=https://www.www.betop24.com%2F
www.escapers-zone.net/ucp.php?mode=logout&redirect=http%3A%2F%2Fwww.betop24.com%2F
http://canuckstuff.com/store/trigger.php?rlink=http://www.betop24.com%2F
members.practicegreenhealth.org/eweb/Logout.aspx?RedirectURL=http://www.betop24.com%2F/
http://fiinpro.com/Home/ChangeLanguage?lang=vi-VN&returnUrl=http://www.betop24.com%2F/
https://visit-thassos.com/index.php/language/en?redirect=http://www.betop24.com%2F
adserverv6.oberberg.net/adserver/www/delivery/ck.php?ct=1&oaparams=2bannerid=2zoneid=35cb=88915619faoadest=http://www.betop24.com%2F
www.homuta.co.jp/link/?link=http%3A%2F%2Fwww.betop24.com%2F
klvr.link/redirect/venividivici/spotify?linkUrl=http%3A%2F%2Fwww.betop24.com%2F
www.pcstore.com.tw/adm/act.htm?src=vipad_click&store_type=SUP_TOP&big_exh=STOREAD-%A7%E950&reurl=http%3A%2F%2Fwww.betop24.com%2F
http://baantawanchandao.com/change_language.asp?language_id=th&MemberSite_session=site_47694_&link=http://www.betop24.com%2F/
grannyfuck.in/cgi-bin/atc/out.cgi?id=139&u=http://www.betop24.com%2F
www.joserodriguez.info/?wptouch_switch=desktop&redirect=http://www.betop24.com%2F
https://navigraph.com/redirect.ashx?url=http://www.betop24.com%2F
edu54.ru/bitrix/redirect.php?goto=http://www.betop24.com%2F
https://honolulufestival.com/ja/?wptouch_switch=desktop&redirect=http%3A%2F%2Fwww.betop24.com%2F
www.perinosboilingpot.com/site.php?pageID=1&bannerID=19&vmoment=1430132758&url=http%3A%2F%2Fwww.betop24.com%2F
www.hardcoreoffice.com/tp/out.php?link=txt&url=https://www.www.betop24.com%2F
best.amateursecrets.net/cgi-bin/out.cgi?ses=onmfsqgs6c&id=318&url=http://www.betop24.com%2F/
elitesm.ru/bitrix/rk.php?id=102&site_id=ru&event1=banner&event2=click&event3=1+%2F+%5B102%5D+%5Bright_group_bot%5D+%DD%CA%CE+3%C4&goto=http://www.betop24.com%2F/
www.iasb.com/sso/login/?userToken=Token&returnURL=http://www.betop24.com%2F
prominentjobs.co.uk/jobclick/?RedirectURL=http%3A%2F%2Fwww.betop24.com%2F
http://workshopweekend.net/er?url=http://www.betop24.com%2F/
go.pnuna.com/go.php?url=http://www.betop24.com%2F
https://przyjazniseniorom.com/language/en/?returnUrl=http%3A%2F%2Fwww.betop24.com%2F
motorrad-stecki.de/trigger.php?r_link=http%3A%2F%2Fwww.betop24.com%2F
news.animravel.fr/retrolien.aspx?id_dest=1035193&id_envoi=463&url=www.betop24.com%2F
http://aldenfamilyonline.com/KathySite/MomsSite/MOM_SHARE_MEMORIES/msg_system/go.php?url=http://www.betop24.com%2F
www.sports-central.org/cgi-bin/axs/ax.pl?http://www.betop24.com%2F/
shop.mediaport.cz/redirect.php?action=url&goto=www.betop24.com%2F
adlogic.ru/?goto=jump&url=http://www.betop24.com%2F/
www.medicumlaude.de/index.php/links/index.php?url=http://www.betop24.com%2F
www.figurama.eu/cz/redirect.php?path=http://www.betop24.com%2F
nagranitse.ru/url.php?q=http://www.betop24.com%2F/
www.wien-girls.at/out-link?url=http://www.betop24.com%2F/
www.blackpictures.net/jcet/tiov.cgi?cvns=1&s=65&u=http://www.betop24.com%2F
https://athleticforum.biz/redirect/?to=http%3A%2F%2Fwww.betop24.com%2F
www.telehaber.com/redir.asp?url=http://www.betop24.com%2F
www.elmore.ru/go.php?to=http://www.betop24.com%2F
www.tido.al/vazhdo.php?url=http://www.betop24.com%2F/
os-company.ru/bitrix/redirect.php?goto=http://www.betop24.com%2F
duma-slog.ru/bitrix/redirect.php?event1=file&event2=www.betop24.com%2F&event3=29.01.2015_312_rd.doc&goto=http://www.betop24.com%2F/
www.interempresas.net/estadisticas/r.asp?idsector=129&e=221083&c=195&d=http://www.betop24.com%2F
pravoslavieru.trckmg.com/app/click/30289/561552041/?goto_url=www.betop24.com%2F
jump.fan-site.biz/rank.cgi?mode=link&id=342&url=http://www.betop24.com%2F/
http://anti-kapitalismus.org/sites/all/modules/pubdlcnt/pubdlcnt.php?file=http://www.betop24.com%2F&nid=435
rubyconnection.com.au/umbraco/newsletterstudio/tracking/trackclick.aspx?nid=207065033113056034011005043041220243180024215107&e=011204127253056232044128247253046214192002250116195220062107112232157159227010159247231011081075001197133136091194134170178051032155159001112047&url=http://www.betop24.com%2F/
video.childsheroes.com/Videos/SetCulture?culture=en-US&returnURL=http%3A%2F%2Fwww.betop24.com%2F
buecher-teneues.de/mlm/lm/lm.php?tk=CQkJRkRhdW1AdGVuZXVlcy5jb20JU3BlY2lhbCBPZmZlcnMgYmVpIHRlTmV1ZXMgCTM3CQkzNzQ1CWNsaWNrCXllcwlubw==&url=http://www.betop24.com%2F
karir.imslogistics.com/language/en?return=https://www.www.betop24.com%2F
yun.smartlib.cn/widgets/fulltext/?url=http://www.betop24.com%2F
www.okhba.org/clicks.php?bannerid=51&url=http%3A%2F%2Fwww.betop24.com%2F
www.all1.co.il/goto.php?url=https://www.www.betop24.com%2F
https://jipijapa.net/jobclick/?RedirectURL=http%3A%2F%2Fwww.betop24.com%2F&Domain=jipijapa.net&rgp_m=co3&et=4495
chaku.tv/i/rank/out.cgi?url=http://www.betop24.com%2F
www.mendocino.com/?id=4884&url=www.betop24.com%2F
doc.enervent.com/op/op.SetLanguage.php?lang=de_DE&referer=http%3A%2F%2Fwww.betop24.com%2F
www.feg-jena.de/link/?link=http://www.betop24.com%2F
b2b.psmlighting.be/en-GB/Base/ChangeCulture?currentculture=de-DE¤turl=http%3A%2F%2Fwww.betop24.com%2F¤turl=http%3A%2F%2Fbatmanapollo.ru
http://dstats.net/redir.php?url=http://www.betop24.com%2F/
crescent.netcetra.com/inventory/military/dfars/?saveme=MS51957-42*&redirect=http://www.betop24.com%2F/
r5.dir.bg/rem.php?word_id=0&place_id=9&ctype=mp&fromemail=&iid=3770&aid=4&cid=0&url=http://www.betop24.com%2F/
www.store-datacomp.eu/Home/ChangeLanguage?lang=en&returnUrl=http%3A%2F%2Fwww.betop24.com%2F
http://hotgrannyworld.com/cgi-bin/crtr/out.cgi?id=41&l=toplist&u=http://www.betop24.com%2F
t.wxb.com/order/sourceUrl/1894895?url=www.betop24.com%2F
shop.merchtable.com/users/authorize?return_url=http://www.betop24.com%2F/
zharpizza.ru/bitrix/rk.php?goto=http://www.betop24.com%2F
elinks.qp.land.to/link.php?url=http://www.betop24.com%2F
www.powerflexweb.com/centers_redirect_log.php?idDivision=25&nameDivision=Homepage&idModule=m551&nameModule=myStrength&idElement=298&nameElement=Provider%20Search&url=http://www.betop24.com%2F/
rbs-crm.ru/?redirect_url=http%3A%2F%2Fwww.betop24.com%2F
www.theukhighstreet.com/perl/jump.cgi?ID=12&URL=http://www.betop24.com%2F
ekonomka.dn.ua/out.php?link=http://www.www.betop24.com%2F
www.perpetuumsoft.com/Out.ashx?href=http://www.betop24.com%2F
digital.fijitimes.com/api/gateway.aspx?f=https://www.www.betop24.com%2F
https://amateurdorado.com/wp-content/plugins/AND-AntiBounce/redirector.php?url=http://www.betop24.com%2F
www.canakkaleaynalipazar.com/advertising.php?r=3&l=http://www.betop24.com%2F
www.tascher-de-la-pagerie.org/fr/liens.php?l=http://www.betop24.com%2F/
youngpussy.ws/out.php?http://www.betop24.com%2F
www.hartje.name/go?r=1193&jumpto=http://www.betop24.com%2F
www.yplf.com/cgi-bin/a2/out.cgi?id=141&l=toplist&u=http://www.betop24.com%2F
www.joblinkapply.com/Joblink/5972/Account/ChangeLanguage?lang=es-MX&returnUrl=http://www.betop24.com%2F
smedia.ru/bitrix/rk.php?goto=http://www.betop24.com%2F
navitrinu.ru/redirect/?go=http%3A%2F%2Fwww.betop24.com%2F
https://hatboroalive.com/abnrs/countguideclicks.cfm?targeturl=http%3A%2F%2Fwww.betop24.com%2F&businessid=29277
www.irrigationnz.co.nz/ClickThru?mk=5120.0&Redir=http://www.betop24.com%2F
api.buu.ac.th/getip/?url=http://www.betop24.com%2F
winehall.ru/bitrix/rk.php?goto=http://www.betop24.com%2F
efir-kazan.ru/bitrix/rk.php?id=367&site_id=s1&event1=banner&event2=click&event3=47+/367[Main_middle1]%D0%90%D0%BA%D0%91%D0%B0%D1%80%D1%81+%D0%A1%D0%BF%D0%BE%D1%80%D1%82&goto=http://www.betop24.com%2F/
yiwu.0579.com/jump.asp?url=http://www.betop24.com%2F
t.goadservices.com/optout?url=http://www.betop24.com%2F
auth.philpapers.org/login?service=http://www.betop24.com%2F
forsto.ru/bitrix/redirect.php?goto=http://www.betop24.com%2F
www.etslousberg.be/ViewSwitcher/SwitchView?mobile=False&returnUrl=http%3A%2F%2Fwww.betop24.com%2F
www.slavenibas.lv/bancp/www/delivery/ck.php?ct=1&oaparams=2bannerid=82zoneid=2cb=008ea50396oadest=http%3A%2F%2Fwww.betop24.com%2F
neso.r.niwepa.com/ts/i5536875/tsc?tst=!&amc=con.blbn.490450.485278.164924&pid=6508&rmd=3&trg=http://www.betop24.com%2F/
italiantrip.it/information_about_cookie_read.php?url=https%3A%2F%2Fwww.betop24.com%2F
cgi1.bellacoola.com/adios.cgi/630?http://www.betop24.com%2F/
adengine.old.rt.ru/go.jsp?to=http://www.betop24.com%2F
www.simpleet.lu/Home/ChangeCulture?lang=de-DE&returnUrl=http%3A%2F%2Fwww.betop24.com%2F
enseignants.flammarion.com/Banners_Click.cfm?ID=86&URL=www.betop24.com%2F
content.sixflags.com/news/director.aspx?gid=0&iid=72&cid=3714&link=http://www.betop24.com%2F/
www.offendorf.fr/spip_cookie.php?url=http://www.betop24.com%2F/
suche6.ch/count.php?url=http://www.betop24.com%2F/
shiftlink.ca/AbpLocalization/ChangeCulture?cultureName=de&returnUrl=http%3A%2F%2Fwww.betop24.com%2F
www.mir-stalkera.ru/go?http://www.betop24.com%2F
duhocphap.edu.vn/?wptouch_switch=desktop&redirect=http%3A%2F%2Fwww.betop24.com%2F
www.millerovo161.ru/go?http://www.betop24.com%2F/
www.naturaltranssexuals.com/cgi-bin/a2/out.cgi?id=97&l=toplist&u=http://www.betop24.com%2F
https://amanaimages.com/lsgate/?lstid=pM6b0jdQgVM-Y9ibFgTe6Zv1N0oD2nYuMA&lsurl=http://www.betop24.com%2F
planszowkiap.pl/trigger.php?r_link=http%3A%2F%2Fwww.betop24.com%2F
http://covenantpeoplesministry.org/cpm/wp/sermons/?show&url=http://www.betop24.com%2F/
www.sporteasy.net/redirect/?url=http://www.betop24.com%2F
getwimax.jp/st-manager/click/track?id=3894&type=raw&url=http://www.betop24.com%2F/&source_url=https://getwimax.jp/&source_title=GetWiMAX.jp%EF%BD%9CWiMAX%EF%BC%88%E3%83%AF%E3%82%A4%E3%83%9E%E3%83%83%E3%82%AF%E3%82%B9%EF%BC%89%E6%AF%94%E8%BC%83%EF%BC%81%E3%81%8A%E3%81%99%E3%81%99%E3%82%81%E3%83%97%E3%83%AD%E3%83%90%E3%82%A4%E3%83%803%E9%81%B8
mobo.osport.ee/Home/SetLang?lang=cs&returnUrl=http://www.betop24.com%2F
materinstvo.ru/forward?link=http%3A%2F%2Fwww.betop24.com%2F
http://computer-chess.org/lib/exe/fetch.php?media=http://www.betop24.com%2F
https://enchantedcottageshop.com/shop/trigger.php?r_link=http%3A%2F%2Fwww.betop24.com%2F
https://buist-keatch.org/sphider/include/click_counter.php?url=http%3A%2F%2Fwww.betop24.com%2F
www.mistress-and-slave.com/cgi-bin/out.cgi?id=123crush&url=http://www.betop24.com%2F/
rel.chubu-gu.ac.jp/soumokuji/cgi-bin/go.cgi?http://www.betop24.com%2F/
fallout3.ru/utils/ref.php?url=http://www.betop24.com%2F/
www.veloxbox.us/link/?h=http%3A%2F%2Fwww.betop24.com%2F
www.adult-plus.com/ys/rank.php?mode=link&id=592&url=http://www.betop24.com%2F/
mobilize.org.br/handlers/anuncioshandler.aspx?anuncio=55&canal=2&redirect=https://www.www.betop24.com%2F
sparktime.justclick.ru/lms/api-login/?hash=MO18szcRUQdzpT%2FrstSCW5K8Gz6ts1NvTJLVa34vf1A%3D&authBhvr=1&email=videotrend24%40mail.ru&expire=1585462818&lms%5BrememberMe%5D=1&targetPath=http%3A%2F%2Fwww.betop24.com%2F
redirect.icurerrors.com/http/www.betop24.com%2F
http://vcteens.com/cgi-bin/at3/out.cgi?trade=http://www.betop24.com%2F
www.bquest.org/Links/Redirect.aspx?ID=164&url=http://www.betop24.com%2F/
www.stipendije.info/phpAdsNew/adclick.php?bannerid=129&zoneid=1&source=&dest=http://www.betop24.com%2F/
today.od.ua/redirect.php?url=http://www.betop24.com%2F/
laskma.megastart-slot.ru/redirect/?g=http://www.betop24.com%2F
mightypeople.asia/link.php?id=M0ZGNHFISkd2bFh0RmlwSFU4bDN4QT09&destination=http://www.betop24.com%2F
www.chinaleatheroid.com/redirect.php?url=http://www.betop24.com%2F
i.s0580.cn/module/adsview/content/?action=click&bid=5&aid=163&url=http%3A%2F%2Fwww.betop24.com%2F&variable=&source=https%3A%2F%2Fcutepix.info%2Fsex%2Friley-reyes.php
jsv3.recruitics.com/redirect?rx_cid=506&rx_jobId=39569207&rx_url=http://www.betop24.com%2F/
https://jobinplanet.com/away?link=www.betop24.com%2F
www.supermoto8.com/sidebanner/62?href=http://www.betop24.com%2F
presse.toyota.dk/login.aspx?returnurl=http://www.betop24.com%2F/
www.uktrademarkregistration.co.uk/JumpTo.aspx?url=http://www.betop24.com%2F/
https://www.youtube.fr/redirect?q=www.betop24.com%2F
https://www.youtube.es/redirect?q=www.betop24.com%2F
https://www.youtube.jp/redirect?q=www.betop24.com%2F
https://www.youtube.co.uk/redirect?q=www.betop24.com%2F
https://www.youtube.ru/redirect?q=www.betop24.com%2F
https://www.youtube.pl/redirect?q=www.betop24.com%2F
https://www.youtube.gr/redirect?q=www.betop24.com%2F
https://www.youtube.nl/redirect?q=www.betop24.com%2F
https://www.youtube.ca/redirect?q=www.betop24.com%2F
https://www.youtube.cz/redirect?q=www.betop24.com%2F
https://www.youtube.com/redirect?q=www.betop24.com%2F&gl=AU
https://www.youtube.com.tw/redirect?q=www.betop24.com%2F
https://www.youtube.com/redirect?q=www.betop24.com%2F
http://www.youtube.de/redirect?event=channel_description&q=www.betop24.com%2F
http://www.youtube.com/redirect?event=channel_description&q=www.betop24.com%2F
http://www.youtube.co/redirect?event=channel_description&q=www.betop24.com%2F
http://www.youtube.es/redirect?event=channel_description&q=www.betop24.com%2F
http://www.youtube.ca/redirect?event=channel_description&q=www.betop24.com%2F
http://www.youtube.nl/redirect?event=channel_description&q=www.betop24.com%2F
http://www.youtube.pl/redirect?event=channel_description&q=www.betop24.com%2F
http://www.youtube.ch/redirect?event=channel_description&q=www.betop24.com%2F
http://www.youtube.be/redirect?event=channel_description&q=www.betop24.com%2F
http://www.youtube.se/redirect?event=channel_description&q=www.betop24.com%2F
http://www.youtube.dk/redirect?event=channel_description&q=www.betop24.com%2F
http://www.youtube.pt/redirect?event=channel_description&q=www.betop24.com%2F
http://www.youtube.no/redirect?event=channel_description&q=www.betop24.com%2F
http://www.youtube.gr/redirect?event=channel_description&q=www.betop24.com%2F
http://www.youtube.cl/redirect?event=channel_description&q=www.betop24.com%2F
http://www.youtube.at/redirect?event=channel_description&q=www.betop24.com%2F
http://www.youtube.bg/redirect?event=channel_description&q=www.betop24.com%2F
http://www.youtube.co.ke/redirect?event=channel_description&q=www.betop24.com%2F
http://www.youtube.co.cr/redirect?event=channel_description&q=www.betop24.com%2F
http://www.youtube.kz/redirect?event=channel_description&q=www.betop24.com%2F
http://www.youtube.cat/redirect?event=channel_description&q=www.betop24.com%2F
http://www.youtube.ge/redirect?event=channel_description&q=www.betop24.com%2F
https://www.youtube.com/redirect?event=channel_description&q=www.betop24.com%2F&gl=ml
https://www.youtube.com/redirect?q=www.betop24.com%2F&gl=DE
https://www.youtube.com/redirect?q=www.betop24.com%2F&gl=IT
https://www.youtube.com/redirect?q=https%3A%2F%2Fwww.betop24.com%2F&gl=AR
https://www.youtube.at/redirect?q=www.betop24.com%2F
https://www.youtube.ch/redirect?q=www.betop24.com%2F
http://www.youtube.sk/redirect?event=channel_description&q=www.betop24.com%2F
http://www.youtube.rs/redirect?event=channel_description&q=www.betop24.com%2F
http://www.youtube.lt/redirect?event=channel_description&q=www.betop24.com%2F
http://www.youtube.si/redirect?event=channel_description&q=www.betop24.com%2F
http://www.youtube.hr/redirect?event=channel_description&q=www.betop24.com%2F
http://www.youtube.ee/redirect?event=channel_description&q=www.betop24.com%2F
http://www.youtube.lu/redirect?event=channel_description&q=www.betop24.com%2F
http://www.youtube.tn/redirect?event=channel_description&q=www.betop24.com%2F
rtb-asiamax.tenmax.io/bid/click/1462922913409/e95f2c30-1706-11e6-a9b4-a9f6fe33c6df/3456/5332/?rUrl=http://www.betop24.com%2F
www.joeshouse.org/booking?link=http%3A%2F%2Fwww.betop24.com%2F&ID=1112
hanhphucgiadinh.vn/ext-click.php?url=http://www.betop24.com%2F/
www.gmwebsite.com/web/redirect.asp?url=http://www.betop24.com%2F
www.nymfer.dk/atc/out.cgi?s=60&l=topgallery&c=1&u=http://www.betop24.com%2F/
https://seocodereview.com/redirect.php?url=http://www.betop24.com%2F
swra.backagent.net/ext/rdr/?http%3A%2F%2Fwww.betop24.com%2F
namiotle.pl/?wptouch_switch=mobile&redirect=http://www.betop24.com%2F/
gfb.gameflier.com/func/actionRewriter.aspx?pro=http&url=www.betop24.com%2F
golfpark.jp/banner/counter.aspx?url=http://www.betop24.com%2F
www.mexicolore.co.uk/click.php?url=http://www.betop24.com%2F
www.tiersertal.com/clicks/uk_banner_click.php?url=http://www.betop24.com%2F/
www.invisalign-doctor.in/api/redirect?url=http://www.betop24.com%2F
www.isadatalab.com/redirect?clientId=ee5a64e1-3743-9b4c-d923-6e6d092ae409&appId=69&value=[EMV%20FIELD]EMAIL[EMV%20/FIELD]&cat=Techniques+culturales&url=http://www.betop24.com%2F/
www.immunallergo.ru/bitrix/redirect.php?goto=https://www.www.betop24.com%2F
dort.brontosaurus.cz/forum/go.php?url=http://www.betop24.com%2F/
eatart.dk/Home/ChangeCulture?lang=da&returnUrl=http%3A%2F%2Fwww.betop24.com%2F
trackingapp4.embluejet.com/Mod_Campaigns/tracking.asp?idem=31069343&em=larauz@untref.edu.ar&ca=73143&ci=0&me=72706&of=581028&adirecta=0&url=http://www.betop24.com%2F/
smils.ru/bitrix/redirect.php?goto=http://www.betop24.com%2F
www.hschina.net/ADClick.aspx?SiteID=206&ADID=1&URL=http://www.betop24.com%2F
cipresso.ru/bitrix/redirect.php?goto=http://www.betop24.com%2F
www.vacacionartravel.com/DTCSpot/public/banner_redirect.aspx?idca=286&ids=75665176&cp=167&idcli=0&ci=2&p=http://www.betop24.com%2F
www.mydosti.com/Advertisement/updateadvhits.aspx?adid=48&gourl=http://www.betop24.com%2F
www.malles-bertault.com/?change_langue=en&url=http%253a%252f%252fwww.betop24.com%2F
www.accesslocksmithatlantaga.com/?wptouch_switch=mobile&redirect=http%3A%2F%2Fwww.betop24.com%2F
www.beeicons.com/redirect.php?site=http://www.betop24.com%2F/
hirlevel.pte.hu/site/redirect?newsletter_id=UFV1UG5yZ3hOaWFyQVhvSUFoRmRQUT09&recipient=Y25zcm1ZaGxvR0xJMFNtNmhwdmpPNFlVSzlpS2c4ZnA1NzRPWjJKY3QrND0=&address=www.betop24.com%2F
www.cccowe.org/lang.php?lang=en&url=http://www.betop24.com%2F
www.mytokachi.jp/index.php?type=click&mode=sbm&code=2981&url=http://www.betop24.com%2F/
apiv2.nextgenshopping.com/api/click?domainName=www.betop24.com%2F&partnerTag4=portal&partnerTag2=coupon&clickId=A499UMEpK&partnerWebsiteKey=QatCO22&syncBack=true
www.wqketang.com/logout?goto=http://www.betop24.com%2F
access.bridges.com/externalRedirector.do?url=www.betop24.com%2F
www.cossa.ru/bitrix/redirect.php?event1=click&event2=&event3=&goto=http://www.betop24.com%2F
bot.buymeapie.com/recipe?url=http://www.betop24.com%2F/
www.frodida.org/BannerClick.php?BannerID=29&LocationURL=http://www.betop24.com%2F
extremaduraempresarial.juntaex.es/cs/c/document_library/find_file_entry?p_l_id=47702&noSuchEntryRedirect=http%3A%2F%2Fwww.betop24.com%2F
www.quanmama.com/t/goto.aspx?url=http://www.betop24.com%2F
quartiernetz-friesenberg.ch/links-go.php?to=http://www.betop24.com%2F
www.maultalk.com/url.php?to=http://www.betop24.com%2F
www.infohakodate.com/ps/ps_search.cgi?act=jump&url=http://www.betop24.com%2F
www.e-expo.net/category/click_url.html?url=http://www.betop24.com%2F
www.chitaitext.ru/bitrix/redirect.php?event1=utw&event2=utw1&event3=&goto=http://www.betop24.com%2F
www.realsubliminal.com/newsletter/t/c/11098198/c?dest=http%3A%2F%2Fwww.betop24.com%2F
http://getdatasheet.com/url.php?url=http://www.betop24.com%2F
www.rechnungswesen-portal.de/bitrix/redirect.php?event1=KD37107&event2=https2F/www.universal-music.de2880%25-100%25)(m/w/d)&goto=http://www.betop24.com%2F
https://kirei-style.info/st-manager/click/track?id=7643&type=raw&url=http%3A%2F%2Fwww.betop24.com%2F
www.aldolarcher.com/tools/esstat/esdown.asp?File=http%3A%2F%2Fwww.betop24.com%2F
embed.gabrielny.com/embedlink?key=f12cc3d5-e680-47b0-8914-a6ce19556f96&width=100%25&height=1200&division=bridal&nochat=1&domain=http%3A%2F%2Fwww.betop24.com%2F
cs.payeasy.com.tw/click?url=http://www.betop24.com%2F
http://blackwhitepleasure.com/cgi-bin/atx/out.cgi?trade=http://www.betop24.com%2F
www.knet-web.net/m/pRedirect.php?uID=2&iID=259&iURL=http%3A%2F%2Fwww.betop24.com%2F
azlan.techdata.com/InTouch/GUIBnrT3/BnrTrackerPublic.aspx?CountryCode=18&BannerLangCulture=nl-nl&URL=http://www.betop24.com%2F/&Target=2&BannerId=41919&Zoneid=281&Parameters=&cos=Azlan
holmss.lv/bancp/www/delivery/ck.php?ct=1&oaparams=2bannerid=44zoneid=1cb=7743e8d201oadest=http%3A%2F%2Fwww.betop24.com%2F
www.mintmail.biz/track/clicks/v2/?messageid=1427&cid=54657&url=http://www.betop24.com%2F
www.lzmfjj.com/Go.asp?URL=http://www.betop24.com%2F/
http://alexmovs.com/cgi-bin/atx/out.cgi?id=148&tag=topatx&trade=http://www.betop24.com%2F
marciatravessoni.com.br/revive/www/delivery/ck.php?ct=1&oaparams=2bannerid=40zoneid=16cb=fc1d72225coadest=http://www.betop24.com%2F/
psylive.ru/success.aspx?id=0&goto=www.betop24.com%2F
https://sohodiffusion.com/mod/mod_langue.asp?action=francais&url=http://www.betop24.com%2F
www.mybunnies.net/te3/out.php?u=http://www.betop24.com%2F/
lubaczowskie.pl/rdir/?l=http%3A%2F%2Fwww.betop24.com%2F&lid=1315
www.kowaisite.com/bin/out.cgi?id=kyouhuna&url=http://www.betop24.com%2F/
www.ieslaasuncion.org/enlacesbuscar/clicsenlaces.asp?Idenlace=411&url=http://www.betop24.com%2F/
www.168web.com.tw/in/front/bin/adsclick.phtml?Nbr=114_02&URL=http://www.betop24.com%2F
http://tswzjs.com/go.asp?url=http://www.betop24.com%2F/
asstomouth.guru/out.php?url=http://www.betop24.com%2F
advtest.exibart.com/adv/adv.php?id_banner=7201&link=http%3A%2F%2Fwww.betop24.com%2F
https://thairesidents.com/l.php?b=85&p=2,5&l=http%3A%2F%2Fwww.betop24.com%2F
www.latestnigeriannews.com/link_channel.php?channel=http%3A%2F%2Fwww.betop24.com%2F
www.haogaoyao.com/proad/default.aspx?url=www.betop24.com%2F
globalmedia51.ru/bitrix/redirect.php?goto=http://www.betop24.com%2F
citysafari.nl/Home/setCulture?language=en&returnUrl=http%3A%2F%2Fwww.betop24.com%2F
es.catholic.net/ligas/ligasframe.phtml?liga=http://www.betop24.com%2F
https://slashwrestling.com/cgi-bin/redirect.cgi?http://www.betop24.com%2F
lorena-kuhni.kz/redirect?link=www.betop24.com%2F
www.webshoptrustmark.fr/Change/en?returnUrl=http%3A%2F%2Fwww.betop24.com%2F
www.wave24.net/cgi-bin/linkrank/out.cgi?id=106248&cg=1&url=www.betop24.com%2F
www.tssweb.co.jp/?wptouch_switch=mobile&redirect=http%253a%252f%252fwww.betop24.com%2F
e.ourger.com/?c=scene&a=link&id=47154371&url=http://www.betop24.com%2F/
zh-hk.guitarians.com/home/redirect/ubid/1015?r=http%3A%2F%2Fwww.betop24.com%2F
www.mastercleaningsupply.com/trigger.php?r_link=http%3A%2F%2Fwww.betop24.com%2F
www.cumshoter.com/cgi-bin/at3/out.cgi?id=98&tag=top&trade=http://www.betop24.com%2F
shp.hu/hpc_uj/click.php?ml=5&url=https://www.www.betop24.com%2F
lrnews.ru/xgo.php?url=www.betop24.com%2F
https://indonesianmma.com/modules/mod_jw_srfr/redir.php?url=http://www.betop24.com%2F
www.dive-international.net/places/redirect.php?b=797&web=www.www.betop24.com%2F
www.themza.com/redirect.php?r=www.betop24.com%2F
lambda.ecommzone.com/lz/srr/00as0z/06e397d17325825ee6006c3c5ee495f922/actions/redirect.aspx?url=http://www.betop24.com%2F
v.wcj.dns4.cn/?c=scene&a=link&id=8833621&url=http://www.betop24.com%2F/
spb-medcom.ru/redirect.php?http://www.betop24.com%2F
forest.ru/links.php?go=http%3A%2F%2Fwww.betop24.com%2F
reefcentral.ru/bitrix/rk.php?goto=http://www.betop24.com%2F
bsau.ru/bitrix/redirect.php?event1=news_out&event2=2Fiblock9CB0%D1D0D0D0%B0BB87B0%D1D1D0D1%82B5%D1D0%B8B0%D0D1D0D1%81828C+2.pdf&goto=http://www.betop24.com%2F
https://trackdaytoday.com/redirect-out?url=http%3A%2F%2Fwww.betop24.com%2F
空の最安値.travel.jp/smart/pc.asp?url=http://www.betop24.com%2F/
https://bigjobslittlejobs.com/jobclick/?RedirectURL=http%3A%2F%2Fwww.betop24.com%2F&Domain=bigjobslittlejobs.com&rgp_m=title23&et=4495
ekovjesnik.hr/ads/www/delivery/ck.php?ct=1&oaparams=2bannerid=4zoneid=4cb=68dbdae1d1oadest=http%3A%2F%2Fwww.betop24.com%2F
www.obertaeva.com/include/get.php?go=http://www.betop24.com%2F
https://studiohire.com/admin-web-tel-process.php?memberid=4638&indentifier=weburl&websitelinkhitnumber=7&telnumberhitnumber=0&websiteurl=http://www.betop24.com%2F
www.bobclubsau.com/cmshome/WebsiteAuditor/6744?url=www.betop24.com%2F
www.moonbbs.com/dm/dmlink.php?dmurl=http://www.betop24.com%2F
www.sparetimeteaching.dk/forward.php?link=http://www.betop24.com%2F
https://paspn.net/default.asp?p=90&gmaction=40&linkid=52&linkurl=http://www.betop24.com%2F
www.dialogportal.com/Services/Forward.aspx?link=http://www.betop24.com%2F
www.poddebiczak.pl/?action=set-desktop&url=http%3A%2F%2Fwww.betop24.com%2F
ant53.ru/file/link.php?url=http://www.betop24.com%2F
www.docin.com/jsp_cn/mobile/tip/android_v1.jsp?forward=http://www.betop24.com%2F
old.magictower.ru/cgi-bin/redir/redir.pl?http://www.betop24.com%2F/
go.flx1.com/click?id=1&m=11&pl=113&dmcm=16782&euid=16603484876&out=http://www.betop24.com%2F
moba-hgh.de/link2http.php?href=www.betop24.com%2F
https://gazetablic.com/ads/www/delivery/ck.php?ct=1&oaparams=2bannerid=34zoneid=26cb=0e0dfef92boadest=http%3A%2F%2Fwww.betop24.com%2F
www.genderpsychology.com/http/www.betop24.com%2F
http://chtbl.com/track/118167/www.betop24.com%2F
www.360wichita.com/amp-banner-tracking?adid=192059&url=http%3A%2F%2Fwww.betop24.com%2F
www.tsv-bad-blankenburg.de/cms/page/mod/url/url.php?eid=16&urlpf=www.betop24.com%2F
https://fishki.net/click?http://www.betop24.com%2F
https://zerlong.com/bitrix/redirect.php?goto=http://www.betop24.com%2F
catalog.flexcom.ru/go?z=36047&i=55&u=http://www.betop24.com%2F
www.wellvit.nl/response/forward/c1e41491e30c5af3c20f80a2af44e440.php?link=0&target=http%3A%2F%2Fwww.betop24.com%2F
www.ident.de/adserver/www/delivery/ck.php?ct=1&oaparams=2bannerid=76zoneid=2cb=8a18c95a9eoadest=http%3A%2F%2Fwww.betop24.com%2F
www.widgetinfo.net/read.php?sym=FRA_LM&url=http%3A%2F%2Fwww.betop24.com%2F
www.sdam-snimu.ru/redirect.php?url=http://www.betop24.com%2F
school.mosreg.ru/soc/moderation/abuse.aspx?link=http://www.betop24.com%2F
affiliates.kanojotoys.com/affiliate/scripts/click.php?a_aid=widdi77&desturl=http%3A%2F%2Fwww.betop24.com%2F
www.gotomypctech.com/affiliates/scripts/click.php?a_aid=ed983915&a_bid=&desturl=http://www.betop24.com%2F
www.my-sms.ru/ViewSwitcher/SwitchView?mobile=False&returnUrl=http%3A%2F%2Fwww.betop24.com%2F&rel=external
shop-uk.fmworld.com/Queue/Index?url=http://www.betop24.com%2F
nieuws.rvent.nl/bitmailer/statistics/mailstatclick/42261?link=http://www.betop24.com%2F
https://jobanticipation.com/jobclick/?RedirectURL=http%3A%2F%2Fwww.betop24.com%2F&Domain=jobanticipation.com
www.xsbaseball.com/tracker/index.html?t=ad&pool_id=3&ad_id=5&url=http://www.betop24.com%2F
eos.ru/bitrix/redirect.php?goto=http://www.betop24.com%2F
b.sm.su/click.php?bannerid=56&zoneid=10&source=&dest=http://www.betop24.com%2F/
https://processon.com/setting/locale?language=zh&back=http%3A%2F%2Fwww.betop24.com%2F
c.yam.com/srh/wsh/r.c?http://www.betop24.com%2F
www.jolletorget.no/J/l.php?l=http%3A%2F%2Fwww.betop24.com%2F
watchvideo.co/go.php?url=http://www.betop24.com%2F
www.todoku.info/gpt/rank.cgi?mode=link&id=29649&url=http://www.betop24.com%2F
www.fotochki.com/redirect.php?go=www.betop24.com%2F
bayerwald.tips/plugins/bannerverwaltung/bannerredirect.php?bannerid=1&url=http%3A%2F%2Fwww.betop24.com%2F
belantara.or.id/lang/s/ID?url=http://www.betop24.com%2F/
ele-market.ru/consumer.php?url=http://www.betop24.com%2F
https://www.хорошие-сайты.рф/r.php?r=http://www.betop24.com%2F/
https://jobsflagger.com/jobclick/?RedirectURL=http%3A%2F%2Fwww.betop24.com%2F
https://gpoltava.com/away/?go=www.betop24.com%2F
www.cardexchange.com/index.php/tools/packages/tony_mailing_list/services/?mode=link&mlm=62&mlu=0&u=2&url=http%3A%2F%2Fwww.betop24.com%2F
services.nfpa.org/Authentication/GetSSOSession.aspx?return=http://www.betop24.com%2F/
http://spaceup.org/?wptouch_switch=mobile&redirect=http://www.betop24.com%2F
yarko-zhivi.ru/redirect?url=http://www.betop24.com%2F
https://kekeeimpex.com/Home/ChangeCurrency?urls=http%3A%2F%2Fwww.betop24.com%2F&cCode=GBP&cRate=77.86247
mycounter.com.ua/go.php?http://www.betop24.com%2F
l2base.su/go?http://www.betop24.com%2F
www.duomodicagliari.it/reg_link.php?link_ext=http%3A%2F%2Fwww.betop24.com%2F&prov=1
assine.hostnet.com.br/cadastro/?rep=17&url=http://www.betop24.com%2F/
www.dvnlp.de/profile/gruppe/redirect/5?url=www.betop24.com%2F
www.smkn5pontianak.sch.id/redirect/?alamat=http://www.betop24.com%2F
www.cheapdealuk.co.uk/go.php?url=http://www.betop24.com%2F/
bilometro.brksedu.com.br/tracking?url=www.betop24.com%2F&zorigem=hotsite-blackfriday
tramplintk.ru/bitrix/redirect.php?goto=http://www.betop24.com%2F
cnc.extranet.gencat.cat/treball_cnc/AppJava/FileDownload.do?pdf=http%3A%2F%2Fwww.betop24.com%2F&codi_cnv=9998045
www.gamecollections.co.uk/search/redirect.php?retailer=127&deeplink=http://www.betop24.com%2F
bearcong.no1.sexy/hobby-delicious/rank.cgi?mode=link&id=19&url=http://www.betop24.com%2F/
www.omschweiz.ch/select-your-country?publicUrl=http%3A%2F%2Fwww.betop24.com%2F
https://anacolle.net/?wptouch_switch=desktop&redirect=http%3A%2F%2Fwww.betop24.com%2F
www.cubamusic.com/Home/ChangeLanguage?lang=es-ES&returnUrl=http%3A%2F%2Fwww.betop24.com%2F
expoclub.ru/bitrix/redirect.php?goto=http://www.betop24.com%2F
365sekretov.ru/redirect.php?action=url&goto=www.betop24.com%2F%20
www.sgdrivingtest.com/redirect.php?page=www.betop24.com%2F
www.jxren.com/news/link/link.asp?id=7&url=http://www.betop24.com%2F
particularcareers.co.uk/jobclick/?RedirectURL=http%3A%2F%2Fwww.betop24.com%2F
https://bsaonline.com/MunicipalDirectory/SelectUnit?unitId=411&returnUrl=http%3A%2F%2Fwww.betop24.com%2F&sitetransition=true
www.elit-apartament.ru/go?http://www.betop24.com%2F
sendai.japansf.net/rank.cgi?mode=link&id=1216&url=http%3A%2F%2Fwww.betop24.com%2F
www.bmwfanatics.ru/goto.php?l=http://www.betop24.com%2F
www.saabsportugal.com/forum/index.php?thememode=full;redirect=http://www.betop24.com%2F
www.interecm.com/interecm/tracker?op=click&id=5204.db2&url=http://www.betop24.com%2F/
cms.sive.it/Jump.aspx?gotourl=http%3A%2F%2Fwww.betop24.com%2F
largusladaclub.ru/go/url=https:/www.betop24.com%2F
https://bethlehem-alive.com/abnrs/countguideclicks.cfm?targeturl=http%3A%2F%2Fwww.betop24.com%2F&businessid=29579
www.bookmark-favoriten.com/?goto=http://www.betop24.com%2F
shop.yuliyababich.eu/RU/ViewSwitcher/SwitchView?mobile=False&returnUrl=http%3A%2F%2Fwww.betop24.com%2F
www.depmode.com/go.php?http://www.betop24.com%2F
https://urgankardesler.com/anasayfa/yonlen?link=http%3A%2F%2Fwww.betop24.com%2F
www.metalindex.ru/netcat/modules/redir/?&site=http://www.betop24.com%2F/
www.rprofi.ru/bitrix/redirect.php?goto=http://www.betop24.com%2F
https://darudar.org/external/?link=http://www.betop24.com%2F
www.postsabuy.com/autopost4/page/generate/?link=http%3A%2F%2Fwww.betop24.com%2F&list=PL9d7lAncfCDSkF4UPyhzO59Uh8cOoD-8q&fb_node=942812362464093&picture&name=%E0%B9%82%E0%B8%9B%E0%B8%A3%E0%B9%81%E0%B8%81%E0%B8%A3%E0%B8%A1%E0%B9%82%E0%B8%9E%E0%B8%AA%E0%B8%82%E0%B8%B2%E0%B8%A2%E0%B8%AA%E0%B8%B4%E0%B8%99%E0%B8%84%E0%B9%89%E0%B8%B2%E0%B8%AD%E0%B8%AD%E0%B8%99%E0%B9%84%E0%B8%A5%E0%B8%99%E0%B9%8C+&caption=%E0%B9%80%E0%B8%A5%E0%B8%82%E0%B8%B2%E0%B8%AA%E0%B9%88%E0%B8%A7%E0%B8%99%E0%B8%95%E0%B8%B1%E0%B8%A7+%E0%B8%97%E0%B8%B5%E0%B8%84%E0%B8%B8%E0%B8%93%E0%B8%A5%E0%B8%B7%E0%B8%A1%E0%B9%84%E0%B8%A1%E0%B9%88%E0%B8%A5%E0%B8%87+Line+%40postsabuy&description=%E0%B8%A3%E0%B8%B2%E0%B8%84%E0%B8%B2%E0%B8%96%E0%B8%B9%E0%B8%81%E0%B8%97%E0%B8%B5%E0%B9%88%E0%B8%AA%E0%B8%B8%E0%B8%94%E0%B9%83%E0%B8%99+3+%E0%B9%82%E0%B8%A5%E0%B8%81+%E0%B8%AD%E0%B8%B4%E0%B8%AD%E0%B8%B4
www.perimeter.org/track.pdf?url=http%3A%2F%2Fwww.betop24.com%2F
forum.darievna.ru/go.php?http://www.betop24.com%2F
techlab.rarus.ru/bitrix/rk.php?goto=http://www.betop24.com%2F
www.spiritualforums.com/vb/redir.php?link=http://www.betop24.com%2F
www.review-mag.com/cdn/www/delivery/view.php?ct=1&oaparams=2bannerid=268zoneid=1cb=8c1317f219oadest=http%3A%2F%2Fwww.betop24.com%2F
polo-v1.feathr.co/v1/analytics/crumb?flvr=email_link_click&rdr=http://www.betop24.com%2F/
sintesi.provincia.mantova.it/portale/LinkClick.aspx?link=http://www.betop24.com%2F
www.surinenglish.com/backend/conectar.php?url=http://www.betop24.com%2F
www.reference-cannabis.com/interface/sortie.php?adresse=http://www.betop24.com%2F/
login.0×69416d.co.uk/sso/logout?tenantId=tnl&gotoUrl=http%3A%2F%2Fwww.betop24.com%2F&domain=0×69416d.co.uk
blog.link-usa.jp/emi?wptouch_switch=mobile&redirect=http://www.betop24.com%2F/
http://damki.net/go/?http://www.betop24.com%2F
opensesame.wellymulia.zaxaa.com/b/66851136?s=1&redir=http://www.betop24.com%2F
ism3.infinityprosports.com/ismdata/2009100601/std-sitebuilder/sites/200901/www/en/tracker/index.html?t=ad&pool_id=1&ad_id=112&url=http://www.betop24.com%2F
www.winxuan.com/page/cps/eqifacookieinterface.jsp?from=yiqifa&wid=8&url=http://www.betop24.com%2F
www.packmage.net/uc/goto/?url=http://www.betop24.com%2F
my.9991.com/login_for_index_0327.php?action=logout&forward=http://www.betop24.com%2F/
www.sdchamber.biz/admin/mod_newsletter/redirect.aspx?message_id=986&redirect=http://www.betop24.com%2F
www.naturum.co.jp/ad/linkshare/?siteID=p_L785d6UQY-V4Fh4Rxs7wNzOPgtzv95Tg&lsurl=http%3A%2F%2Fwww.betop24.com%2F
www.d-e-a.eu/newsletter/redirect.php?link=http://www.betop24.com%2F
www.bom.ai/goweburl?go=http%3A%2F%2Fwww.betop24.com%2F
enews2.sfera.net/newsletter/redirect.php?id=sabricattani@gmail.com_0000006566_144&link=http://www.betop24.com%2F/
underwater.com.au/redirect_url/id/7509/?redirect=http://www.betop24.com%2F
https://eqsoftwares.com/languages/setlanguage?languagesign=en&redirect=http://www.betop24.com%2F
www.jagdambasarees.com/Home/ChangeCurrency?urls=http%3A%2F%2Fwww.betop24.com%2F&cCode=MYR&cRate=14.554
www.priegeltje.nl/gastenboek/go.php?url=http://www.betop24.com%2F
www.serie-a.ru/bitrix/redirect.php?goto=http://www.betop24.com%2F
www.rz114.cn/url.html?url=http://www.betop24.com%2F/
www.greatdealsindia.com/redirects/infibeam.aspx?url=http://www.betop24.com%2F
rs.345kei.net/rank.php?id=37&mode=link&url=http://www.betop24.com%2F/
webapp.jgz.la/?c=scene&a=link&id=8665466&url=http://www.betop24.com%2F
www.erotiikkalelut.com/url.php?link=http://www.betop24.com%2F
https://vse-doski.com/redirect/?go=http://www.betop24.com%2F
www.karatetournaments.net/link7.asp?LRURL=http://www.betop24.com%2F/&LRTYP=O
simracing.su/go/?http://www.betop24.com%2F
click.cheshi.com/go.php?proid=218&clickid=1393306648&url=http://www.betop24.com%2F/
fdeam.finanzen-partnerprogramm.de/tracking/?as_id=9257&c_id=595&url=http://www.betop24.com%2F
flypoet.toptenticketing.com/index.php?url=http://www.betop24.com%2F
www.horsesmouth.com/LinkTrack.aspx?u=http://www.betop24.com%2F
d-click.fiemg.com.br/u/18081/131/75411/137_0/82cb7/?url=http://www.betop24.com%2F/
vicsport.com.au/analytics/outbound?url=http://www.betop24.com%2F/
https://fachowiec.com/zliczanie-bannera?id=24&url=http://www.betop24.com%2F
ieea.ir/includes/change_lang.php?lang=en&goto=http://www.betop24.com%2F/
scribe.mmonline.io/click?evt_nm=Clicked+Registration+Completion&evt_typ=clickEmail&app_id=m4marry&eml_sub=Registration+Successful&usr_did=4348702&cpg_sc=NA&cpg_md=email&cpg_nm=&cpg_cnt=&cpg_tm=NA&link_txt=Live+Chat&em_type=Notification&url=http://www.betop24.com%2F
apps.cancaonova.com/ads/www/delivery/ck.php?ct=1&oaparams=2bannerid=149zoneid=20cb=87d2c6208doadest=http%3A%2F%2Fwww.betop24.com%2F
www.lespritjardin.be/?advp_click_bimage_id=19&url=http%253a%252f%252fwww.betop24.com%2F&shortcode_id=10
www.dresscircle-net.com/psr/rank.cgi?mode=link&id=14&url=http://www.betop24.com%2F
www.counterwelt.com/charts/click.php?user=14137&link=http://www.betop24.com%2F/
fms.csonlineschool.com.au/changecurrency/1?returnurl=http://www.betop24.com%2F
www.v-archive.ru/bitrix/rk.php?goto=http://www.betop24.com%2F
www.jagat.co.jp/analysis/analysis.php?url=http%3A%2F%2Fwww.betop24.com%2F
https://careerchivy.com/jobclick/?RedirectURL=http%3A%2F%2Fwww.betop24.com%2F
shinsekai.type.org/?wptouch_switch=desktop&redirect=http://www.betop24.com%2F/
https://maned.com/scripts/lm/lm.php?tk=CQkJZWNuZXdzQGluZm90b2RheS5jb20JW05ld3NdIE1FSSBBbm5vdW5jZXMgUGFydG5lcnNoaXAgV2l0aCBUd2l4bCBNZWRpYQkxNjcyCVBSIE1lZGlhIENvbnRhY3RzCTI1OQljbGljawl5ZXMJbm8=&url=http://www.betop24.com%2F
www.etaigou.com/turn2.php?ad_id=276&link=http://www.betop24.com%2F
www.asensetranslations.com/modules/babel/redirect.php?newlang=en_US&newurl=http://www.betop24.com%2F/
gameshock.jeez.jp/rank.cgi?mode=link&id=307&url=http://www.betop24.com%2F/
https://tripyar.com/go.php?http://www.betop24.com%2F/
www.floridafilmofficeinc.com/?goto=http://www.betop24.com%2F/
tsvc1.teachiworld.com/bin/checker?mode=4&module=11&mailidx=19130&dmidx=0&emidx=0&service=0&cidx=&etime=20120328060000&seqidx=3&objidx=22&encoding=0&url=http://www.betop24.com%2F/
rechner.atikon.at/lbg.at/newsletter/linktracking?subscriber=&delivery=38116&url=http%3A%2F%2Fwww.betop24.com%2F
www.pcreducator.com/Common/SSO.aspx?returnUrl=http://www.betop24.com%2F/
go.eniro.dk/lg/ni/cat-2611/http:/www.betop24.com%2F
www.fisherly.com/redirect?type=website&ref=listing_detail&url=http://www.betop24.com%2F
fid.com.ua/redirect/?go=http://www.betop24.com%2F/
www.modernipanelak.cz/?b=618282165&redirect=http://www.betop24.com%2F/
h5.hbifeng.com/index.php?c=scene&a=link&id=14240604&url=http://www.betop24.com%2F
www.bkdc.ru/bitrix/redirect.php?event1=news_out&event2=32reg.roszdravnadzor.ru/&event3=A0A0B5A09180D0%A09582A0BBA1A085%D0E2A084D0D1C2D0%A085+A0A0B5A182B0A0%C2D0D0D096+A1A0BBA0B180D0%A09795+A0A0B0A09582A1%D1D0D0D0A182B5+A0A091A08695A0%D1D0A6A185A0A085%D0D1D0D082A1A085%D0D0D1D0A095B1A0%C2D0D0D091&goto=http://www.betop24.com%2F/
record.affiliatelounge.com/WS-jvV39_rv4IdwksK4s0mNd7ZgqdRLk/7/?deeplink=http://www.betop24.com%2F/
d-click.artenaescola.org.br/u/3806/290/32826/1416_0/53052/?url=http://www.betop24.com%2F
www.morroccoaffiliate.com/aff.php?id=883&url=http://www.betop24.com%2F/
www.omegon.eu/de/?r=http%3A%2F%2Fwww.betop24.com%2F
www.flooble.com/cgi-bin/clicker.pl?id=grabbadl&url=http://www.betop24.com%2F/
www.sportsbook.ag/ctr/acctmgt/pl/openLink.ctr?ctrPage=http://www.betop24.com%2F/
www.quantixtickets3.com/php-bin-8/kill_session_and_redirect.php?redirect=http://www.betop24.com%2F/
www.photokonkurs.com/cgi-bin/out.cgi?url=http://www.betop24.com%2F/
crewroom.alpa.org/SAFETY/LinkClick.aspx?link=http://www.betop24.com%2F/&mid=12872
www.tagirov.org/out.php?url=www.betop24.com%2F
startlist.club/MSF/Language/Set?languageIsoCode=en&returnUrl=http%3A%2F%2Fwww.betop24.com%2F
www.tetsumania.net/search/rank.cgi?mode=link&id=947&url=http://www.betop24.com%2F/
https://imperial-info.net/link?idp=125&url=http://www.betop24.com%2F
www.brainlanguage-sa.com/setcookie.php?lang=en&file=http://www.betop24.com%2F/
www.ra2d.com/directory/redirect.asp?id=596&url=http://www.betop24.com%2F/
www.anorexicnudes.net/cgi-bin/atc/out.cgi?u=http://www.betop24.com%2F
www.zjjiajiao.com.cn/ad/adredir.asp?url=http://www.betop24.com%2F/
https://hakobo.com/wp/?wptouch_switch=desktop&redirect=http%3A%2F%2Fwww.betop24.com%2F
t.agrantsem.com/tt.aspx?cus=216&eid=1&p=216-2-71016b553a1fa2c9.3b14d1d7ea8d5f86&d=http://www.betop24.com%2F/
moscowdesignmuseum.ru/bitrix/rk.php?goto=http://www.betop24.com%2F
www.cheek.co.jp/location/location.php?id=keibaseminar&url=http://www.betop24.com%2F
https://www.nbda.org/?URL=www.betop24.com%2F
https://www.kichink.com/home/issafari?uri=http://www.betop24.com%2F/
https://blogranking.fc2.com/out.php?id=1032500&url=http://www.betop24.com%2F/
5cfxm.hxrs6.servertrust.com/v/affiliate/setCookie.asp?catId=1180&return=http://www.betop24.com%2F/
http://3dcreature.com/cgi-bin/at3/out.cgi?id=187&trade=http://www.betop24.com%2F/
https://nowlifestyle.com/redir.php?k=9a4e080456dabe5eebc8863cde7b1b48&url=https://www.www.betop24.com%2F
http://slipknot1.info/go.php?url=https://www.www.betop24.com%2F
www.acutenet.co.jp/cgi-bin/lcount/lcounter.cgi?link=https://www.www.betop24.com%2F
http://news-matome.com/method.php?method=1&url=http://www.betop24.com%2F/
https://lifecollection.top/site/gourl?url=https://www.www.betop24.com%2F
https://www.toscanapiu.com/web/lang.php?lang=DEU&oldlang=ENG&url=http%3A%2F%2Fwww.www.betop24.com%2F
speakrus.ru/links.php?go=http://www.betop24.com%2F/
https://edcommunity.ru/bitrix/rk.php?goto=http://www.betop24.com%2F/
https://www.gudarjavalambre.com/sections/miscelany/link.php?url=https://www.www.betop24.com%2F
akademik.tkyd.org/Home/SetCulture?culture=en-US&returnUrl=https://www.www.betop24.com%2F
www.interracialhall.com/cgi-bin/atx/out.cgi?trade=https://www.www.betop24.com%2F
https://www.tumimusic.com/link.php?url=http://www.betop24.com%2F/
www.glorioustronics.com/redirect.php?link=http://www.betop24.com%2F/
mail.resen.gov.mk/redir.hsp?url=http://www.betop24.com%2F/
https://www.pompengids.net/followlink.php?id=495&link=https://www.www.betop24.com%2F
https://www.hirforras.net/scripts/redir.php?url=http://www.betop24.com%2F/
https://lens-club.ru/link?go=http://www.betop24.com%2F/
https://www.thislife.net/cgi-bin/webcams/out.cgi?id=playgirl&url=http://www.betop24.com%2F/
https://www.dans-web.nu/klick.php?url=http://www.betop24.com%2F/
https://voobrajulya.ru/bitrix/redirect.php?goto=https://www.www.betop24.com%2F
www.resnichka.ru/partner/go.php?https://www.www.betop24.com%2F
www.nafta-him.com/bitrix/redirect.php?goto=http://www.betop24.com%2F/
www.kyoto-osaka.com/search/rank.cgi?mode=link&id=9143&url=http://www.betop24.com%2F/
https://t.raptorsmartadvisor.com/.lty?url=http://www.betop24.com%2F/&loyalty_id=14481&member_id=b01bbee6-4592-4345-a0ee-5d71ed6f1929
https://evoautoshop.com/?wptouch_switch=mobile&redirect=http%3A%2F%2Fwww.www.betop24.com%2F
http://beautycottageshop.com/change.php?lang=cn&url=http://www.betop24.com%2F/
http://1000love.net/lovelove/link.php?url=http://www.betop24.com%2F/
https://www.contactlenshouse.com/currency.asp?c=CAD&r=http%3A%2F%2Fwww.www.betop24.com%2F
https://www.bartaz.lt/wp-content/plugins/clikstats/ck.php?Ck_id=70&Ck_lnk=https://www.www.betop24.com%2F
https://sogo.i2i.jp/link_go.php?url=http://www.betop24.com%2F/
https://ceb.bg/catalog/statistic/?id=61&location=http%3A%2F%2Fwww.www.betop24.com%2F
https://malehealth.ie/redirect/?age=40&part=waist&illness=obesity&refer=http://www.betop24.com%2F/
https://magicode.me/affiliate/go?url=http://www.betop24.com%2F/
www.findingfarm.com/redir?url=http://www.betop24.com%2F/
www.fairpoint.net/~jensen1242/gbook/go.php?url=http://www.betop24.com%2F/
www.cnainterpreta.it/redirect.asp?url=https://www.www.betop24.com%2F
red.ribbon.to/~zkcsearch/zkc-search/rank.cgi?mode=link&id=156&url=http://www.betop24.com%2F/
iphoneapp.impact.co.th/i/r.php?u=https://www.www.betop24.com%2F
www.rencai8.com/web/jump_to_ad_url.php?id=642&url=http://www.betop24.com%2F/
http://crackstv.com/redirect.php?bnn=CabeceraHyundai&url=https://www.www.betop24.com%2F
https://www.baby22.com.tw/Web/turn.php?ad_id=160&link=http%3A%2F%2Fwww.www.betop24.com%2F
forum.marillion.com/forum/index.php?thememode=full;redirect=https://www.www.betop24.com%2F
https://jobregistry.net/jobclick/?RedirectURL=http%3A%2F%2Fwww.www.betop24.com%2F&Domain=jobregistry.net&rgp_m=title13&et=4495
https://www.goatzz.com/adredirect.aspx?adType=SiteAd&ItemID=9595&ReturnURL=http://www.betop24.com%2F/
https://www.emiratesvoice.com/footer/comment_like_dislike_ajax/?code=like&commentid=127&redirect=http://www.betop24.com%2F/
https://envios.uces.edu.ar/control/click.mod.php?idenvio=8147&email=gramariani@gmail.com&url=http://www.www.betop24.com%2F
https://www.rexart.com/cgi-rexart/al/affiliates.cgi?aid=872&redirect=http://www.betop24.com%2F
https://ulfishing.ru/forum/go.php?http://www.betop24.com%2F
https://www.ab-search.com/rank.cgi?mode=link&id=107&url=http://www.betop24.com%2F/
https://www.studyrama.be/tracking.php?origine=ficheform5683&lien=http://www.www.betop24.com%2F
https://experts.richdadworld.com/assets/shared/php/noabp.php?oaparams=2bannerid=664zoneid=5cb=0902f987cboadest=http%3A%2F%2Fwww.betop24.com%2F
https://miyagi.lawyer-search.tv/details/linkchk.aspx?type=o&url=http://www.betop24.com%2F/
https://www.snwebcastcenter.com/event/page/count_download_time.php?url=http://www.betop24.com%2F/
https://go.onelink.me/v1xd?pid=Patch&c=Mobile%20Footer&af_web_dp=https%3A%2F%2Fwww.betop24.com%2F
https://www.weather.net/cgi-bin/redir?http://www.betop24.com%2F
https://www.luckyplants.com/cgi-bin/toplist/out.cgi?id=rmontero&url=http://www.betop24.com%2F
https://webankety.cz/dalsi.aspx?site=http://www.betop24.com%2F
https://runkeeper.com/apps/authorize?redirect_uri=http://www.betop24.com%2F
https://gaggedtop.com/cgi-bin/top/out.cgi?ses=sBZFnVYGjF&id=206&url=http://www.betop24.com%2F/
https://e-bike-test.net/wp-content/plugins/AND-AntiBounce/redirector.php?url=http://www.betop24.com%2F/
https://www.morhipo.com/shared/partnercookie?k=gort&url=http://www.betop24.com%2F
https://www.shop-bell.com/out.php?id=kibocase&category=ladies&url=http://www.betop24.com%2F/
https://m.addthis.com/live/redirect/?url=http://www.betop24.com%2F/
https://www.pcreducator.com/Common/SSO.aspx?returnUrl=http://www.betop24.com%2F/
https://horsesmouth.com/LinkTrack.aspx?u=http://www.betop24.com%2F/
https://soom.cz/projects/get2mail/redir.php?id=c2e52da9ad&url=http://www.betop24.com%2F/
https://www.iaai.com/VehicleInspection/InspectionProvidersUrl?name=AA%20Transit%20Pros%20Inspection%20Service&url=http://www.betop24.com%2F/
https://shop.merchtable.com/users/authorize?return_url=http://www.betop24.com%2F/
https://nudewwedivas.forumcommunity.net/m/ext.php?url=http://www.betop24.com%2F/
https://cztt.ru/redir.php?url=http://www.betop24.com%2F/
https://ageoutloud.gms.sg/visit.php?item=54&uri=http://www.betop24.com%2F/
https://multimedia.inrap.fr/redirect.php?li=287&R=http://www.betop24.com%2F/
https://games4ever.3dn.ru/go?http://www.betop24.com%2F/
https://de.reasonable.shop/SetCurrency.aspx?currency=CNY&returnurl=http://www.betop24.com%2F/
https://www.pcbheaven.com/forum/index.php?thememode=full;redirect=http://www.betop24.com%2F/
https://mobo.osport.ee/Home/SetLang?lang=cs&returnUrl=http://www.betop24.com%2F/
https://www.rechnungswesen-portal.de/bitrix/redirect.php?event1=KD37107&event2=https2F/www.universal-music.de2880%25-100%25)(m/w/d)&goto=http://www.betop24.com%2F/
https://monarchbeachmembers.play18.com/ViewSwitcher/SwitchView?mobile=False&returnUrl=http://www.betop24.com%2F/
https://wayi.com.tw/wayi_center.aspx?flag=banner&url=http://www.betop24.com%2F/&idno=443
https://d.agkn.com/pixel/2389/?che=2979434297&col=22204979,1565515,238211572,435508400,111277757&l1=http://www.betop24.com%2F/
https://mobilize.org.br/handlers/anuncioshandler.aspx?anuncio=55&canal=2&redirect=http://www.betop24.com%2F/
https://bemidji.bigdealsmedia.net/include/sort.php?return_url=http://www.betop24.com%2F/&sort=a:3:{s:9:%E2%80%9Ddirection%E2%80%9D;s:3:%E2%80%9DASC%E2%80%9D;s:5:%E2%80%9Dfield%E2%80%9D;s:15:%E2%80%9DItems.PriceList%E2%80%9D;s:5:%E2%80%9Dlabel%E2%80%9D;s:9:%E2%80%9Dvalue_asc%E2%80%9D;}
https://www.dodeley.com/?action=show_ad&url=http://www.betop24.com%2F/
https://www.ignicaodigital.com.br/affiliate/?idev_id=270&u=http://www.betop24.com%2F/
https://www.cheerunion.org/tracker/index.html?t=ad&pool_id=2&ad_id=5&url=http://www.betop24.com%2F/
https://area51.to/go/out.php?s=100&l=site&u=http://www.betop24.com%2F/
http://mientaynet.com/advclick.php?o=textlink&u=15&l=http://www.betop24.com%2F/
https://atlanticleague.com/tracker/index.html?t=ad&pool_id=11&ad_id=5&url=http://www.betop24.com%2F/
https://www.castellodivezio.it/lingua.php?lingua=EN&url=http://www.betop24.com%2F/
https://www.podcastone.com/site/rd?satype=40&said=4&aaid=email&camid=-4999600036534929178&url=http://www.betop24.com%2F/
https://hslda.org/content/a/LinkTracker.aspx?id=4015475&appeal=385&package=36&uri=http://www.betop24.com%2F/
https://www.sign-in-china.com/newsletter/statistics.php?type=mail2url&bs=88&i=114854&url=http://www.betop24.com%2F/
https://panarmenian.net/eng/tofv?tourl=http://www.betop24.com%2F/
https://www.kvinfo.dk/visit.php?linkType=2&linkValue=http://www.betop24.com%2F/
https://lavoro.provincia.como.it/portale/LinkClick.aspx?link=http://www.betop24.com%2F/&mid=935
https://sete.gr/app_plugins/newsletterstudio/pages/tracking/trackclick.aspx?nid=218221206039243162226109144018149003034132019130&e=000220142174231130224127060133189018075115154134&url=http://www.betop24.com%2F/
https://ovatu.com/e/c?url=http://www.betop24.com%2F/
https://primepartners.globalprime.com/afs/wcome.php?c=427|0|1&e=GP204519&url=http://www.betop24.com%2F/
https://ltp.org/home/setlocale?locale=en&returnUrl=http://www.betop24.com%2F/
https://www.morhipo.com/shared/partnercookie?k=gort&url=http://www.betop24.com%2F/
https://cingjing.fun-taiwan.com/AdRedirector.aspx?padid=303&target=http://www.betop24.com%2F/
https://rtb-asiamax.tenmax.io/bid/click/1462922913409/e95f2c30-1706-11e6-a9b4-a9f6fe33c6df/3456/5332/?rUrl=http://www.betop24.com%2F/
https://campaign.unitwise.com/click?emid=31452&emsid=ee720b9f-a315-47ce-9552-fd5ee4c1c5fa&url=http://www.betop24.com%2F/
https://m.17ll.com/apply/tourl/?url=http://www.betop24.com%2F/
https://crewroom.alpa.org/SAFETY/LinkClick.aspx?link=http://www.betop24.com%2F/&mid=12872
https://rs.businesscommunity.it/snap.php?u=http://www.betop24.com%2F/
https://www.aiac.world/pdf/October-December2015Issue?pdf_url=http://www.betop24.com%2F/
https://union.591.com.tw/stats/event/redirect?e=eyJpdiI6IjdUd1B5Z2FPTmNWQzBmZk1LblR2R0E9PSIsInZhbHVlIjoiQTI4TnVKMzdjMkxrUjcrSWlkcXdzbjRQeGRtZ0ZGbXdNSWxkSkVieENwNjQ1cHF5aDZmWmFobU92ZGVyUk5jRTlxVnI2TG5pb0dJVHZSUUlHcXFTbGo3UDliYWU5UE5MSjlMY0xOQnFmbVRQSFNoZDRGd2dqVDZXZEU4WFoyajJ0S0JITlQ2XC9SXC9jRklPekdmcnFGb09vRllqNHVtTHlYT284ZmN3d0ozOHFkclRYYnU5UlY2NTFXSGRheW5SbGxJb3BmYjQ2Mm9TWUFCTEJuXC9iT25nYkg4QXpOd2pHVlBWTWxWXC91aWRQMVhKQmVJXC9qMW9IdlZaVVlBdWlCYW4rS0JualhSMElFeVZYN3NnUW1qcUdxcWUrSlFROFhKbWttdkdvMUJ3aWVRa2I3MVV5TXpER3doa2ZuekFWNWd3OGpuQ1VSczFDREVKaklaUks0TTRIY2pUeXYrQmdZYUFTK1F4RWpTY0RRaW5Nc0krdVJ2N2VUT1wvSUxVVWVKN3hnQU92QmlCbjQyMUpRdTZKVWJcL0RCSVFOcWl0azl4V2pBazBHWmVhdWptZGREVXh0VkRNWWxkQmFSYXhBRmZtMHA5dTlxMzIzQzBVaWRKMEFqSG0wbGkxME01RDBaaElTaU5QKzIxbSswaUltS0FYSzViZlFmZjZ1XC9Yclg0U2VKdHFCc0pTNndcL09FWklUdjlQM2dcL2RuN0szQ3plWmcyYWdpQzJDQ2NIcWROczVua3dIM1Q3OXpJY3Z0XC93MVk3SHUyODZHU3Z5aHFVbWEwRFU1ZFdyMGt0YWpsb3BkQitsZER5aWk4YWMrZWYzSFNHNERhOGdDeUJWeEtoSm9wQ0hQb2EzOHZ3eHFGVTQ2Mk1QSEZERzlXZWxRRTJldjJkdnZUM0ZwaW1KcEVVc3ZXWjRHaTZWRDJOK0YxR3d4bXhMR3BhWmZBNkJ6eUYxQjR4ODVxc0d0YkFpYU8yZ2tuWGdzelBpU3dFUjJVYUVtYUlpZllSUTVpaHZMbjhySFp4VEpQR3EyYnRLTmdcLzRvKzQwRmtGNUdWWnQ0VjFpcTNPc0JubEdWenFiajRLRFg5a2dRZFJOZ1wvaUEwVHR3ZnYzakpYVmVtT294aFk1TXBUZ3FmVnF2dnNSVWJ5VEE0WGZpV3o3Y0k2SjJhM2RDK2hoQ0FvV2YrSW9QWnhuZG5QN1hlOEFaTVZrcFZ3c0pXVHhtNkRTUkpmenpibG8zdTM0cGF6Q3oxTEJsdDdiOUgwWXFOUkNHWjlEbTBFYzdIRUcyalYrcW4wYklFbnlGYlZJUG00R1VDQTZLZEVJRklIbFVNZFdpS3RkeCt5bVpTNUkrOXE3dDlxWmZ2bitjSGlSeE9wZTg5Yk9wS0V6N1wvd1EzUTNVenNtbjlvQUJhdGsxMzNkZTdjTU1LNkd4THJMYTBGUEJ4elEycFNTNGZabEJnalhJc0pYZit1c1wvWDBzSm1JMzRad3F3PT0iLCJtYWMiOiI4MjNhNDJlYTMwOTlmY2VlYzgxNmU1N2JiM2QzODk5YjI5MDFhYThhMDBkYzNhODljOTRmMTMzMzk0YTM5OGIzIn0=&source=BANNER.2913&url=http://www.betop24.com%2F/
https://fdeam.finanzen-partnerprogramm.de/tracking/?as_id=9257&c_id=595&url=http://www.betop24.com%2F/
https://pdcn.co/e/http://www.betop24.com%2F/
https://api.webconnex.com/v1/postmaster/track/click/4f8036d14ee545798599c8921fbfcd22/db005310dba511e89fb606f49a4ee876?url=http://www.betop24.com%2F/
https://www.lecake.com/stat/goto.php?url=http://www.betop24.com%2F/
https://www.im-harz.com/counter/counter.php?url=http://www.betop24.com%2F/
https://pzz.to/click?uid=8571&target_url=http://www.betop24.com%2F/
https://www.swipeclock.com/sc/cookie.asp?sitealias=79419397&redirect=http://www.betop24.com%2F/
https://home.uceusa.com/Redirect.aspx?r=http://www.betop24.com%2F/
https://www.seankenney.com/include/jump.php?num=http://www.betop24.com%2F/
https://runningcheese.com/go?url=http://www.betop24.com%2F/
http://chtbl.com/track/118167/http://www.betop24.com%2F/
https://www.ricacorp.com/Ricapih09/redirect.aspx?link=http://www.betop24.com%2F/
https://www.tremblant.ca/Shared/LanguageSwitcher/ChangeCulture?culture=en&url=http://www.betop24.com%2F/
https://www.winxuan.com/page/cps/eqifacookieinterface.jsp?from=yiqifa&wid=8&url=http://www.betop24.com%2F/
https://moscowdesignmuseum.ru/bitrix/rk.php?goto=http://www.betop24.com%2F/
https://igert2011.videohall.com/to_client?target=http://www.betop24.com%2F/
https://hr.pecom.ru/bitrix/rk.php?goto=http://www.betop24.com%2F/
https://cse.google.ru/url?q=http://www.betop24.com%2F/
https://jump2.bdimg.com/mo/q/checkurl?url=http://www.betop24.com%2F/
https://severeweather.wmo.int/cgi-bin/goto?where=http://www.betop24.com%2F/
https://beam.jpn.org/rank.cgi?mode=link&url=http://www.betop24.com%2F/
https://cse.google.com/url?sa=t&url=http://www.betop24.com%2F/
https://blogranking.fc2.com/out.php?id=414788&url=http://www.betop24.com%2F/
https://wtk.db.com/777554543598768/optout?redirect=http://www.betop24.com%2F/
https://community.rsa.com/t5/custom/page/page-id/ExternalRedirect?url=http://www.betop24.com%2F
https://click.alamode.com/?adcode=CPEMAQM0913_1&url=http://www.betop24.com%2F/
https://images.google.sk/url?q=http://www.betop24.com%2F/
https://images.google.gr/url?q=http://www.betop24.com%2F/
https://click.alamode.com/?adcode=CPEMAQM0913_1&url=http://www.betop24.com%2F/%20
https://images.google.lv/url?sa=t&url=http://www.betop24.com%2F
https://m.caijing.com.cn/member/logout?referer=http://www.betop24.com%2F/
https://jamesattorney.agilecrm.com/click?u=http://www.betop24.com%2F/
https://wikimapia.org/external_link?url=http://www.betop24.com%2F/
https://rsv.nta.co.jp/affiliate/set/af100101.aspx?site_id=66108024&redi_url=http://www.betop24.com%2F/
https://adengine.old.rt.ru/go.jsp?to=http://www.betop24.com%2F/
https://thediplomat.com/ads/books/ad.php?i=4&r=http://www.betop24.com%2F/
https://mitsui-shopping-park.com/lalaport/iwata/redirect.html?url=http://www.betop24.com%2F/
https://toolbarqueries.google.lt/url?q=http%3A%2F%2Fwww.betop24.com%2F
https://www.google.co.jp/url?q=http://www.betop24.com%2F/
https://images.google.de/url?q=http://www.betop24.com%2F/
https://images.google.bg/url?sa=t&url=http://www.betop24.com%2F
https://maps.google.ca/url?q=http://www.betop24.com%2F/
https://za.zalo.me/v3/verifyv2/pc?token=OcNsmjfpL0XY2F3BtHzNRs4A-hhQ5q5sPXtbk3O&continue=http://www.betop24.com%2F/%20
https://cse.google.cz/url?q=http://www.betop24.com%2F/
https://scanmail.trustwave.com/?c=8510&d=4qa02KqxZJadHuhFUvy7ZCUfI_2L10yeH0EeBz7FGQ&u=http://www.betop24.com%2F/
https://images.google.co.kr/url?sa=t&url=http://www.betop24.com%2F
https://images.google.ki/url?q=http://www.betop24.com%2F/
https://images.google.ga/url?q=http://www.betop24.com%2F/
https://maps.google.nu/url?q=http%3A%2F%2Fwww.betop24.com%2F
https://maps.google.bt/url?q=http://www.betop24.com%2F/
https://cse.google.bt/url?sa=i&url=http%3A%2F%2Fwww.www.betop24.com%2F
https://cse.google.bt/url?q=http%3A%2F%2Fwww.betop24.com%2F
https://images.google.bt/url?q=http%3A%2F%2Fwww.www.betop24.com%2F
https://images.google.bt/url?q=https%3A%2F%2Fwww.www.betop24.com%2F
https://www.google.bt/url?q=http%3A%2F%2Fwww.www.betop24.com%2F
https://ditu.google.com/url?q=http%3A%2F%2Fwww.www.betop24.com%2F
https://ditu.google.com/url?q=http%3A%2F%2Fwww.betop24.com%2F
https://asia.google.com/url?q=http%3A%2F%2Fwww.www.betop24.com%2F
https://clients1.google.de/url?q=http://www.betop24.com%2F
https://clients1.google.es/url?q=http://www.betop24.com%2F
https://clients1.google.co.uk/url?q=http://www.betop24.com%2F
https://clients1.google.co.jp/url?q=http://www.betop24.com%2F
https://clients1.google.fr/url?q=http://www.betop24.com%2F
https://clients1.google.it/url?q=http://www.betop24.com%2F
https://clients1.google.com.br/url?q=http://www.betop24.com%2F
https://clients1.google.co.in/url?q=http://www.betop24.com%2F
https://clients1.google.ca/url?q=http://www.betop24.com%2F
https://clients1.google.ru/url?q=http://www.betop24.com%2F
https://clients1.google.com.hk/url?q=http://www.betop24.com%2F
https://clients1.google.com.au/url?q=http://www.betop24.com%2F
https://clients1.google.co.id/url?q=http://www.betop24.com%2F
https://clients1.google.nl/url?q=http://www.betop24.com%2F
https://clients1.google.com.tw/url?q=http://www.betop24.com%2F
https://clients1.google.pl/url?q=http://www.betop24.com%2F
https://clients1.google.be/url?q=http://www.betop24.com%2F
https://clients1.google.co.th/url?q=http://www.betop24.com%2F
https://clients1.google.at/url?q=http://www.betop24.com%2F
https://clients1.google.cz/url?q=http://www.betop24.com%2F
https://clients1.google.se/url?q=http://www.betop24.com%2F
https://clients1.google.com.mx/url?q=http://www.betop24.com%2F
https://clients1.google.ch/url?q=http://www.betop24.com%2F
https://clients1.google.com.vn/url?q=http://www.betop24.com%2F
https://clients1.google.pt/url?q=http://www.betop24.com%2F
https://clients1.google.com.ua/url?q=http://www.betop24.com%2F
https://clients1.google.com.tr/url?q=http://www.betop24.com%2F
https://clients1.google.ro/url?q=http://www.betop24.com%2F
https://clients1.google.com.my/url?q=http://www.betop24.com%2F
https://clients1.google.gr/url?q=http://www.betop24.com%2F
https://clients1.google.dk/url?q=http://www.betop24.com%2F
https://clients1.google.hu/url?q=http://www.betop24.com%2F
https://clients1.google.com.ar/url?q=http://www.betop24.com%2F
https://clients1.google.fi/url?q=http://www.betop24.com%2F
https://clients1.google.co.il/url?q=http://www.betop24.com%2F
https://clients1.google.co.nz/url?q=http://www.betop24.com%2F
https://clients1.google.co.za/url?q=http://www.betop24.com%2F
https://clients1.google.cl/url?q=http://www.betop24.com%2F
https://clients1.google.com.co/url?q=http://www.betop24.com%2F
https://clients1.google.com.sg/url?q=http://www.betop24.com%2F
https://clients1.google.ie/url?q=http://www.betop24.com%2F
https://clients1.google.sk/url?q=http://www.betop24.com%2F
https://clients1.google.co.kr/url?q=http://www.betop24.com%2F
https://clients1.google.com.ph/url?q=http://www.betop24.com%2F
https://clients1.google.no/url?q=http://www.betop24.com%2F
https://clients1.google.lt/url?q=http://www.betop24.com%2F
https://clients1.google.bg/url?q=http://www.betop24.com%2F
https://clients1.google.com.sa/url?q=http://www.betop24.com%2F
https://clients1.google.hr/url?q=http://www.betop24.com%2F
https://clients1.google.com.pe/url?q=http://www.betop24.com%2F
https://clients1.google.ae/url?q=http://www.betop24.com%2F
https://clients1.google.co.ve/url?q=http://www.betop24.com%2F
https://clients1.google.ee/url?q=http://www.betop24.com%2F
https://clients1.google.com.pk/url?q=http://www.betop24.com%2F
https://clients1.google.rs/url?q=http://www.betop24.com%2F
https://clients1.google.com.eg/url?q=http://www.betop24.com%2F
https://clients1.google.si/url?q=http://www.betop24.com%2F
https://clients1.google.com.ec/url?q=http://www.betop24.com%2F
https://clients1.google.com.qa/url?q=http://www.betop24.com%2F
https://clients1.google.com.pr/url?q=http://www.betop24.com%2F
https://clients1.google.mu/url?q=http://www.betop24.com%2F
https://clients1.google.li/url?q=http://www.betop24.com%2F
https://clients1.google.lv/url?q=http://www.betop24.com%2F
https://clients1.google.mn/url?q=http://www.betop24.com%2F
https://clients1.google.com.gt/url?q=http://www.betop24.com%2F
https://clients1.google.co.cr/url?q=http://www.betop24.com%2F
https://clients1.google.com.uy/url?q=http://www.betop24.com%2F
https://clients1.google.lu/url?q=http://www.betop24.com%2F
https://clients1.google.ba/url?q=http://www.betop24.com%2F
https://clients1.google.is/url?q=http://www.betop24.com%2F
https://clients1.google.dz/url?q=http://www.betop24.com%2F
https://clients1.google.kg/url?q=http://www.betop24.com%2F
https://clients1.google.co.ke/url?q=http://www.betop24.com%2F
https://clients1.google.az/url?q=http://www.betop24.com%2F
https://clients1.google.com.ng/url?q=http://www.betop24.com%2F
https://clients1.google.com.np/url?q=http://www.betop24.com%2F
https://clients1.google.com.mt/url?q=http://www.betop24.com%2F
https://clients1.google.bi/url?q=http://www.betop24.com%2F
https://clients1.google.by/url?q=http://www.betop24.com%2F
https://clients1.google.com.bd/url?q=http://www.betop24.com%2F
https://clients1.google.as/url?q=http://www.betop24.com%2F
https://clients1.google.com.do/url?q=http://www.betop24.com%2F
https://clients1.google.kz/url?q=http://www.betop24.com%2F
https://clients1.google.co.ma/url?q=http://www.betop24.com%2F
https://clients1.google.jo/url?q=http://www.betop24.com%2F
https://clients1.google.lk/url?q=http://www.betop24.com%2F
https://clients1.google.com.cu/url?q=http://www.betop24.com%2F
https://clients1.google.com.ai/url?q=http://www.betop24.com%2F
https://clients1.google.com.gi/url?q=http://www.betop24.com%2F
https://clients1.google.cf/url?q=http://www.betop24.com%2F
https://clients1.google.com.ni/url?q=http://www.betop24.com%2F
https://clients1.google.md/url?q=http://www.betop24.com%2F
https://clients1.google.mg/url?q=http://www.betop24.com%2F
https://clients1.google.la/url?q=http://www.betop24.com%2F
https://clients1.google.com.jm/url?q=http://www.betop24.com%2F
https://clients1.google.com.vc/url?q=http://www.betop24.com%2F
https://clients1.google.com.tj/url?q=http://www.betop24.com%2F
https://clients1.google.com.cy/url?q=http://www.betop24.com%2F
https://clients1.google.com.sv/url?q=http://www.betop24.com%2F
https://clients1.google.rw/url?q=http://www.betop24.com%2F
https://clients1.google.com.om/url?q=http://www.betop24.com%2F
https://clients1.google.ps/url?q=http://www.betop24.com%2F
https://clients1.google.com.bo/url?q=http://www.betop24.com%2F
https://clients1.google.tk/url?q=http://www.betop24.com%2F
https://clients1.google.co.mz/url?q=http://www.betop24.com%2F
https://clients1.google.bs/url?q=http://www.betop24.com%2F
https://clients1.google.mk/url?q=http://www.betop24.com%2F
https://clients1.google.co.bw/url?q=http://www.betop24.com%2F
https://clients1.google.al/url?q=http://www.betop24.com%2F
https://clients1.google.sm/url?q=http://www.betop24.com%2F
https://clients1.google.co.zw/url?q=http://www.betop24.com%2F
https://clients1.google.tm/url?q=http://www.betop24.com%2F
https://clients1.google.com.bh/url?q=http://www.betop24.com%2F
https://clients1.google.com.af/url?q=http://www.betop24.com%2F
https://clients1.google.com.fj/url?q=http://www.betop24.com%2F
https://clients1.google.com.kh/url?q=http://www.betop24.com%2F
https://clients1.google.cg/url?q=http://www.betop24.com%2F
https://clients1.google.ki/url?q=http://www.betop24.com%2F
https://clients1.google.mw/url?q=http://www.betop24.com%2F
https://clients1.google.com.kw/url?q=http://www.betop24.com%2F
https://clients1.google.bf/url?q=http://www.betop24.com%2F
https://clients1.google.com.lb/url?q=http://www.betop24.com%2F
https://clients1.google.co.ls/url?q=http://www.betop24.com%2F
https://clients1.google.ms/url?q=http://www.betop24.com%2F
https://clients1.google.ci/url?q=http://www.betop24.com%2F
https://clients1.google.dm/url?q=http://www.betop24.com%2F
https://clients1.google.com.sb/url?q=http://www.betop24.com%2F
https://clients1.google.co.vi/url?q=http://www.betop24.com%2F
https://clients1.google.so/url?q=http://www.betop24.com%2F
https://clients1.google.nu/url?q=http://www.betop24.com%2F
https://clients1.google.dj/url?q=http://www.betop24.com%2F
https://clients1.google.hn/url?q=http://www.betop24.com%2F
https://clients1.google.nr/url?q=http://www.betop24.com%2F
https://clients1.google.co.tz/url?q=http://www.betop24.com%2F
https://clients1.google.mv/url?q=http://www.betop24.com%2F
https://clients1.google.tn/url?q=http://www.betop24.com%2F
https://clients1.google.sc/url?q=http://www.betop24.com%2F
https://clients1.google.com.py/url?q=http://www.betop24.com%2F
https://clients1.google.sn/url?q=http://www.betop24.com%2F
https://clients1.google.am/url?q=http://www.betop24.com%2F
https://clients1.google.ad/url?q=http://www.betop24.com%2F
https://clients1.google.com.gh/url?q=http://www.betop24.com%2F
https://clients1.google.com.bz/url?q=http://www.betop24.com%2F
https://clients1.google.iq/url?q=http://www.betop24.com%2F
https://clients1.google.to/url?q=http://www.betop24.com%2F
https://clients1.google.com.bn/url?q=http://www.betop24.com%2F
https://clients1.google.cat/url?q=http://www.betop24.com%2F
https://clients1.google.sh/url?q=http://www.betop24.com%2F
https://clients1.google.cm/url?q=http://www.betop24.com%2F
https://clients1.google.gg/url?q=http://www.betop24.com%2F
https://clients1.google.co.ug/url?q=http://www.betop24.com%2F
https://clients1.google.com.ly/url?q=http://www.betop24.com%2F
https://clients1.google.co.uz/url?q=http://www.betop24.com%2F
https://clients1.google.co.zm/url?q=http://www.betop24.com%2F
https://clients1.google.com.na/url?q=http://www.betop24.com%2F
https://clients1.google.com.ag/url?q=http://www.betop24.com%2F
https://clients1.google.me/url?q=http://www.betop24.com%2F
https://clients1.google.cd/url?q=http://www.betop24.com%2F
https://clients1.google.fm/url?q=http://www.betop24.com%2F
https://clients1.google.co.ck/url?q=http://www.betop24.com%2F
https://clients1.google.com.et/url?q=http://www.betop24.com%2F
https://clients1.google.com.pa/url?q=http://www.betop24.com%2F
https://clients1.google.je/url?q=http://www.betop24.com%2F
https://clients1.google.gl/url?q=http://www.betop24.com%2F
https://clients1.google.ge/url?q=http://www.betop24.com%2F
https://clients1.google.ht/url?q=http://www.betop24.com%2F
https://clients1.google.im/url?q=http://www.betop24.com%2F
https://clients1.google.gy/url?q=http://www.betop24.com%2F
https://clients1.google.com.sl/url?q=http://www.betop24.com%2F
https://clients1.google.bj/url?q=http://www.betop24.com%2F
https://clients1.google.ml/url?q=http://www.betop24.com%2F
https://clients1.google.cv/url?q=http://www.betop24.com%2F
https://clients1.google.tl/url?q=http://www.betop24.com%2F
https://clients1.google.com.mm/url?q=http://www.betop24.com%2F
https://clients1.google.bt/url?q=http://www.betop24.com%2F
https://clients1.google.ac/url?q=http://www.betop24.com%2F
https://clients1.google.st/url?q=http://www.betop24.com%2F
https://clients1.google.td/url?q=http://www.betop24.com%2F
https://clients1.google.com.pg/url?q=http://www.betop24.com%2F
https://clients1.google.co.ao/url?q=http://www.betop24.com%2F
https://clients1.google.gp/url?q=http://www.betop24.com%2F
https://clients1.google.com.nf/url?q=http://www.betop24.com%2F
https://clients1.google.ne/url?q=http://www.betop24.com%2F
https://clients1.google.pn/url?q=http://www.betop24.com%2F
https://images.google.com.om/url?q=http://www.betop24.com%2F/
https://maps.google.dz/url?q=http://www.betop24.com%2F/
https://images.google.dz/url?q=http://www.betop24.com%2F/
https://images.google.ga/url?q=http%3A%2F%2Fwww.betop24.com%2F
https://images.google.nu/url?q=http://www.betop24.com%2F/
https://images.google.nu/url?q=http%3A%2F%2Fwww.betop24.com%2F
https://maps.google.nu/url?q=http://www.betop24.com%2F/
https://www.google.nu/url?q=http%3A%2F%2Fwww.betop24.com%2F
https://maps.google.kg/url?q=http://www.betop24.com%2F/
https://maps.google.sc/url?q=http%3A%2F%2Fwww.betop24.com%2F
https://maps.google.sc/url?q=http://www.betop24.com%2F/
https://images.google.sc/url?q=http%3A%2F%2Fwww.betop24.com%2F
https://images.google.sc/url?q=http://www.betop24.com%2F/
https://images.google.sc/url?q=https%3A%2F%2Fwww.betop24.com%2F
https://www.google.sc/url?q=http%3A%2F%2Fwww.betop24.com%2F
https://maps.google.iq/url?q=http://www.betop24.com%2F/
http://www.cnainterpreta.it/redirect.asp?url=http://www.betop24.com%2F/
http://www.site-navi.net/sponavi/rank.cgi?mode=link&id=890&url=http://www.betop24.com%2F/
https://www.mattias.nu/cgi-bin/redirect.cgi?http://www.betop24.com%2F/
http://teenstgp.us/cgi-bin/out.cgi?u=http://www.betop24.com%2F/
http://www.qlt-online.de/cgi-bin/click/clicknlog.pl?link=http://www.betop24.com%2F/
http://ww.sdam-snimu.ru/redirect.php?url=http://www.betop24.com%2F/
http://www.uktrademarkregistration.co.uk/JumpTo.aspx?url=http://www.betop24.com%2F/
http://www.macro.ua/out.php?link=http://www.betop24.com%2F/
http://www.aurki.com/jarioa/redirect?id_feed=510&url=http://www.betop24.com%2F/
http://congovibes.com/index.php?thememode=full;redirect=http://www.betop24.com%2F/
http://www.humaniplex.com/jscs.html?hj=y&ru=http://www.betop24.com%2F/
http://ds-media.info/url?q=http://www.betop24.com%2F/
http://excitingperformances.com/?URL=www.betop24.com%2F
http://familie-huettler.de/link.php?link=www.betop24.com%2F
http://forum.vizslancs.hu/lnks.php?uid=net&url=http://www.betop24.com%2F/
http://forum.wonaruto.com/redirection.php?redirection=http://www.betop24.com%2F/
http://fosteringsuccessmichigan.com/?URL=www.betop24.com%2F
http://fotos24.org/url?q=http://www.betop24.com%2F/
http://fouillez-tout.com/cgi-bin/redirurl.cgi?www.betop24.com%2F
http://frag-den-doc.de/index.php?s=verlassen&url=http://www.betop24.com%2F/
http://freethailand.com/goto.php?url=http://www.betop24.com%2F/
http://freshcannedfrozen.ca/?URL=www.betop24.com%2F
http://funkhouse.de/url?q=http://www.betop24.com%2F/
http://distributors.hrsprings.com/?URL=www.betop24.com%2F
http://dorf-v8.de/url?q=http://www.betop24.com%2F/
http://doverwx.com/template/pages/station/redirect.php?url=http://www.betop24.com%2F/
http://dr-guitar.de/quit.php?url=http://www.betop24.com%2F/
http://drdrum.biz/quit.php?url=http://www.betop24.com%2F/
http://ga.naaar.nl/link/?url=http://www.betop24.com%2F/
http://gb.poetzelsberger.org/show.php?c453c4=www.betop24.com%2F
http://getmethecd.com/?URL=www.betop24.com%2F
http://goldankauf-oberberg.de/out.php?link=http://www.betop24.com%2F/
https://www.fairsandfestivals.net/?URL=http://www.betop24.com%2F/
http://dayviews.com/externalLinkRedirect.php?url=http://www.betop24.com%2F/
http://db.cbservices.org/cbs.nsf/forward?openform&http://www.betop24.com%2F/
http://chatx2.whocares.jp/redir.jsp?u=http://www.betop24.com%2F/
http://chuanroi.com/Ajax/dl.aspx?u=http://www.betop24.com%2F/
http://club.dcrjs.com/link.php?url=http://www.betop24.com%2F/
http://d-quintet.com/i/index.cgi?id=1&mode=redirect&no=494&ref_eid=33&url=http://www.betop24.com%2F/
http://data.allie.dbcls.jp/fct/rdfdesc/usage.vsp?g=http://www.betop24.com%2F/
http://data.linkedevents.org/describe/?url=http://www.betop24.com%2F/
http://conny-grote.de/url?q=http://www.betop24.com%2F/
http://crazyfrag91.free.fr/?URL=http://www.betop24.com%2F
http://crewe.de/url?q=http://www.betop24.com%2F/
http://cwa4100.org/uebimiau/redir.php?http://www.betop24.com%2F/
https://www.hradycz.cz/redir.php?b=445&t=http://www.betop24.com%2F/
https://www.moneydj.com/ads/adredir.aspx?bannerid=39863&url=http://www.betop24.com%2F/
https://uk.kindofbook.com/redirect.php/?red=http://www.betop24.com%2F/
http://m.17ll.com/apply/tourl/?url=http://www.betop24.com%2F/
http://smile.wjp.am/link-free/link3.cgi?mode=cnt&no=8&hpurl=http://www.betop24.com%2F/
https://www.usjournal.com/go.php?campusID=190&url=http://www.betop24.com%2F/
https://temptationsaga.com/buy.php?url=http://www.betop24.com%2F/
http://ads.cars.cz/adclick.php?bannerid=333&zoneid=237&source=&dest=http://www.betop24.com%2F/
https://spb-medcom.ru/redirect.php?http://www.betop24.com%2F/
https://www.info-realty.ru/bitrix/rk.php?goto=http://www.betop24.com%2F/
http://www.sermemole.com/public/serbook/redirect.php?url=http://www.betop24.com%2F/
http://simvol-veri.ru/xp/?goto=http://www.betop24.com%2F/
http://www.skladcom.ru/(S(qdiwhk55jkcyok45u4ti0a55))/banners.aspx?url=http://www.betop24.com%2F/
https://stroim100.ru/redirect?url=http://www.betop24.com%2F/
https://meguro.keizai.biz/banner.php?type=image_banner&position=right&id=13&uri=http://www.betop24.com%2F/
https://kakaku-navi.net/items/detail.php?url=http://www.betop24.com%2F/
http://www.paladiny.ru/go.php?url=http://www.betop24.com%2F/
http://tido.al/vazhdo.php?url=http://www.betop24.com%2F/
http://financialallorner.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://mjghouthernmatron.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://katihfmaxtron.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://ikkemandar.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://googlejfgdlenewstoday.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://bhrecadominicana.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://anupam-bestprice89.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://bhrepublica.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://allinspirations.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://bestandroid.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://discoverable.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://puriagatratt.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://azizlemon.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://prasat.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://meri.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://prabu.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://easehipranaam.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://nidatyi.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://krodyit.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://naine.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://mmurugesamnfo.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://esujkamien.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://ujkaeltnx.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://ausnangck2809.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://supermu.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://buyfcyclingteam.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://allthingnbeyondblog.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://usafunworldt.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://chandancomputers.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://bingshopping.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://ptritam.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://tech-universes.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://littleboy.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://correctinspiration.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://weallfreieds.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://shanmegurad.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://manualmfuctional.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://verybeayurifull.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://breajwasi.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://beithe.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://tumari.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://hariomm.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://lejano.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://jotumko.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://auyttrv.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://bartos.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://cllfather.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://littlejohnny.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://semesmemos.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://faultypirations.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://ptrfsiitan.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://boardgame-breking.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://flowwergulab.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://bingcreater-uk.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://skinnyskin.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://amil.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://mohan.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://kripa.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://charanraj.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://pream.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://sang.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://pasas.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://kanchan.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://udavhav.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://matura.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://patana.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://bopal.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://gwl.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://delhi.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://gopi.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://dilip.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://avinasg.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://virat.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://hsadyttk.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://ashu-quality.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://tinko.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://konkaruna.blog.idnes.cz/redir.aspx?url=http://www.betop24.com%2F/
http://atchs.jp/jump?u=http://www.betop24.com%2F/
http://away3d.com/?URL=www.betop24.com%2F
http://baseballpodcasts.net/Feed2JS/feed2js.php?src=http://www.betop24.com%2F/
http://bbs.mottoki.com/index?bbs=hpsenden&act=link&page=94&linkk=http://www.betop24.com%2F/
http://bernhardbabel.com/url?q=http://www.betop24.com%2F/
http://business.eatonton.com/list?globaltemplate=http://www.betop24.com%2F/
http://cdiabetes.com/redirects/offer.php?URL=http://www.betop24.com%2F/
https://padletcdn.com/cgi/fetch?disposition=attachment&url=http://www.betop24.com%2F/
http://cdn.iframe.ly/api/iframe?url=http://www.betop24.com%2F/
http://centuryofaction.org/?URL=www.betop24.com%2F
http://Somewh.a.T.dfqw@www.newsdiffs.org/article-history/www.findabeautyschool.com/map.aspx?url=http://www.betop24.com%2F/
http://actontv.org/?URL=www.betop24.com%2F
http://albertaaerialapplicators.com/?URL=www.betop24.com%2F
http://alexanderroth.de/url?q=http://www.betop24.com%2F/
http://bigbarganz.com/g/?http://www.betop24.com%2F/
http://archives.midweek.com/?URL=www.betop24.com%2F
http://armdrag.com/mb/get.php?url=http://www.betop24.com%2F/
http://asai-kota.com/acc/acc.cgi?REDIRECT=http://www.betop24.com%2F/
http://ass-media.de/wbb2/redir.php?url=http://www.betop24.com%2F/
http://assadaaka.nl/?URL=www.betop24.com%2F
http://blackberryvietnam.net/proxy.php?link=http://www.betop24.com%2F/
http://boogiewoogie.com/?URL=www.betop24.com%2F
http://bsumzug.de/url?q=http://www.betop24.com%2F/
http://alt.baunetzwissen.de/rd/nl-track/?obj=bnw&cg1=redirect&cg2=wissen-newsletter:beton&cg3=partner&datum=2017-01&titel=partnerklick-beton&firma=informationszentrumbeton&link=http://www.betop24.com%2F/
http://andreasgraef.de/url?q=http://www.betop24.com%2F/
http://app.espace.cool/ClientApi/SubscribeToCalendar/1039?url=http://www.betop24.com%2F/
http://kupiauto.zr.ru/bitrix/rk.php?goto=http://www.betop24.com%2F/
http://ipx.bcove.me/?url=http://www.betop24.com%2F/
http://www.interempresas.net/estadisticas/r.asp?idsector=129&e=221083&c=195&d=http://www.betop24.com%2F/
http://fishbiz.seagrant.uaf.edu/click-thru.html?url=http://www.betop24.com%2F/
http://client.paltalk.com/client/webapp/client/External.wmt?url=http://www.betop24.com%2F/
http://ceskapozice.lidovky.cz/redir.aspx?url=http://www.betop24.com%2F/
http://mitsui-shopping-park.com/lalaport/ebina/redirect.html?url=http://www.betop24.com%2F/
http://community.nxp.com/t5/custom/page/page-id/ExternalRedirect?url=http://www.betop24.com%2F/
http://foro.infojardin.com/proxy.php?link=http://www.betop24.com%2F/
http://twindish-electronics.de/url?q=http://www.betop24.com%2F/
http://www.beigebraunapartment.de/url?q=http://www.betop24.com%2F/
http://www.mix-choice.com/yomi/rank.cgi?mode=link&id=391&url=http://www.betop24.com%2F/
https://cse.google.fm/url?q=http://www.betop24.com%2F/
http://www.1soft-tennis.com/search/rank.cgi?mode=link&id=17&url=http://www.betop24.com%2F/
http://go.e-frontier.co.jp/rd2.php?uri=http://www.betop24.com%2F/
http://www.reddotmedia.de/url?q=http://www.betop24.com%2F/
https://10ways.com/fbredir.php?orig=http://www.betop24.com%2F/
https://emailtrackerapi.leadforensics.com/api/URLOpen?EmailSentRecordID=17006&URL=http://www.betop24.com%2F/
http://search.haga-f.net/rank.cgi?mode=link&url=http://www.betop24.com%2F/
http://7ba.ru/out.php?url=http://www.betop24.com%2F/
http://big-data-fr.com/linkedin.php?lien=http://www.betop24.com%2F/
http://reg.kost.ru/cgi-bin/go?http://www.betop24.com%2F/
http://www.kirstenulrich.de/url?q=http://www.betop24.com%2F/
http://www.inkwell.ru/redirect/?url=http://www.betop24.com%2F/
https://cse.google.co.je/url?q=http://www.betop24.com%2F/
http://click-navi.jp/cgi/service-search/rank.cgi?mode=link&id=121&url=http://www.betop24.com%2F/
https://n1653.funny.ge/redirect.php?url=http://www.betop24.com%2F/
https://www.livecmc.com/?lang=fr&id=Ld9efT&url=http://www.betop24.com%2F/
https://images.google.ee/url?sa=j&source=web&rct=j&url=http://www.betop24.com%2F/
http://www.arndt-am-abend.de/url?q=http://www.betop24.com%2F/
https://maps.google.com.vc/url?q=http://www.betop24.com%2F/
http://maxnetworks.org/searchlink/rank.cgi?mode=link&id=321&url=http://www.betop24.com%2F/
https://image.google.com.ng/url?rct=j&sa=t&url=http://www.betop24.com%2F/
https://www.kenzai-navi.com/location/location_topbanner.php?bs=238&m=899&href=http://www.betop24.com%2F/
https://www.anybeats.jp/jump/?http://www.betop24.com%2F/
http://url-collector.appspot.com/positiveVotes?topic=Hate%20speech&url=http://www.betop24.com%2F/
https://cse.google.com.cy/url?q=http://www.betop24.com%2F/
https://maps.google.be/url?sa=j&url=http://www.betop24.com%2F/
https://top.hange.jp/linkdispatch/dispatch?targetUrl=http://www.betop24.com%2F/
http://www.51queqiao.net/link.php?url=http://www.betop24.com%2F/
https://cse.google.dm/url?q=http://www.betop24.com%2F/
https://rsyosetsu.bookmarks.jp/ys4/rank.cgi?mode=link&id=3519&url=http://www.betop24.com%2F/
http://www.wildner-medien.de/url?q=http://www.betop24.com%2F/
http://www.tifosy.de/url?q=http://www.betop24.com%2F/
https://image.google.co.ck/url?sa=j&source=web&rct=j&url=http://www.betop24.com%2F/
http://ads.icorp.ro/others/STS/?t=CeNortjKxUjK0NDFVsgZcMBADAm4-&g=http://www.betop24.com%2F/
http://deai-ranking.org/search/rank.cgi?mode=link&id=28&url=http://www.betop24.com%2F/
https://izispicy.com/go.php?url=http://www.betop24.com%2F/
http://demo.vieclamcantho.vn/baohiemthatnghiep/Redirect.aspx?sms=90bb20bb20tbb20thc3%B4ng&link=http://www.betop24.com%2F/
http://www.city-fs.de/url?q=http://www.betop24.com%2F/
http://p.profmagic.com/urllink.php?url=http://www.betop24.com%2F/
https://www.google.md/url?q=http://www.betop24.com%2F/
https://maps.google.com.pa/url?q=http://www.betop24.com%2F/
http://www.link.gokinjyo-eikaiwa.com/rank.cgi?mode=link&id=5&url=http://www.betop24.com%2F/
http://www.air-dive.com/au/mt4i.cgi?mode=redirect&ref_eid=697&url=http://www.betop24.com%2F/
http://okozukai.j-web.jp/j-web/okozukai/ys4/rank.cgi?mode=link&id=6817&url=http://www.betop24.com%2F/
https://sfmission.com/rss/feed2js.php?src=http://www.betop24.com%2F/
https://www.watersportstaff.co.uk/extern.aspx?src=http://www.betop24.com%2F/&cu=60096&page=1&t=1&s=42"
http://siamcafe.net/board/go/go.php?http://www.betop24.com%2F/
http://www.whitening-navi.info/cgi/search-smartphone/rank.cgi?mode=link&id=1431&url=http://www.betop24.com%2F/
http://www.youtube.com/redirect?q=http://www.betop24.com%2F/
http://www.youtube.com/redirect?event=channeldescription&q=http://www.betop24.com%2F/
http://www.google.com/url?sa=t&url=http://www.betop24.com%2F/
http://maps.google.com/url?q=http://www.betop24.com%2F/
http://maps.google.com/url?sa=t&url=http://www.betop24.com%2F/
http://plus.google.com/url?q=http://www.betop24.com%2F/
http://cse.google.de/url?sa=t&url=http://www.betop24.com%2F/
http://maps.google.de/url?sa=t&url=http://www.betop24.com%2F/
http://clients1.google.de/url?sa=t&url=http://www.betop24.com%2F/
http://es.catholic.net/ligas/ligasframe.phtml?liga=http://www.betop24.com%2F/
https://www.google.to/url?q=http://www.betop24.com%2F/
https://maps.google.bi/url?q=http://www.betop24.com%2F/
http://www.nickl-architects.com/url?q=http://www.betop24.com%2F/
https://www.ocbin.com/out.php?url=http://www.betop24.com%2F/
http://www.lobenhausen.de/url?q=http://www.betop24.com%2F/
https://image.google.bs/url?q=http://www.betop24.com%2F/
http://redirect.me/?http://www.betop24.com%2F/
http://kank.o.oo7.jp/cgi-bin/ys4/rank.cgi?mode=link&id=569&url=http://www.betop24.com%2F/
http://ruslog.com/forum/noreg.php?http://www.betop24.com%2F/
http://forum.ahigh.ru/away.htm?link=http://www.betop24.com%2F/
https://toolbarqueries.google.td/url?sa=j&source=web&rct=j&url=http://www.betop24.com%2F/
https://home.guanzhuang.org/link.php?url=http://www.betop24.com%2F/
http://dvd24online.de/url?q=http://www.betop24.com%2F/
https://smartservices.ru/bitrix/rk.php?goto=http://www.betop24.com%2F/
http://www.topkam.ru/gtu/?url=http://www.betop24.com%2F/
https://torggrad.ru/bitrix/rk.php?goto=http://www.betop24.com%2F/
http://torgi-rybinsk.ru/bitrix/rk.php?goto=http://www.betop24.com%2F/
http://tpprt.ru/bitrix/rk.php?goto=http://www.betop24.com%2F/
http://www.capitalbikepark.se/bok/go.php?url=http://www.betop24.com%2F/
http://cooltgp.org/tgp/click.php?id=370646&u=http://www.betop24.com%2F/
https://joomlinks.org/?url=http://www.betop24.com%2F/
https://managementportal.de/modules/mod_jw_srfr/redir.php?url=http://www.betop24.com%2F/
http://www.green-cross.pro/bitrix/redirect.php?goto=http://www.betop24.com%2F/
http://m.adlf.jp/jump.php?l=http://www.betop24.com%2F/
https://clients1.google.al/url?q=http://www.betop24.com%2F/
https://cse.google.al/url?q=http://www.betop24.com%2F/
https://images.google.al/url?q=http://www.betop24.com%2F/
http://toolbarqueries.google.al/url?q=http://www.betop24.com%2F/
https://www.google.al/url?q=http://www.betop24.com%2F/
https://www.snek.ai/redirect?url=http://www.betop24.com%2F/
http://gals.graphis.ne.jp/mkr/out.cgi?id=01019&go=http://www.betop24.com%2F/
https://ulfishing.ru/forum/go.php?http://www.betop24.com%2F/
https://underwood.ru/away.html?url=http://www.betop24.com%2F/
https://unicom.ru/links.php?go=http://www.betop24.com%2F/
http://www.unifin.ru/bitrix/redirect.php?goto=http://www.betop24.com%2F/
http://uvbnb.ru/go?http://www.betop24.com%2F/
https://skibaza.ru/bitrix/rk.php?goto=http://www.betop24.com%2F/
http://request-response.com/blog/ct.ashx?url=http://www.betop24.com%2F/
https://turbazar.ru/url/index?url=http://www.betop24.com%2F/
http://members.asoa.org/sso/logout.aspx?returnurl=http://www.betop24.com%2F/
http://login.mediafort.ru/autologin/mail/?code=14844×02ef859015x290299&url=http://www.betop24.com%2F/
https://www.sports-central.org/cgi-bin/axs/ax.pl?http://www.betop24.com%2F/
http://dot.boss.sk/o/rs.php?ssid=1&szid=4&dsid=12&dzid=32&url=http://www.betop24.com%2F
http://www.americanstylefridgefreezer.co.uk/go.php?url=http://www.betop24.com%2F/
https://www.newzealandgirls.co.nz/auckland/escorts/clicks.php?click_id=NavBar_AF_AdultDirectory&target=http://www.betop24.com%2F/
https://wdesk.ru/go?http://www.betop24.com%2F/
https://www.moonbbs.com/dm/dmlink.php?dmurl=http://www.betop24.com%2F/
http://www.mac52ipod.cn/urlredirect.php?go=http://www.betop24.com%2F/
http://chrison.net/ct.ashx?id=dad2849a-8862-4a6d-b842-ef628cbfd3c3&url=http://www.betop24.com%2F/
http://datasheetcatalog.biz/url.php?url=http://www.betop24.com%2F/
http://minlove.biz/out.html?id=nhmode&go=http://www.betop24.com%2F/
http://www.diwaxx.ru/win/redir.php?redir=http://www.betop24.com%2F/
http://www.gearguide.ru/phpbb/go.php?http://www.betop24.com%2F/
https://es.catholic.net/ligas/ligasframe.phtml?liga=http://www.betop24.com%2F/
https://passport.sfacg.com/LoginOut.aspx?Returnurl=http://www.betop24.com%2F/
https://offers.sidex.ru/stat_ym_new.php?redir=http://www.betop24.com%2F/&hash=1577762
http://edcommunity.ru/bitrix/rk.php?goto=http://www.betop24.com%2F/
http://page.yicha.cn/tp/j?url=http://www.betop24.com%2F/
http://www.internettrafficreport.com/cgi-bin/cgirdir.exe?http://www.betop24.com%2F/
https://globalmedia51.ru/bitrix/redirect.php?goto=http://www.betop24.com%2F/
https://ramset.com.au/document/url/?url=http://www.betop24.com%2F/
https://www.vicsport.com.au/analytics/outbound?url=http://www.betop24.com%2F/
http://www.interfacelift.com/goto.php?url=http://www.betop24.com%2F/
https://good-surf.ru/r.php?g=http://www.betop24.com%2F/
http://mbrf.ae/knowledgeaward/language/ar/?redirect_url=http://www.betop24.com%2F/
https://gektor-nsk.ru/bitrix/redirect.php?goto=http://www.betop24.com%2F/
https://lekoufa.ru/banner/go?banner_id=4&link=http://www.betop24.com%2F/
http://www.global-flat.com/mobile/mobile_switch.php?dir=tofull&url=http://www.betop24.com%2F/
https://golden-resort.ru/out.php?out=http://www.betop24.com%2F/
http://avalon.gondor.ru/away.php?link=http://www.betop24.com%2F/
http://www.laosubenben.com/home/link.php?url=http://www.betop24.com%2F/
http://www.lifeshow.com.tw/show.php?ty5_id=1599&url=http://www.betop24.com%2F/
https://forsto.ru/bitrix/redirect.php?goto=http://www.betop24.com%2F/
https://www.cossa.ru/bitrix/redirect.php?event1=click&event2=&event3=&goto=http://www.betop24.com%2F/
http://www.gigatran.ru/go?url=http://www.betop24.com%2F/
http://www.gigaalert.com/view.php?h=&s=http://www.betop24.com%2F/
http://www.jkes.tyc.edu.tw/dyna/webs/gotourl.php?id=357&url=http://www.betop24.com%2F/
http://www.laopinpai.com/gourl.asp?url=/gourl.asp?url=http://www.betop24.com%2F/
http://www.humanbrainmapping.org/i4a/etrack/track.cfm?rType=2&campaignID=3572&contactID=4524&origURL=http://www.betop24.com%2F/
http://www.cyprus-net.com/banner_click.php?banid=4&link=http://www.betop24.com%2F/
http://a.gongkong.com/db/adredir.asp?id=16757&url=http://www.betop24.com%2F/
http://gfaq.ru/go?http://www.betop24.com%2F/
http://gbi-12.ru/links.php?go=http://www.betop24.com%2F/
https://gcup.ru/go?http://www.betop24.com%2F/
http://www.appenninobianco.it/ads/adclick.php?bannerid=159&zoneid=8&source=&dest=http://www.betop24.com%2F/
https://www.eduplus.hk/special/emailalert/goURL.jsp?clickURL=http://www.betop24.com%2F/
http://www.japan.road.jp/navi/navi.cgi?jump=226&url=http://www.betop24.com%2F
http://www.arch.iped.pl/artykuly.php?id=1&cookie=1&url=http://www.betop24.com%2F/
http://hirlevelcenter.eu/click.php?hirlevel_id=13145441085508&url=http://www.betop24.com%2F/
https://birds.cz/avif/redirect.php?from=avif.obs.php&url=http://www.betop24.com%2F/
http://blackhistorydaily.com/black_history_links/link.asp?linkid=5&URL=http://www.betop24.com%2F/
http://www.kitchencabinetsdirectory.com/redirect.asp?url=http://www.betop24.com%2F/
https://perezvoni.com/blog/away?url=http://www.betop24.com%2F/
https://www.ciymca.org/af/register-redirect/71104?url=http://www.betop24.com%2F
https://whizpr.nl/tracker.php?u=http://www.betop24.com%2F/
http://bw.irr.by/knock.php?bid=252583&link=http://www.betop24.com%2F/
http://www.isadatalab.com/redirect?clientId=ee5a64e1-3743-9b4c-d923-6e6d092ae409&appId=69&value=[EMVFIELD]EMAIL[EMV/FIELD]&cat=Techniquesculturales&url=http://www.betop24.com%2F
https://www.jukujo.gs/bin/out.cgi?id=kimiomof&url=http://www.betop24.com%2F/
http://gals.graphis.ne.jp/mkr/out.cgi?id=04489&go=http://www.betop24.com%2F/
http://dir.tetsumania.net/search/rank.cgi?mode=link&id=3267&url=http://www.betop24.com%2F/
http://www.project24.info/mmview.php?dest=http://www.betop24.com%2F
http://buildingreputation.com/lib/exe/fetch.php?media=http://www.betop24.com%2F/
http://moscowdesignmuseum.ru/bitrix/rk.php?goto=http://www.betop24.com%2F/
http://www.fairpoint.net/~jensen1242/gbook/go.php?url=http://www.betop24.com%2F/
https://lirasport.com.ar/bloques/bannerclick.php?id=7&url=http://www.betop24.com%2F/
http://www.knabstrupper.se/guestbook/go.php?url=http://www.betop24.com%2F/
https://www.voxlocalis.net/enlazar/?url=http://www.betop24.com%2F/
http://metalist.co.il/redirect.asp?url=http://www.betop24.com%2F/
http://i-marine.eu/pages/goto.aspx?link=http://www.betop24.com%2F/
http://www.teenlove.biz/cgibin/atc/out.cgi?id=24&u=http://www.betop24.com%2F/
http://www.altoona.com/newsletter/click.php?volume=52&url=http://www.betop24.com%2F/
http://www.katjushik.ru/link.php?to=http://www.betop24.com%2F/
http://tfads.testfunda.com/TFServeAds.aspx?strTFAdVars=4a086196-2c64-4dd1-bff7-aa0c7823a393,TFvar,00319d4f-d81c-4818-81b1-a8413dc614e6,TFvar,GYDH-Y363-YCFJ-DFGH-5R6H,TFvar,http://www.betop24.com%2F/
http://www.beeicons.com/redirect.php?site=http://www.betop24.com%2F/
http://www.diwaxx.ru/hak/redir.php?redir=http://www.betop24.com%2F/
http://www.kyoto-osaka.com/search/rank.cgi?mode=link&url=http://www.betop24.com%2F/
http://games.cheapdealuk.co.uk/go.php?url=http://www.betop24.com%2F/
https://www.netwalkerstore.com/redirect.asp?accid=18244&adcampaignid=2991&adcampaigntype=2&affduration=30&url=http://www.betop24.com%2F/
http://hansolav.net/blog/ct.ashx?id=0af6301b-e71f-44be-838f-905709eee792&url=http://www.betop24.com%2F/
http://bitrix.adlr.ru/bitrix/rk.php?goto=http://www.betop24.com%2F/
https://m.snek.ai/redirect?url=http://www.betop24.com%2F/
http://www.officeservice.com.br/trackMKT/trackerFolha.aspx?DESCRIPTION=HotSite_EcrmContabil&FROM=Workshop&DESTINATION=http://www.betop24.com%2F
https://www.arbsport.ru/gotourl.php?url=http://www.betop24.com%2F/
http://cityprague.ru/go.php?go=http://www.betop24.com%2F/
https://www.hachimantaishi.com/click3/click3.cgi?cnt=c5&url=http://www.betop24.com%2F/
http://www.careerbuilder.com.cn/cn/tallyredirector.aspx?task=Ushi&objName=SnsShare&redirectUrl=http://www.betop24.com%2F&methodName=SetSnsShareLink
http://banners.knollenstein.com/adnetwork/servlet/advertBeans.TrackingServlet?seid=27709655&t=100&redirectTo=www.betop24.com%2F&cid=DECONL&vid=986809&externalsource=jobbsquare_ppc
https://meyeucon.org/ext-click.php?url=http://www.betop24.com%2F
http://www.cnmhe.fr/spip.php?action=cookie&url=http://www.betop24.com%2F
http://tsugarubrand.jp/blog/?wptouch_switch=mobile&redirect=http://www.betop24.com%2F
http://sdchamber.biz/admin/mod_newsletter/redirect.aspx?message_id=785&redirect=http://www.betop24.com%2F/
http://www.p1-uranai.com/rank.cgi?mode=link&id=538&url=http://www.betop24.com%2F/
http://earthsciencescanada.com/modules/babel/redirect.php?newlang=en_us&newurl=http://www.betop24.com%2F/
http://psykodynamiskt.nu/?wptouch_switch=desktop&redirect=http://www.betop24.com%2F
http://www.pcbheaven.com/forum/index.php?thememode=full;redirect=http://www.betop24.com%2F/
https://bondage-guru.net/bitrix/rk.php?goto=http://www.betop24.com%2F/
http://www.westbloomfieldlibrary.org/includes/statistics.php?StatType=Link&StatID=Facebook&weblink=http://www.betop24.com%2F
https://tenisnews.com.br/clickTracker/clickTracker.php?u=http://www.betop24.com%2F/
http://www.stevestechspot.com/ct.ashx?id=9fdcf4a4-6e09-4807-bc31-ac1adf836f6c&url=http://www.betop24.com%2F/
http://rcoi71.ru/bitrix/redirect.php?goto=http://www.betop24.com%2F/
http://www.laselection.net/redir.php3?cat=int&url=www.betop24.com%2F
http://www.masai-mara.com/cgi-bin/link2.pl?grp=mm&link=http://www.betop24.com%2F
http://earnupdates.com/goto.php?url=http://www.betop24.com%2F/
http://www.strana.co.il/finance/redir.aspx?site=http://www.betop24.com%2F
http://www.actuaries.ru/bitrix/rk.php?goto=http://www.betop24.com%2F/
https://naviking.localking.com.tw/about/redirect.aspx?mid=7&url=http://www.betop24.com%2F/
http://sms-muzeji.si/Home/ChangeCulture?lang=sl&returnUrl=http://www.betop24.com%2F
http://r.emeraldexpoinfo.com/s.ashx?ms=EXI2:125462_115115&e=krubin723@aol.com&eId=440186886&c=h&url=http://www.betop24.com%2F
https://apps.firstrain.com/service/openurl.jsp?action=titleclick&src=rss&u=http://www.betop24.com%2F/
http://cdp.thegoldwater.com/click.php?id=101&url=http://www.betop24.com%2F/
http://www.request-response.com/blog/ct.ashx?id=d827b163-39dd-48f3-b767-002147c94e05&url=http://www.betop24.com%2F/
http://www.cooltgp.org/tgp/click.php?id=370646&u=http://www.betop24.com%2F/
http://gondor.ru/go.php?url=http://www.betop24.com%2F/
http://proekt-gaz.ru/go?http://www.betop24.com%2F/
https://www.tricitiesapartmentguide.com/MobileDefault.aspx?reff=http://www.betop24.com%2F/
http://www.vietnamsingle.com/vn/banners/redirect.asp?url=http://www.betop24.com%2F/
https://www.tourplanisrael.com/redir/?url=http://www.betop24.com%2F/
http://www.mutualgravity.com/archangel/webcontact/d_signinsimple.php?action=signup&CID=240&EID=&S=default.css&return=http://www.betop24.com%2F/
http://evenemangskalender.se/redirect/?id=15723&lank=http://www.betop24.com%2F/
http://anifre.com/out.html?go=http://www.betop24.com%2F
http://hobbyplastic.co.uk/trigger.php?r_link=http://www.betop24.com%2F
http://www.castellodivezio.it/lingua.php?lingua=EN&url=http://www.betop24.com%2F
http://ads.pukpik.com/myads/click.php?banner_id=316&banner_url=http://www.betop24.com%2F/
http://no-smok.net/nsmk/InterWiki?action=goto&oe=EUC-KR&url=http://www.betop24.com%2F
http://www.stalker-modi.ru/go?http://www.betop24.com%2F/
https://www.pba.ph/redirect?url=http://www.betop24.com%2F&id=3&type=tab
http://youmydaddy.com/cgi-bin/at3/out.cgi?id=88&trade=http://www.betop24.com%2F
http://barykin.com/go.php?www.betop24.com%2F
https://www.adminer.org/redirect/?sa=t&url=www.betop24.com%2F
http://www.bigblackmamas.com/cgi-bin/sites/out.cgi?url=http://www.betop24.com%2F
http://www.bigblackbootywatchers.com/cgi-bin/sites/out.cgi?url=http://www.betop24.com%2F
https://www.yeaah.com/disco/DiscoGo.asp?ID=3435&Site=http://www.betop24.com%2F
https://primorye.ru/go.php?id=19&url=http://www.betop24.com%2F/
https://www.123gomme.it/it/ViewSwitcher/SwitchView?mobile=True&returnUrl=http://www.betop24.com%2F
https://csirealty.com/?URL=http://www.betop24.com%2F/
http://asadi.de/url?q=http://www.betop24.com%2F/
http://treblin.de/url?q=http://www.betop24.com%2F/
https://kentbroom.com/?URL=http://www.betop24.com%2F/
http://0845.boo.jp/cgi/mt3/mt4i.cgi?id=24&mode=redirect&no=15&ref_eid=3387&url=http://www.betop24.com%2F/
http://www.cercasostituto.it/index.php?name=GestBanner&file=counter&idbanner=40&dir_link=http://www.betop24.com%2F/
http://www.meteo-leran.fr/meteotemplate/template/plugins/deviations/redirect.php?url=http://www.betop24.com%2F/
https://www.eurobichons.com/fda%20alerts.php?url=http://www.betop24.com%2F/
http://search.pointcom.com/k.php?ai=&url=http://www.betop24.com%2F/
https://s-p.me/template/pages/station/redirect.php?url=http://www.betop24.com%2F/
http://ww.thesteelbrothers.com/buy.php?store=iBooks&url=http://www.betop24.com%2F/
http://thdt.vn/convert/convert.php?link=http://www.betop24.com%2F/
http://www.doitweb365.de/scripts/doitweb.exe/rasklickzaehler2?http://www.betop24.com%2F/
http://www.sprang.net/url?q=http://www.betop24.com%2F/
http://nishiyama-takeshi.com/mobile2/mt4i.cgi?id=3&mode=redirect&no=67&ref_eid=671&url=http://www.betop24.com%2F/
https://www.guadamur.eu/template/pages/station/redirect.php?url=http://www.betop24.com%2F/
https://weather.cube.com.gr/pages/station/redirect.php?url=http://www.betop24.com%2F/
http://110.164.66.211/ULIB6//dublin.linkout.php?url=http://www.betop24.com%2F/
http://110.164.92.12/ULIB//dublin.linkout.php?url=http://www.betop24.com%2F/
http://198.54.125.86.myopenlink.net/describe/?url=http://www.betop24.com%2F/
http://202.144.225.38/jmp?url=http://www.betop24.com%2F/
https://forum.everleap.com/proxy.php?link=http://www.betop24.com%2F/
http://www.mosig-online.de/url?q=http://www.betop24.com%2F/
http://www.hccincorporated.com/?URL=http://www.betop24.com%2F/
http://fatnews.com/?URL=http://www.betop24.com%2F/
https://student-helpr.rminds.dev/redirect?redirectTo=http://www.betop24.com%2F/
http://www.kalinna.de/url?q=http://www.betop24.com%2F/
http://www.hartmanngmbh.de/url?q=http://www.betop24.com%2F/
https://www.the-mainboard.com/proxy.php?link=http://www.betop24.com%2F/
http://4coma.net/cgi/mt4/mt4i.cgi?cat=12&mode=redirect&ref_eid=3231&url=http://www.betop24.com%2F/
http://4travel.jp/dynamic/redirect.php?mode=dm_tour&url=http://www.betop24.com%2F/
https://img.2chan.net/bin/jump.php?http://www.betop24.com%2F/
http://www.is.kyusan-u.ac.jp/htmllint/htmllint.cgi?ViewSource=on;URL=http://www.betop24.com%2F/
http://local.rongbachkim.com/rdr.php?url=http://www.betop24.com%2F/
http://bachecauniversitaria.it/link/frm_top.php?url=http://www.betop24.com%2F/
http://www.fimmgviterbo.org/mobfimmgviterbo/index.php?nametm=counter&idbanner=4&dir_link=http://www.betop24.com%2F/
http://www.mckinneyfarm.com/template/plugins/stationExtremes/redirect.php?url=http://www.betop24.com%2F/
https://rb.gy/mixjhy
안전 카지노사이트_https://betop24.com/
온라인카지노 추천_https://betop24.com/
바카라사이트 추천_https://betop24.com/
파라오카지노_https://betop24.com/pharaoh-casino/
쿨카지노_https://betop24.com/cool-casino/
뉴헤븐카지노_https://betop24.com/nhcasino/
솔카지노_https://betop24.com/solcasino/
펀카지노_https://betop24.com/fun-casino/
헤라카지노_https://betop24.com/hera-casino/
J9카지노_https://betop24.com/j9-casino/
https://apaste.info/Ald3
https://bitbin.it/3uzmIXhm/
https://tinyurl.com/598thjcu
https://sites.google.com/view/betop24online/%ED%99%88
https://forms.gle/KZ6riHkPr4o4CtE56
https://linktr.ee/betop24
https://taplink.cc/betop24
https://sleek.bio/betop24
https://betop24.carrd.co/
https://solo.to/betop24
https://biolinky.co/betop24
https://paste.enginehub.org/IDSg9YSy_
https://pasteio.com/xVgdrVZYdk0p
https://apaste.info/nYTW
https://apaste.info/x5f0
https://pasteio.com/xsgR7oZ7rU9Q
http://bit.ly/3mSEOju
https://cutt.ly/E4furTX
https://www.google.co.zw/url?q=https://betop24.com/safe-casino-site%2F
https://www.adminer.org/redirect/?url=https://betop24.com/safe-casino-site/
https://sfwater.org/redirect.aspx?url=https://betop24.com/safe-casino-site/
https://web.archive.org/web/20180804180141/https://betop24.com/safe-casino-site/
https://baoviet.com.vn/Redirect.aspx?url=https://betop24.com/safe-casino-site/
https://sc.sie.gov.hk/TuniS/https://betop24.com/safe-casino-site/
https://www.ric.edu/Pages/link_out.aspx?target=https://betop24.com/safe-casino-site/
https://cse.google.com.pe/url?q=https://betop24.com/safe-casino-site/
https://cse.google.com.pk/url?q=https://betop24.com/safe-casino-site/
https://cse.google.com.np/url?q=https://betop24.com/safe-casino-site/
https://cse.google.com.jm/url?q=https://betop24.com/safe-casino-site/
https://cse.google.li/url?q=https://betop24.com/safe-casino-site/
https://cse.google.com.kh/url?q=https://betop24.com/safe-casino-site/
https://www.google.com.co/url?q=https://betop24.com/safe-casino-site/
https://cse.google.cm/url?q=https://betop24.com/safe-casino-site/
https://cse.google.com.gi/url?q=https://betop24.com/safe-casino-site/
https://cse.google.la/url?q=https://betop24.com/safe-casino-site/
https://cse.google.tk/url?q=https://betop24.com/safe-casino-site/
https://cse.google.co.mz/url?q=https://betop24.com/safe-casino-site/
https://cse.google.com.sb/url?q=https://betop24.com/safe-casino-site/
https://cse.google.sh/url?q=https://betop24.com/safe-casino-site/
https://cse.google.com.ly/url?q=https://betop24.com/safe-casino-site/
https://cse.google.fm/url?q=https://betop24.com/safe-casino-site/
https://cse.google.vu/url?q=https://betop24.com/safe-casino-site/
https://cse.google.cf/url?q=https://betop24.com/safe-casino-site/
https://cse.google.com.tj/url?q=https://betop24.com/safe-casino-site/
https://cse.google.cv/url?q=https://betop24.com/safe-casino-site/
https://cse.google.st/url?q=https://betop24.com/safe-casino-site/
https://cse.google.sr/url?q=https://betop24.com/safe-casino-site/
https://www.google.iq/url?q=https://betop24.com/safe-casino-site/
https://www.google.tk/url?q=https://betop24.com/safe-casino-site/
https://posts.google.com/url?q=https://betop24.com/safe-casino-site/
https://www.google.nu/url?q=https://betop24.com/safe-casino-site/
https://clients1.google.com/url?q=https://betop24.com/safe-casino-site/
https://asia.google.com/url?q=https://betop24.com/safe-casino-site/
https://www.google.com/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.de/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.de/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.uk/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.co.uk/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.co.jp/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.jp/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.co.jp/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.fr/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.es/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.es/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.es/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.it/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.it/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.it/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.br/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.com.br/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.br/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.ca/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.ca/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.ca/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.hk/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.com.hk/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.hk/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.nl/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.nl/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.co.in/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.in/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.co.in/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.ru/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.ru/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.ru/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.pl/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.pl/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.pl/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.com.au/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.au/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.au/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.tw/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.com.tw/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.tw/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.co.id/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.id/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.co.id/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.ch/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.ch/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.be/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.be/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.at/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.at/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.at/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.cz/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.cz/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.th/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.co.th/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.co.th/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.ua/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.ua/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.com.ua/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.tr/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.tr/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.mx/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.mx/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.dk/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.dk/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.hu/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.hu/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.hu/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.fi/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.fi/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.fi/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.nz/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.co.nz/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.co.nz/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.vn/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.com.vn/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.pt/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.pt/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.ro/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.ro/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.ro/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.my/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.my/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.com.my/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.co.za/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.za/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.co.za/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.com.sg/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.sg/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.sg/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.gr/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.gr/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.gr/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.co.il/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.il/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.co.il/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.cl/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.cl/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.cl/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.ie/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.ie/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.ie/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.sk/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.sk/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.sk/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.bg/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.bg/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.bg/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.pe/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.com.pe/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.ae/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.ae/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.ae/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.com.pk/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.pk/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.co/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.co/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.com.co/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.com.eg/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.eg/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.eg/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.lt/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.sa/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.sa/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.com.sa/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.hr/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.hr/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.co.ve/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.co.ve/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.ve/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.ee/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.ee/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.ee/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.si/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.si/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.si/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.by/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.ec/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.com.ec/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.lv/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.lv/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.ba/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.com.ng/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.ng/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.gt/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.gt/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.cr/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.co.cr/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.lu/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.kw/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.com.kw/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.is/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.dz/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.tn/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.tn/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.iq/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.iq/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.cm/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.com.bd/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.bd/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.do/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.do/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.com.do/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.kz/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.ge/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.lk/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.ni/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.la/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.com.jm/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.cy/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.qa/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.com.sv/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.sv/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.ps/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.bo/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.li/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.mn/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.com.bh/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.bh/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.kh/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.lb/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.lb/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.tt/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.tt/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.ci/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.hn/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.hn/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.sn/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.sn/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.com.mt/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.mt/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.cat/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.bi/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.bi/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.cd/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.fm/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.fm/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.fm/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.as/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.pa/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.pa/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.om/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.om/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.mz/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.co.bw/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.ms/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.kg/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.com.bz/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.bz/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.bn/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.sh/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.ag/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.com.ag/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
http://app.feedblitz.com/f/f.fbz?track=http%3A%2F%2Fbetop24.com/safe-casino-site/
http://archives.midweek.com/?URL=http%253A%252F%252Fhttps://betop24.com/safe-casino-site/
http://fooyoh.com/wcn.php?url=http%3A%2F%2Fbetop24.com/safe-casino-site/&keyid=42973
http://m.odnoklassniki.ru/dk?st.cmd=outLinkWarning&st.rfn=http%3A%2F%2Fbetop24.com/safe-casino-site/
http://onlinemanuals.txdot.gov/help/urlstatusgo.html?url=http%3A%2F%2Fbetop24.com/safe-casino-site/
http://onlinemanuals.txdot.gov/help/urlstatusgo.html?url=https://betop24.com/safe-casino-site/
http://sc.sie.gov.hk/TuniS/https://betop24.com/safe-casino-site/
http://www.aa.org/pages/en_US/disclaimer?u=http%3A%2F%2Fbetop24.com/safe-casino-site/
http://www.aastocks.com/sc/changelang.aspx?lang=sc&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
http://www.blueletterbible.org/tools/redirect.cfm?Site=http%3A%2F%2Fbetop24.com/safe-casino-site/
http://www.cineuropa.org/el.aspx?el=http%3A%2F%2Fbetop24.com/safe-casino-site/
http://www.drinksmixer.com/redirect.php?url=http%3A%2F%2Fbetop24.com/safe-casino-site/
http://www.justjared.com/flagcomment.php?el=http%3A%2F%2Fbetop24.com/safe-casino-site/
http://www.kaskus.co.id/redirect?url=http%3A%2F%2Fbetop24.com/safe-casino-site/
http://www.meetme.com/apps/redirect/?url=http%3A%2F%2Fbetop24.com/safe-casino-site/
http://www.office.xerox.com/perl-bin/reseller_exit.pl?url=http%3A%2F%2Fbetop24.com/safe-casino-site/
http://www.popcouncil.org/scripts/leaving.asp?URL=http%3A%2F%2Fbetop24.com/safe-casino-site/
http://www.ric.edu/Pages/link_out.aspx?target=http%3A%2F%2Fbetop24.com/safe-casino-site/
http://www.sinp.msu.ru/ru/ext_link?url=http%3A%2F%2Fbetop24.com/safe-casino-site/
http://www.webclap.com/php/jump.php?url=https://betop24.com/safe-casino-site/
http://www2.sandbox.google.co.jp/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
http://www2.sandbox.google.co.kr/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
http://cr.naver.com/rd?u=http%3A%2F%2Fbetop24.com/safe-casino-site/
http://devicedoctor.com/driver-feedback.php?device=PCI%20bus&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
http://domain.opendns.com/https://betop24.com/safe-casino-site/
http://my.dek-d.com/username/link/link.php?out=http%3A%2F%2Fbetop24.com/safe-casino-site/
http://panchodeaonori.sakura.ne.jp/feed/aonori/feed2js.php?src=http%3A%2F%2Fbetop24.com/safe-casino-site/
http://refer.ash1.ccbill.com/cgi-bin/clicks.cgi?CA=933914&PA=1785830&HTML=http%3A%2F%2Fbetop24.com/safe-casino-site/
http://refer.ccbill.com/cgi-bin/clicks.cgi/http:/?CA=928498&PA=1458253&HTML=http%3A%2F%2Fbetop24.com/safe-casino-site/
http://sc.youth.gov.hk/TuniS/https://betop24.com/safe-casino-site/
http://wasearch.loc.gov/e2k/*/https://betop24.com/safe-casino-site/
http://wikimapia.org/external_link?url=https://betop24.com/safe-casino-site/
http://www.astro.wisc.edu/?URL=http%3A%2F%2Fbetop24.com/safe-casino-site/
http://www.astro.wisc.edu/?URL=https://betop24.com/safe-casino-site/
http://www.cssdrive.com/?URL=https://betop24.com/safe-casino-site/
http://www.earth-policy.org/?URL=https://betop24.com/safe-casino-site/
http://www.fmvz.unam.mx/fmvz/biblioteca/revistas_electronicas/control/envioClics.php?cnH57w=1118&kqJm89=http%3A%2F%2Fbetop24.com/safe-casino-site/
http://www.ric.edu/Pages/link_out.aspx?target=https://betop24.com/safe-casino-site/
http://www.shinobi.jp/etc/goto.html?http%3A%2F%2Fbetop24.com/safe-casino-site/kuttymovies-2020-kuttymovies-hd-tamil-movies-download
http://www.spiritfanfiction.com/link?l=http%3A%2F%2Fbetop24.com/safe-casino-site/
http://www.webclap.com/php/jump.php?url=http%3A%2F%2Fbetop24.com/safe-casino-site/
http://www.zerocarts.com/demo/index.php?url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://advisor.wmtransfer.com/SiteDetails.aspx?url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://blog.ss-blog.jp/pages/mobile/step/index?u=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://blogs.rtve.es/libs/getfirma_footer_prod.php?blogurl=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://bukkit.org/proxy.php?link=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://client.paltalk.com/client/webapp/client/External.wmt?url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://club.panasonic.jp/member/terms/?siteId=B1&returnURL=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://community.cypress.com/external-link.jspa?url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://community.esri.com/external-link.jspa?url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://community.rsa.com/external-link.jspa?url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://cse.google.ac/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.ad/url?q=https://betop24.com/safe-casino-site/
https://cse.google.ad/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.ae/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.al/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.am/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.as/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.at/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.az/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.ba/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.bg/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.bi/url?q=https://betop24.com/safe-casino-site/
https://cse.google.bi/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.bj/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.bs/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.bt/url?q=https://betop24.com/safe-casino-site/
https://cse.google.bt/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.by/url?q=https://betop24.com/safe-casino-site/
https://cse.google.by/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.ca/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.cat/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.cd/url?q=https://betop24.com/safe-casino-site/
https://cse.google.cd/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.cf/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.cg/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.ch/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.ci/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.cl/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.cm/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.co.ao/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.co.bw/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.co.ck/url?q=https://betop24.com/safe-casino-site/
https://cse.google.co.cr/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.co.id/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.co.in/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.co.jp/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.co.ke/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.co.kr/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.co.ls/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.co.ma/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.co.mz/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.co.nz/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.co.th/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.co.tz/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.co.ug/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.co.uz/url?q=https://betop24.com/safe-casino-site/
https://cse.google.co.uz/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.co.ve/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.co.vi/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.co.za/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.co.zm/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.co.zw/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.com.af/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.com.ag/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.com.ai/url?q=https://betop24.com/safe-casino-site/
https://cse.google.com.ai/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.com.au/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.com.bd/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.com.bh/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.com.bn/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.com.bo/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.com.co/url?q=https://betop24.com/safe-casino-site/
https://cse.google.com.co/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.com.cu/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.com.cy/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.com.ec/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.com.eg/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.com.et/url?q=https://betop24.com/safe-casino-site/
https://cse.google.com.et/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.com.fj/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.com.gh/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.com.gi/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.com.gt/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.com.hk/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.com.jm/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.com.kh/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.com.kw/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.com.ly/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.com.mm/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.com.mt/url?q=https://betop24.com/safe-casino-site/
https://cse.google.com.mt/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.com.mx/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.com.my/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.com.na/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.com.nf/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.com.ng/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.com.ni/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.com.np/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.com.om/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.com.pa/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.com.pe/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.com.pg/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.com.ph/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.com.pr/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.com.sa/url?q=https://betop24.com/safe-casino-site/
https://cse.google.com.sa/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.com.sg/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.com.sv/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.com.tj/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.com.tr/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.com.tw/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.com.ua/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.com.uy/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.com.vc/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.com/url?q=https://betop24.com/safe-casino-site/
https://cse.google.com/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.cz/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.de/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.dk/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.dz/url?q=https://betop24.com/safe-casino-site/
https://cse.google.dz/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.ee/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.es/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.fi/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.fr/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.ga/url?q=https://betop24.com/safe-casino-site/
https://cse.google.ga/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.gg/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.gl/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.gm/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.gp/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.gr/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.gy/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.hn/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.hr/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.ht/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.hu/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.ie/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.im/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.iq/url?q=https://betop24.com/safe-casino-site/
https://cse.google.is/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.it/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.je/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.jo/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.kg/url?q=https://betop24.com/safe-casino-site/
https://cse.google.kg/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.ki/url?q=https://betop24.com/safe-casino-site/
https://cse.google.ki/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.kz/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.la/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.li/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.lk/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.lt/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.lu/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.lv/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.me/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.mg/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.mk/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.mn/url?q=https://betop24.com/safe-casino-site/
https://cse.google.mn/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.ms/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.mu/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.mv/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.mw/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.ne/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.nl/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.no/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.nr/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.nu/url?q=https://betop24.com/safe-casino-site/
https://cse.google.nu/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.ps/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.ro/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.rs/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.ru/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.rw/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.sc/url?q=https://betop24.com/safe-casino-site/
https://cse.google.se/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.sh/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.si/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.sk/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.sm/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.sn/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.so/url?q=https://betop24.com/safe-casino-site/
https://cse.google.sr/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.td/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.tg/url?q=https://betop24.com/safe-casino-site/
https://cse.google.tg/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.tl/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.tm/url?q=https://betop24.com/safe-casino-site/
https://cse.google.tm/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.tn/url?q=https://betop24.com/safe-casino-site/
https://cse.google.to/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.tt/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.vg/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.vu/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.ws/url?q=https://betop24.com/safe-casino-site/
https://cse.google.ws/url?sa=i&url=https://betop24.com/safe-casino-site/
https://ditu.google.com/url?q=https://betop24.com/safe-casino-site/
https://foro.infojardin.com/proxy.php?link=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://foro.infojardin.com/proxy.php?link=https://betop24.com/safe-casino-site/
https://forum.solidworks.com/external-link.jspa?url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://galter.northwestern.edu/exit?url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://id.telstra.com.au/register/crowdsupport?gotoURL=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.ad/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.ad/url?q=https://betop24.com/safe-casino-site/
https://images.google.ae/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.at/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.be/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.bg/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.bs/url?q=https://betop24.com/safe-casino-site/
https://images.google.bt/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.bt/url?q=https://betop24.com/safe-casino-site/
https://images.google.by/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.by/url?q=https://betop24.com/safe-casino-site/
https://images.google.ca/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.cat/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.ch/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.cl/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.bw/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.id/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.il/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.in/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.jp/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.kr/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.nz/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.th/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.ug/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.ug/url?q=https://betop24.com/safe-casino-site/
https://images.google.co.uk/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.uz/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.uz/url?q=https://betop24.com/safe-casino-site/
https://images.google.co.ve/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.za/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.au/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.bd/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.br/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.co/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.do/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.ec/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.eg/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.et/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.et/url?q=https://betop24.com/safe-casino-site/
https://images.google.com.gt/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.hk/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.jm/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.jm/url?q=https://betop24.com/safe-casino-site/
https://images.google.com.kh/url?q=https://betop24.com/safe-casino-site/
https://images.google.com.lb/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.mx/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.my/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.ng/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.np/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.om/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.om/url?q=https://betop24.com/safe-casino-site/
https://images.google.com.pe/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.pe/url?q=https://betop24.com/safe-casino-site/
https://images.google.com.ph/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.pk/url?q=https://betop24.com/safe-casino-site/
https://images.google.com.pk/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.sa/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.sa/url?q=https://betop24.com/safe-casino-site/
https://images.google.com.sa/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.sg/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.sv/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.tj/url?q=https://betop24.com/safe-casino-site/
https://images.google.com.tr/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.tw/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.ua/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.uy/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.vn/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.cz/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.de/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.dk/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.dm/url?q=https://betop24.com/safe-casino-site/
https://images.google.dz/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.dz/url?q=https://betop24.com/safe-casino-site/
https://images.google.ee/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.es/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.fi/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.fr/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.gr/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.hr/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.hu/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.ie/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.iq/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.iq/url?q=https://betop24.com/safe-casino-site/
https://images.google.it/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.kg/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.kg/url?q=https://betop24.com/safe-casino-site/
https://images.google.lk/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.lt/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.lu/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.lv/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.mn/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.mn/url?q=https://betop24.com/safe-casino-site/
https://images.google.nl/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.pl/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.pt/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.ro/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.ru/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.sc/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.se/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.si/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.sk/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.tm/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.tm/url?q=https://betop24.com/safe-casino-site/
https://images.google.tn/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.tn/url?q=https://betop24.com/safe-casino-site/
https://images.google.tt/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.ws/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.ws/url?q=https://betop24.com/safe-casino-site/
https://legacy.aom.org/verifymember.asp?nextpage=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://m.ok.ru/dk?st.cmd=outLinkWarning&st.rfn=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.ae/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.at/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.be/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.bg/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.bs/url?q=https://betop24.com/safe-casino-site/
https://maps.google.bt/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.bt/url?q=https://betop24.com/safe-casino-site/
https://maps.google.by/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.ca/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.ch/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.cl/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.co.cr/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.co.id/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.co.il/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.co.in/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.co.jp/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.co.ke/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.co.kr/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.co.nz/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.co.th/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.co.ug/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.co.uk/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.co.ve/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.co.za/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.au/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.br/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.co/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.do/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.ec/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.eg/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.et/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.et/url?q=https://betop24.com/safe-casino-site/
https://maps.google.com.gt/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.hk/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.jm/url?q=https://betop24.com/safe-casino-site/
https://maps.google.com.mx/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.my/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.ng/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.ni/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.om/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.om/url?q=https://betop24.com/safe-casino-site/
https://maps.google.com.pe/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.ph/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.sg/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.tr/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.tw/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.cz/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.dk/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.dz/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.dz/url?q=https://betop24.com/safe-casino-site/
https://maps.google.es/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.fi/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.fr/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.ge/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.gr/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.hn/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.hr/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.iq/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.iq/url?q=https://betop24.com/safe-casino-site/
https://maps.google.it/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.kg/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.kg/url?q=https://betop24.com/safe-casino-site/
https://maps.google.li/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.li/url?q=https://betop24.com/safe-casino-site/
https://maps.google.lk/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.lt/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.lu/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.lv/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.mn/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.mn/url?q=https://betop24.com/safe-casino-site/
https://maps.google.pl/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.pt/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.ro/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.ru/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.sc/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.se/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.si/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.sk/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.tn/url?q=https://betop24.com/safe-casino-site/
https://maps.google.tn/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.vu/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.vu/url?q=https://betop24.com/safe-casino-site/
https://maps.google.ws/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.ws/url?q=https://betop24.com/safe-casino-site/
https://optimize.viglink.com/page/pmv?url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://plus.google.com/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://pw.mail.ru/forums/fredirect.php?url=https://betop24.com/safe-casino-site/
https://ref.gamer.com.tw/redir.php?url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://rspcb.safety.fhwa.dot.gov/pageRedirect.aspx?RedirectedURL=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://sfwater.org/redirect.aspx?url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://sfwater.org/redirect.aspx?url=https://betop24.com/safe-casino-site/
https://wfc2.wiredforchange.com/dia/track.jsp?v=2&c=hdorrh%2BHcDlQ%2BzUEnZU5qlfKZ1Cl53X6&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://wikimapia.org/external_link?url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.adminer.org/redirect/?url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.ae/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.as/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.at/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.be/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.bg/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.by/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.ca/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.ch/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.cl/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.co.bw/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.co.id/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.co.il/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.co.in/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.co.jp/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.co.ke/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.co.kr/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.co.ma/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.co.nz/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.co.th/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.co.uk/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.co.ve/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.co.za/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.com.au/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.com.bd/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.com.br/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.com.co/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.com.do/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.com.ec/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.com.eg/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.com.hk/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.com.mx/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.com.my/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.com.ng/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.com.np/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.com.pe/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.com.ph/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.com.pk/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.com.pr/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.com.sa/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.com.sg/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.com.sv/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.com.tr/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.com.tw/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.com.ua/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.com.uy/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.com.vn/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.com/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.cz/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.de/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.dk/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.dz/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.ee/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.fi/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.fr/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.gr/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.hn/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.hr/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.ie/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.is/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.it/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.kg/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.kg/url?q=https://betop24.com/safe-casino-site/
https://www.google.kz/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.lu/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.lv/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.nl/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.pt/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.ro/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.ru/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.se/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.si/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.tn/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.tt/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.transtats.bts.gov/exit.asp?url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.transtats.bts.gov/exit.asp?url=https://betop24.com/safe-casino-site/
https://www.youtube.com/redirect?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://youtube.com/redirect?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://advisor.wmtransfer.com/SiteDetails.aspx?url=https://betop24.com/safe-casino-site/
https://advisor.wmtransfer.com/SiteDetails.aspx?url=https://betop24.com/safe-casino-site/&tab=feedback
https://advisor.wmtransfer.com/SiteDetails.aspx?url=https://betop24.com/safe-casino-site/&tab=rating
https://advisor.wmtransfer.com/SiteDetails.aspx?url=https://betop24.com/safe-casino-site/&tab=wminfo
https://advisor.wmtransfer.com/SiteDetails.aspx?url=https://betop24.com/safe-casino-site/
https://anonym.to/?https://betop24.com/safe-casino-site/
https://anonym.to/?http%3A%2F%2Fbetop24.com/safe-casino-site/
https://blogs.rtve.es/libs/getfirma_footer_prod.php?blogurl=https://betop24.com/safe-casino-site/
https://blogs.rtve.es/libs/getfirma_footer_prod.php?blogurl=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://bukkit.org/proxy.php?link=https://betop24.com/safe-casino-site/
https://bukkit.org/proxy.php?link=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://cdrinfo.com/Sections/Ads/ReviewsAroundTheWebRedirector.aspx?TargetUrl=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://ceskapozice.lidovky.cz/redir.aspx?url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://cgl.ethz.ch/disclaimer.php?dlurl=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://chofu.keizai.biz/banner.php?type=text_banner&position=right&id=3&uri=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.ac/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.ad/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.ae/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.al/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.am/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.as/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.az/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.bf/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.bi/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.bj/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.bs/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.bt/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.by/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.cd/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.cf/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.cg/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.ci/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.cm/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.co.ao/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.co.ck/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.co.il/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.co.in/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.co.jp/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.co.kr/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.co.ls/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.co.ma/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.co.mz/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.co.nz/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.co.th/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.co.tz/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.co.uz/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.co.ve/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.co.vi/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.co.zm/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.co.zw/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.com.af/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.com.ag/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.com.ai/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.com.ar/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.com.bh/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.com.bn/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.com.bo/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.com.bz/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.com.cu/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.com.cy/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.com.eg/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.com.fj/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.com.gh/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.com.gi/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.com.hk/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.com.jm/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.com.kh/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.com.kw/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.com.ly/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.com.mm/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.com.mt/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.com.my/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.com.na/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.com.np/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.com.om/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.com.pe/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.com.pg/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.com.pk/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.com.pr/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.com.qa/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.com.sa/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.com.sl/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.com.tj/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.com.tr/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.com.tw/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.com.ua/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.com.uy/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.com.vn/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.cv/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.de/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.dj/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.dm/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.fm/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.ga/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.gg/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.gl/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.gm/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.gp/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.gy/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.ht/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.ie/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.im/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.is/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.it/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.jo/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.kg/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.ki/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.la/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.li/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.md/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.me/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.mg/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.mn/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.ms/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.mv/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.mw/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.ne/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.nr/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.nu/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.pn/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.ro/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.rw/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.sc/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.sh/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.sm/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.so/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.sr/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.st/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.td/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.tg/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.tl/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.tm/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.to/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.tt/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.vg/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.vu/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.ws/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://club.panasonic.jp/member/terms/?siteId=B1&returnURL=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://community.cypress.com/external-link.jspa?url=https://betop24.com/safe-casino-site/
https://community.cypress.com/external-link.jspa?url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://community.esri.com/external-link.jspa?url=https://betop24.com/safe-casino-site/
https://community.esri.com/external-link.jspa?url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://community.rsa.com/external-link.jspa?url=https://betop24.com/safe-casino-site/
https://contacts.google.com/url?sa=t&source=web&rct=j&url=http%3A%2F%2Fbetop24.com/safe-casino-site/&ved=2ahUKEwip75vMsq7mAhUv06YKHYSzAN44rAIQFjAYegQIKRAB
https://cse.google.ba/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://cse.google.by/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://cse.google.cm/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://cse.google.co.id/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://cse.google.co.in/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://cse.google.co.jp/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://cse.google.co.ke/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://cse.google.com.br/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://cse.google.com.hk/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://cse.google.com.mx/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://cse.google.com.nf/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://cse.google.com.ng/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://cse.google.com.pr/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://cse.google.com.tr/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://cse.google.com.tw/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://cse.google.com.vn/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://cse.google.de/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://cse.google.es/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://cse.google.it/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://cse.google.ru/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://cse.google.se/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://cse.google.tt/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://demo.html5xcss3.com/demo.php?url=https://betop24.com/safe-casino-site/
https://dereferer.org/?https://betop24.com/safe-casino-site/
https://drive.sandbox.google.com.br/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://engawa.kakaku.com/jump/?url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://eventlog.centrum.cz/redir?data=aclick1c68565-349178t12&s=najistong&v=1&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://foro.infojardin.com/proxy.php?link=https://betop24.com/safe-casino-site/%2F
https://forum.solidworks.com/external-link.jspa?url=https://betop24.com/safe-casino-site/
https://forum.solidworks.com/external-link.jspa?url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://forums-archive.eveonline.com/default.aspx?g=warning&l=http%3A%2F%2Fbetop24.com/safe-casino-site/&domain=https://betop24.com/safe-casino-site/
https://galter.northwestern.edu/exit?url=https://betop24.com/safe-casino-site/
https://galter.northwestern.edu/exit?url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://gleam.io/zyxKd-INoWr2EMzH?l=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://hobby.idnes.cz/peruanske-palive-papricky-rocoto-dlz-/redir.aspx?url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://id.telstra.com.au/register/crowdsupport?gotoURL=https://betop24.com/safe-casino-site/
https://images.google.ac/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.ac/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.ad/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.ae/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.ae/url?q=https://betop24.com/safe-casino-site/
https://images.google.al/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.al/url?q=https://betop24.com/safe-casino-site/
https://images.google.al/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.am/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.am/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.as/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.as/url?q=https://betop24.com/safe-casino-site/
https://images.google.as/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.at/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.at/url?q=https://betop24.com/safe-casino-site/
https://images.google.az/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.az/url?q=https://betop24.com/safe-casino-site/
https://images.google.az/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.ba/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.ba/url?q=https://betop24.com/safe-casino-site/
https://images.google.ba/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.be/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.be/url?q=https://betop24.com/safe-casino-site/
https://images.google.bf/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.bf/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.bg/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.bi/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.bi/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.bj/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.bj/url?q=https://betop24.com/safe-casino-site/
https://images.google.bj/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.bs/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.bt/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.by/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.ca/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.cat/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.cat/url?q=https://betop24.com/safe-casino-site/
https://images.google.cd/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.cd/url?q=https://betop24.com/safe-casino-site/
https://images.google.cd/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.cf/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.cf/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.cg/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.cg/url?q=https://betop24.com/safe-casino-site/
https://images.google.cg/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.ch/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.ch/url?q=https://betop24.com/safe-casino-site/
https://images.google.ci/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.ci/url?q=https://betop24.com/safe-casino-site/
https://images.google.ci/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.cl/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.cm/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.cm/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.ao/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.ao/url?q=https://betop24.com/safe-casino-site/
https://images.google.co.ao/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.bw/url?q=https://betop24.com/safe-casino-site/
https://images.google.co.ck/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.ck/url?q=https://betop24.com/safe-casino-site/
https://images.google.co.ck/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.cr/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.cr/url?q=https://betop24.com/safe-casino-site/
https://images.google.co.id/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.id/url?q=https://betop24.com/safe-casino-site/
https://images.google.co.il/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.in/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.jp/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.ke/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.ke/url?q=https://betop24.com/safe-casino-site/
https://images.google.co.ke/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.kr/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.kr/url?q=https://betop24.com/safe-casino-site/
https://images.google.co.ls/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.ls/url?q=https://betop24.com/safe-casino-site/
https://images.google.co.ls/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.ma/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.ma/url?q=https://betop24.com/safe-casino-site/
https://images.google.co.mz/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.mz/url?q=https://betop24.com/safe-casino-site/
https://images.google.co.mz/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.nz/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.th/url?q=https://betop24.com/safe-casino-site/
https://images.google.co.tz/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.tz/url?q=https://betop24.com/safe-casino-site/
https://images.google.co.tz/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.ug/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.uk/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.ve/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.ve/url?q=https://betop24.com/safe-casino-site/
https://images.google.co.vi/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.vi/url?q=https://betop24.com/safe-casino-site/
https://images.google.co.vi/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.za/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.za/url?q=https://betop24.com/safe-casino-site/
https://images.google.co.zm/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.zm/url?q=https://betop24.com/safe-casino-site/
https://images.google.co.zm/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.zw/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.zw/url?q=https://betop24.com/safe-casino-site/
https://images.google.co.zw/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.af/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.af/url?q=https://betop24.com/safe-casino-site/
https://images.google.com.af/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.ag/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.ag/url?q=https://betop24.com/safe-casino-site/
https://images.google.com.ag/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.ai/url?q=https://betop24.com/safe-casino-site/
https://images.google.com.ai/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.ai/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.ar/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.ar/url?q=https://betop24.com/safe-casino-site/
https://images.google.com.ar/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.au/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.bd/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.bh/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.bh/url?q=https://betop24.com/safe-casino-site/
https://images.google.com.bh/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.bn/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.bn/url?q=https://betop24.com/safe-casino-site/
https://images.google.com.bn/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.bo/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.bo/url?q=https://betop24.com/safe-casino-site/
https://images.google.com.bo/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.bz/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.bz/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.co/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.co/url?q=https://betop24.com/safe-casino-site/
https://images.google.com.cy/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.cy/url?q=https://betop24.com/safe-casino-site/
https://images.google.com.cy/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.do/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.do/url?q=https://betop24.com/safe-casino-site/
https://images.google.com.ec/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.eg/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.eg/url?q=https://betop24.com/safe-casino-site/
https://images.google.com.et/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.fj/url?q=https://betop24.com/safe-casino-site/
https://images.google.com.fj/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.gh/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.gh/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.gi/url?q=https://betop24.com/safe-casino-site/
https://images.google.com.gt/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.gt/url?q=https://betop24.com/safe-casino-site/
https://images.google.com.hk/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.hk/url?q=https://betop24.com/safe-casino-site/
https://images.google.com.jm/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.kh/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.kh/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.kw/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.kw/url?q=https://betop24.com/safe-casino-site/
https://images.google.com.kw/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.lb/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.ly/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.ly/url?q=https://betop24.com/safe-casino-site/
https://images.google.com.ly/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.mm/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.mm/url?q=https://betop24.com/safe-casino-site/
https://images.google.com.mm/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.mt/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.mt/url?q=https://betop24.com/safe-casino-site/
https://images.google.com.mt/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.mx/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.mx/url?q=https://betop24.com/safe-casino-site/
https://images.google.com.my/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.my/url?q=https://betop24.com/safe-casino-site/
https://images.google.com.na/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.na/url?q=https://betop24.com/safe-casino-site/
https://images.google.com.na/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.nf/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.ng/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.ni/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.ni/url?q=https://betop24.com/safe-casino-site/
https://images.google.com.np/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.np/url?q=https://betop24.com/safe-casino-site/
https://images.google.com.om/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.pa/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.pa/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.pg/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.pg/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.ph/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.ph/url?q=https://betop24.com/safe-casino-site/
https://images.google.com.pk/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.pr/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.pr/url?q=https://betop24.com/safe-casino-site/
https://images.google.com.pr/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.py/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.py/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.qa/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.qa/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.sg/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.sg/url?q=https://betop24.com/safe-casino-site/
https://images.google.com.sl/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.sl/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.sv/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.tj/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.tj/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.tr/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.tw/url?q=https://betop24.com/safe-casino-site/
https://images.google.com.ua/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.ua/url?q=https://betop24.com/safe-casino-site/
https://images.google.com.uy/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.uy/url?q=https://betop24.com/safe-casino-site/
https://images.google.com.vc/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.vc/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.vn/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.vn/url?q=https://betop24.com/safe-casino-site/
https://images.google.com/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com/url?sa=t&url=https://betop24.com/safe-casino-site/
https://images.google.cv/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.cv/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.cz/url?q=https://betop24.com/safe-casino-site/
https://images.google.de/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.de/url?q=https://betop24.com/safe-casino-site/
https://images.google.dj/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.dj/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.dk/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.dk/url?q=https://betop24.com/safe-casino-site/
https://images.google.dm/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.dm/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.dz/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.ee/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.ee/url?q=https://betop24.com/safe-casino-site/
https://images.google.es/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.es/url?q=https://betop24.com/safe-casino-site/
https://images.google.fi/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.fi/url?q=https://betop24.com/safe-casino-site/
https://images.google.fm/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.fm/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.fr/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.fr/url?q=https://betop24.com/safe-casino-site/
https://images.google.ga/url?q=https://betop24.com/safe-casino-site/
https://images.google.ga/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.ga/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.ge/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.ge/url?q=https://betop24.com/safe-casino-site/
https://images.google.ge/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.gg/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.gg/url?q=https://betop24.com/safe-casino-site/
https://images.google.gg/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.gl/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.gl/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.gm/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.gm/url?q=https://betop24.com/safe-casino-site/
https://images.google.gm/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.gp/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.gp/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.gr/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.gr/url?q=https://betop24.com/safe-casino-site/
https://images.google.gy/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.hn/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.hn/url?q=https://betop24.com/safe-casino-site/
https://images.google.hn/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.hr/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.hr/url?q=https://betop24.com/safe-casino-site/
https://images.google.ht/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.ht/url?q=https://betop24.com/safe-casino-site/
https://images.google.ht/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.hu/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.hu/url?q=https://betop24.com/safe-casino-site/
https://images.google.ie/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.ie/url?q=https://betop24.com/safe-casino-site/
https://images.google.im/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.im/url?q=https://betop24.com/safe-casino-site/
https://images.google.im/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.iq/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.is/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.is/url?q=https://betop24.com/safe-casino-site/
https://images.google.it/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.it/url?q=https://betop24.com/safe-casino-site/
https://images.google.je/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.je/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.jo/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.jo/url?q=https://betop24.com/safe-casino-site/
https://images.google.jo/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.kg/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.ki/url?q=https://betop24.com/safe-casino-site/
https://images.google.ki/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.ki/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.kz/url?q=https://betop24.com/safe-casino-site/
https://images.google.kz/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.la/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.la/url?q=https://betop24.com/safe-casino-site/
https://images.google.la/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.li/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.li/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.lk/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.lk/url?q=https://betop24.com/safe-casino-site/
https://images.google.lu/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.lu/url?q=https://betop24.com/safe-casino-site/
https://images.google.lv/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.lv/url?q=https://betop24.com/safe-casino-site/
https://images.google.md/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.md/url?q=https://betop24.com/safe-casino-site/
https://images.google.md/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.me/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.me/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.mg/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.mg/url?q=https://betop24.com/safe-casino-site/
https://images.google.mg/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.mk/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.mk/url?q=https://betop24.com/safe-casino-site/
https://images.google.mk/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.mn/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.ms/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.ms/url?q=https://betop24.com/safe-casino-site/
https://images.google.ms/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.mu/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.mu/url?q=https://betop24.com/safe-casino-site/
https://images.google.mu/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.mv/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.mv/url?q=https://betop24.com/safe-casino-site/
https://images.google.mv/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.mw/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.mw/url?q=https://betop24.com/safe-casino-site/
https://images.google.mw/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.ne/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.ne/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.nl/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.no/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.no/url?q=https://betop24.com/safe-casino-site/
https://images.google.no/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.nr/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.nr/url?q=https://betop24.com/safe-casino-site/
https://images.google.nr/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.nu/url?q=https://betop24.com/safe-casino-site/
https://images.google.nu/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.nu/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.pl/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.pn/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.pn/url?q=https://betop24.com/safe-casino-site/
https://images.google.pn/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.ps/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.ps/url?q=https://betop24.com/safe-casino-site/
https://images.google.ps/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.pt/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.ro/url?q=https://betop24.com/safe-casino-site/
https://images.google.rs/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.rs/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.ru/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.ru/url?q=https://betop24.com/safe-casino-site/
https://images.google.rw/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.rw/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.sc/url?q=https://betop24.com/safe-casino-site/
https://images.google.sc/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.se/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.se/url?q=https://betop24.com/safe-casino-site/
https://images.google.sh/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.sh/url?q=https://betop24.com/safe-casino-site/
https://images.google.sh/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.si/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.si/url?q=https://betop24.com/safe-casino-site/
https://images.google.sk/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.sk/url?q=https://betop24.com/safe-casino-site/
https://images.google.sm/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.sm/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.sn/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.sn/url?q=https://betop24.com/safe-casino-site/
https://images.google.sn/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.so/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.so/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.sr/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.sr/url?q=https://betop24.com/safe-casino-site/
https://images.google.sr/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.st/url?q=https://betop24.com/safe-casino-site/
https://images.google.st/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.st/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.td/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.td/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.tg/url?q=https://betop24.com/safe-casino-site/
https://images.google.tg/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.tg/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.tl/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.tl/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.tm/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.tn/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.to/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.tt/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.vg/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.vg/url?q=https://betop24.com/safe-casino-site/
https://images.google.vg/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.vu/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.vu/url?q=https://betop24.com/safe-casino-site/
https://images.google.vu/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.ws/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://inquiry.princetonreview.com/away/?value=cconntwit&category=FS&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://ipv4.google.com/url?q=https://betop24.com/safe-casino-site/
https://it.paltalk.com/client/webapp/client/External.wmt?url=http://https://betop24.com/safe-casino-site/
https://jump.2ch.net/?https://betop24.com/safe-casino-site/
https://lcu.hlcommission.org/lcu/pages/auth/forgotPassword.aspx?Returnurl=http://https://betop24.com/safe-casino-site/
https://legacy.aom.org/verifymember.asp?nextpage=https://betop24.com/safe-casino-site/
https://lexsrv2.nlm.nih.gov/fdse/search/search.pl?Match=0&Realm=All&Terms=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://m.addthis.com/live/redirect/?url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://m.odnoklassniki.ru/dk?st.cmd=outLinkWarning&st.rfn=https://betop24.com/safe-casino-site/
https://m.odnoklassniki.ru/dk?st.cmd=outLinkWarning&st.rfn=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://m.ok.ru/dk?st.cmd=outLinkWarning&st.rfn=https://betop24.com/safe-casino-site/
https://m.ok.ru/dk?st.cmd=outLinkWarning&st.rfn=http://https://betop24.com/safe-casino-site/
https://maps.google.ad/url?q=https://betop24.com/safe-casino-site/
https://maps.google.ad/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.ae/url?q=https://betop24.com/safe-casino-site/
https://maps.google.as/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.as/url?q=https://betop24.com/safe-casino-site/
https://maps.google.as/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.at/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.at/url?q=https://betop24.com/safe-casino-site/
https://maps.google.ba/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.ba/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.be/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.be/url?q=https://betop24.com/safe-casino-site/
https://maps.google.bf/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.bf/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.bg/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.bg/url?q=https://betop24.com/safe-casino-site/
https://maps.google.bi/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.bi/url?q=https://betop24.com/safe-casino-site/
https://maps.google.bj/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.bj/url?q=https://betop24.com/safe-casino-site/
https://maps.google.bj/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.bs/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.bs/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.bt/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.by/url?q=https://betop24.com/safe-casino-site/
https://maps.google.by/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.ca/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.cat/url?q=https://betop24.com/safe-casino-site/
https://maps.google.cat/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.cd/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.cf/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.cf/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.cg/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.cg/url?q=https://betop24.com/safe-casino-site/
https://maps.google.cg/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.ch/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.ch/url?q=https://betop24.com/safe-casino-site/
https://maps.google.ci/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.ci/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.cl/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.cl/url?q=https://betop24.com/safe-casino-site/
https://maps.google.cm/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.cm/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.co.ao/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.co.ao/url?q=https://betop24.com/safe-casino-site/
https://maps.google.co.ao/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.co.bw/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.co.bw/url?q=https://betop24.com/safe-casino-site/
https://maps.google.co.bw/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.co.ck/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.co.ck/url?q=https://betop24.com/safe-casino-site/
https://maps.google.co.ck/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.co.cr/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.co.cr/url?q=https://betop24.com/safe-casino-site/
https://maps.google.co.id/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.co.il/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.co.in/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.co.in/url?q=https://betop24.com/safe-casino-site/
https://maps.google.co.ke/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.co.kr/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.co.kr/url?q=https://betop24.com/safe-casino-site/
https://maps.google.co.ls/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.co.ls/url?q=https://betop24.com/safe-casino-site/
https://maps.google.co.ls/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.co.mz/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.co.mz/url?q=https://betop24.com/safe-casino-site/
https://maps.google.co.mz/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.co.nz/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.co.th/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.co.th/url?q=https://betop24.com/safe-casino-site/
https://maps.google.co.tz/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.co.tz/url?q=https://betop24.com/safe-casino-site/
https://maps.google.co.ug/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.co.ug/url?q=https://betop24.com/safe-casino-site/
https://maps.google.co.uk/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.co.ve/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.co.ve/url?q=https://betop24.com/safe-casino-site/
https://maps.google.co.vi/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.co.vi/url?q=https://betop24.com/safe-casino-site/
https://maps.google.co.vi/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.co.za/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.co.za/url?q=https://betop24.com/safe-casino-site/
https://maps.google.co.zm/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.co.zm/url?q=https://betop24.com/safe-casino-site/
https://maps.google.co.zm/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.co.zw/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.co.zw/url?q=https://betop24.com/safe-casino-site/
https://maps.google.com.ag/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.ag/url?q=https://betop24.com/safe-casino-site/
https://maps.google.com.ag/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.ai/url?q=https://betop24.com/safe-casino-site/
https://maps.google.com.ai/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.ai/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.ar/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.ar/url?q=https://betop24.com/safe-casino-site/
https://maps.google.com.ar/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.au/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.bd/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.bd/url?q=https://betop24.com/safe-casino-site/
https://maps.google.com.bd/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.bh/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.bh/url?q=https://betop24.com/safe-casino-site/
https://maps.google.com.bh/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.bn/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.bn/url?q=https://betop24.com/safe-casino-site/
https://maps.google.com.bn/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.bo/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.br/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.bz/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.bz/url?q=https://betop24.com/safe-casino-site/
https://maps.google.com.bz/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.co/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.co/url?q=https://betop24.com/safe-casino-site/
https://maps.google.com.cu/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.cu/url?q=https://betop24.com/safe-casino-site/
https://maps.google.com.cu/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.do/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.do/url?q=https://betop24.com/safe-casino-site/
https://maps.google.com.ec/url?q=https://betop24.com/safe-casino-site/
https://maps.google.com.eg/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.eg/url?q=https://betop24.com/safe-casino-site/
https://maps.google.com.fj/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.fj/url?q=https://betop24.com/safe-casino-site/
https://maps.google.com.fj/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.gh/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.gh/url?q=https://betop24.com/safe-casino-site/
https://maps.google.com.gh/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.gi/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.gi/url?q=https://betop24.com/safe-casino-site/
https://maps.google.com.gi/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.gt/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.gt/url?q=https://betop24.com/safe-casino-site/
https://maps.google.com.hk/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.hk/url?q=https://betop24.com/safe-casino-site/
https://maps.google.com.jm/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.jm/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.kh/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.kh/url?q=https://betop24.com/safe-casino-site/
https://maps.google.com.kh/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.kw/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.kw/url?q=https://betop24.com/safe-casino-site/
https://maps.google.com.kw/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.lb/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.ly/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.ly/url?q=https://betop24.com/safe-casino-site/
https://maps.google.com.ly/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.mm/url?q=https://betop24.com/safe-casino-site/
https://maps.google.com.mm/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.mt/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.mt/url?q=https://betop24.com/safe-casino-site/
https://maps.google.com.mt/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.mx/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.mx/url?q=https://betop24.com/safe-casino-site/
https://maps.google.com.my/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.my/url?q=https://betop24.com/safe-casino-site/
https://maps.google.com.na/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.na/url?q=https://betop24.com/safe-casino-site/
https://maps.google.com.na/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.ng/url?q=https://betop24.com/safe-casino-site/
https://maps.google.com.ni/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.ni/url?q=https://betop24.com/safe-casino-site/
https://maps.google.com.om/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.pa/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.pa/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.pe/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.pe/url?q=https://betop24.com/safe-casino-site/
https://maps.google.com.pg/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.ph/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.pr/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.pr/url?q=https://betop24.com/safe-casino-site/
https://maps.google.com.pr/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.py/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.py/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.qa/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.qa/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.sa/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.sa/url?q=https://betop24.com/safe-casino-site/
https://maps.google.com.sg/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.sg/url?q=https://betop24.com/safe-casino-site/
https://maps.google.com.sl/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.sl/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.sv/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.tr/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.tr/url?q=https://betop24.com/safe-casino-site/
https://maps.google.com.tw/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.tw/url?q=https://betop24.com/safe-casino-site/
https://maps.google.com.ua/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.ua/url?q=https://betop24.com/safe-casino-site/
https://maps.google.com.ua/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.uy/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.uy/url?q=https://betop24.com/safe-casino-site/
https://maps.google.com.uy/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.vc/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.vc/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.cv/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.cv/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.cz/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.cz/url?q=https://betop24.com/safe-casino-site/
https://maps.google.de/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.de/url?q=https://betop24.com/safe-casino-site/
https://maps.google.dj/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.dj/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.dk/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.dk/url?q=https://betop24.com/safe-casino-site/
https://maps.google.dm/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.dm/url?q=https://betop24.com/safe-casino-site/
https://maps.google.dm/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.dz/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.ee/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.ee/url?q=https://betop24.com/safe-casino-site/
https://maps.google.es/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.es/url?q=https://betop24.com/safe-casino-site/
https://maps.google.fi/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.fi/url?q=https://betop24.com/safe-casino-site/
https://maps.google.fm/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.fm/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.fr/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.fr/url?q=https://betop24.com/safe-casino-site/
https://maps.google.ga/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.ge/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.ge/url?q=https://betop24.com/safe-casino-site/
https://maps.google.gg/url?q=https://betop24.com/safe-casino-site/
https://maps.google.gg/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.gl/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.gl/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.gm/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.gm/url?q=https://betop24.com/safe-casino-site/
https://maps.google.gm/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.gp/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.gp/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.gr/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.gr/url?q=https://betop24.com/safe-casino-site/
https://maps.google.gy/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.gy/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.hn/url?q=https://betop24.com/safe-casino-site/
https://maps.google.hr/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.hr/url?q=https://betop24.com/safe-casino-site/
https://maps.google.ht/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.hu/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.hu/url?q=https://betop24.com/safe-casino-site/
https://maps.google.hu/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.ie/url?q=https://betop24.com/safe-casino-site/
https://maps.google.im/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.im/url?q=https://betop24.com/safe-casino-site/
https://maps.google.im/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.iq/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.is/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.is/url?q=https://betop24.com/safe-casino-site/
https://maps.google.is/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.it/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.it/url?q=https://betop24.com/safe-casino-site/
https://maps.google.je/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.je/url?q=https://betop24.com/safe-casino-site/
https://maps.google.je/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.jo/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.jo/url?q=https://betop24.com/safe-casino-site/
https://maps.google.jo/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.kg/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.ki/url?q=https://betop24.com/safe-casino-site/
https://maps.google.ki/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.ki/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.kz/url?q=https://betop24.com/safe-casino-site/
https://maps.google.kz/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.la/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.la/url?q=https://betop24.com/safe-casino-site/
https://maps.google.la/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.li/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.lk/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.lk/url?q=https://betop24.com/safe-casino-site/
https://maps.google.lt/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.lu/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.lu/url?q=https://betop24.com/safe-casino-site/
https://maps.google.lv/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.lv/url?q=https://betop24.com/safe-casino-site/
https://maps.google.mg/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.mg/url?q=https://betop24.com/safe-casino-site/
https://maps.google.mg/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.mk/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.mk/url?q=https://betop24.com/safe-casino-site/
https://maps.google.mk/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.mn/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.ms/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.ms/url?q=https://betop24.com/safe-casino-site/
http://www.fhwa.dot.gov/reauthorization/reauexit.cfm?link=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.fhwa.dot.gov/reauthorization/reauexit.cfm?link=https://betop24.com/safe-casino-site/
https://www.ladan.com.ua/link/go.php?url=http%3A%2F%2Fbetop24.com/safe-casino-site/%2F
https://www.webclap.com/php/jump.php?url=http%3A%2F%2Fbetop24.com/safe-casino-site/%2F
https://schwarzes-bw.de/wbb231/redir.php?url=https://betop24.com/safe-casino-site/
https://www.webclap.com/php/jump.php?url=https://betop24.com/safe-casino-site/
https://cse.google.com.cu/url?q=https://betop24.com/safe-casino-site/
http://www2.ogs.state.ny.us/help/urlstatusgo.html?url=http%3A%2F%2Fbetop24.com/safe-casino-site/
http://go.infomine.com/?re=121&tg=http%3A%2F%2Fbetop24.com/safe-casino-site/
http://link.harikonotora.net/?http%3A%2F%2Fbetop24.com/safe-casino-site/
http://text.usg.edu/tt/https://betop24.com/safe-casino-site/
http://uniportal.huaweidevice.com/uniportal/jsp/clearCrossDomainCookie.jsp?redirectUrl=http%3A%2F%2Fbetop24.com/safe-casino-site/
http://www.fito.nnov.ru/go.php?url=http%3A%2F%2Fbetop24.com/safe-casino-site/
http://www2.ogs.state.ny.us/help/urlstatusgo.html?url=https://betop24.com/safe-casino-site/
https://affiliates.bookdepository.com/scripts/click.php?a_aid=Alexa1202&a_bid=9abb5269&desturl=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://s5.histats.com/stats/r.php?869637&100&47794&urlr=&https://betop24.com/safe-casino-site/
https://15.pro.tok2.com/%7Ekoro/cutlinks/rank.php?url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://affiliates.bookdepository.com/scripts/click.php?a_aid=Alexa1202&a_bid=9abb5269&desturl=https://betop24.com/safe-casino-site/
https://affiliates.bookdepository.com/scripts/click.php?a_aid=Alexa1202&abid=9abb5269&desturl=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://channel.pixnet.net/newdirect.php?blog=cr19h3id&serial=0&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.cu/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://janeyleegrace.worldsecuresystems.com/redirect.aspx?destination=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://cse.google.ba/url?q=https://betop24.com/safe-casino-site/
https://cse.google.be/url?q=https://betop24.com/safe-casino-site/
https://cse.google.bg/url?q=https://betop24.com/safe-casino-site/
https://cse.google.bs/url?q=https://betop24.com/safe-casino-site/
https://cse.google.ca/url?q=https://betop24.com/safe-casino-site/
https://cse.google.cg/url?q=https://betop24.com/safe-casino-site/
https://cse.google.ch/url?q=https://betop24.com/safe-casino-site/
https://cse.google.cl/url?q=https://betop24.com/safe-casino-site/
https://cse.google.co.bw/url?q=https://betop24.com/safe-casino-site/
https://cse.google.co.cr/url?q=https://betop24.com/safe-casino-site/
https://cse.google.co.id/url?q=https://betop24.com/safe-casino-site/
https://cse.google.co.il/url?q=https://betop24.com/safe-casino-site/
https://cse.google.co.il/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.co.in/url?q=https://betop24.com/safe-casino-site/
https://cse.google.co.jp/url?q=https://betop24.com/safe-casino-site/
https://cse.google.co.ke/url?q=https://betop24.com/safe-casino-site/
https://cse.google.co.kr/url?q=https://betop24.com/safe-casino-site/
https://cse.google.co.ma/url?q=https://betop24.com/safe-casino-site/
https://cse.google.co.nz/url?q=https://betop24.com/safe-casino-site/
https://cse.google.co.th/url?q=https://betop24.com/safe-casino-site/
https://cse.google.co.ug/url?q=https://betop24.com/safe-casino-site/
https://cse.google.co.uk/url?q=https://betop24.com/safe-casino-site/
https://cse.google.co.uk/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.co.ve/url?q=https://betop24.com/safe-casino-site/
https://cse.google.co.za/url?q=https://betop24.com/safe-casino-site/
https://cse.google.com.af/url?q=https://betop24.com/safe-casino-site/
https://cse.google.com.au/url?q=https://betop24.com/safe-casino-site/
https://cse.google.com.bd/url?q=https://betop24.com/safe-casino-site/
https://cse.google.com.br/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.com.do/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.com.py/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.com.qa/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.com.sg/url?q=https://betop24.com/safe-casino-site/
https://cse.google.com.sv/url?q=https://betop24.com/safe-casino-site/
https://cse.google.com.tr/url?q=https://betop24.com/safe-casino-site/
https://cse.google.com.tw/url?q=https://betop24.com/safe-casino-site/
https://cse.google.com.ua/url?q=https://betop24.com/safe-casino-site/
https://cse.google.com.uy/url?q=https://betop24.com/safe-casino-site/
https://cse.google.com.vn/url?q=https://betop24.com/safe-casino-site/
https://cse.google.pt/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.rs/url?q=https://betop24.com/safe-casino-site/
https://cse.google.si/url?q=https://betop24.com/safe-casino-site/
https://cse.google.sk/url?q=https://betop24.com/safe-casino-site/
https://cse.google.tn/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.vg/url?q=https%3A%2F%2Fhttps://betop24.com/safe-casino-site/
https://images.google.at/url?sa=t&url=https://betop24.com/safe-casino-site/
https://images.google.be/url?sa=t&url=https://betop24.com/safe-casino-site/
https://images.google.bg/url?sa=t&url=https://betop24.com/safe-casino-site/
https://images.google.ca/url?q=https://betop24.com/safe-casino-site/
https://images.google.ca/url?sa=t&url=https://betop24.com/safe-casino-site/
https://images.google.ch/url?sa=t&url=https://betop24.com/safe-casino-site/
https://images.google.cl/url?sa=t&url=https://betop24.com/safe-casino-site/
https://images.google.co.id/url?sa=t&url=https://betop24.com/safe-casino-site/
https://images.google.co.il/url?sa=t&url=https://betop24.com/safe-casino-site/
https://images.google.co.in/url?sa=t&url=https://betop24.com/safe-casino-site/
https://images.google.co.jp/url?sa=t&url=https://betop24.com/safe-casino-site/
https://images.google.co.nz/url?sa=t&url=https://betop24.com/safe-casino-site/
https://images.google.co.th/url?sa=t&url=https://betop24.com/safe-casino-site/
https://images.google.co.uk/url?sa=t&url=https://betop24.com/safe-casino-site/
https://images.google.co.za/url?sa=t&url=https://betop24.com/safe-casino-site/
https://images.google.com.au/url?sa=t&url=https://betop24.com/safe-casino-site/
https://images.google.com.br/url?q=https://betop24.com/safe-casino-site/
https://images.google.com.br/url?sa=t&url=https://betop24.com/safe-casino-site/
https://images.google.com.cu/url?q=https://betop24.com/safe-casino-site/
https://images.google.com.hk/url?sa=t&url=https://betop24.com/safe-casino-site/
https://images.google.com.mx/url?sa=t&url=https://betop24.com/safe-casino-site/
https://images.google.com.my/url?sa=t&url=https://betop24.com/safe-casino-site/
https://images.google.com.sg/url?sa=t&url=https://betop24.com/safe-casino-site/
https://images.google.com.tr/url?sa=t&url=https://betop24.com/safe-casino-site/
https://images.google.com.tw/url?sa=t&url=https://betop24.com/safe-casino-site/
https://images.google.com.ua/url?sa=t&url=https://betop24.com/safe-casino-site/
https://images.google.com.vn/url?q=https://betop24.com/safe-casino-site/
https://images.google.com.vn/url?sa=t&url=https://betop24.com/safe-casino-site/
https://images.google.cz/url?sa=t&url=https://betop24.com/safe-casino-site/
https://images.google.de/url?sa=t&url=https://betop24.com/safe-casino-site/
https://images.google.dk/url?sa=t&url=https://betop24.com/safe-casino-site/
https://images.google.es/url?sa=t&url=https://betop24.com/safe-casino-site/
https://images.google.fi/url?sa=t&url=https://betop24.com/safe-casino-site/
https://images.google.fr/url?sa=t&url=https://betop24.com/safe-casino-site/
https://images.google.gr/url?sa=t&url=https://betop24.com/safe-casino-site/
https://images.google.hu/url?sa=t&url=https://betop24.com/safe-casino-site/
https://images.google.ie/url?sa=t&url=https://betop24.com/safe-casino-site/
https://images.google.nl/url?sa=t&url=https://betop24.com/safe-casino-site/
https://images.google.pl/url?sa=t&url=https://betop24.com/safe-casino-site/
https://images.google.pt/url?sa=t&url=https://betop24.com/safe-casino-site/
https://images.google.ro/url?sa=t&url=https://betop24.com/safe-casino-site/
https://images.google.ru/url?sa=t&url=https://betop24.com/safe-casino-site/
https://images.google.sk/url?sa=t&url=https://betop24.com/safe-casino-site/
https://maps.google.be/url?sa=t&url=https://betop24.com/safe-casino-site/
https://maps.google.ca/url?sa=t&url=https://betop24.com/safe-casino-site/
https://maps.google.ch/url?sa=t&url=https://betop24.com/safe-casino-site/
https://maps.google.cl/url?q=https://betop24.com/safe-casino-site/
https://maps.google.cl/url?sa=t&url=https://betop24.com/safe-casino-site/
https://maps.google.co.id/url?sa=t&url=https://betop24.com/safe-casino-site/
https://maps.google.co.id/url?sa=t&url=https://betop24.com/safe-casino-site%2F
https://maps.google.co.il/url?sa=t&url=https://betop24.com/safe-casino-site/
https://maps.google.co.in/url?sa=t&url=https://betop24.com/safe-casino-site/
https://maps.google.co.jp/url?sa=t&url=https://betop24.com/safe-casino-site/
https://maps.google.co.nz/url?sa=t&url=https://betop24.com/safe-casino-site/
https://maps.google.co.th/url?sa=t&url=https://betop24.com/safe-casino-site/
https://maps.google.co.uk/url?sa=t&url=https://betop24.com/safe-casino-site/
https://maps.google.co.za/url?sa=t&url=https://betop24.com/safe-casino-site/
https://maps.google.com.au/url?sa=t&url=https://betop24.com/safe-casino-site/
https://maps.google.com.br/url?q=https://betop24.com/safe-casino-site/
https://maps.google.com.hk/url?sa=t&url=https://betop24.com/safe-casino-site/
https://maps.google.com.mx/url?sa=t&url=https://betop24.com/safe-casino-site/
https://maps.google.com.my/url?sa=t&url=https://betop24.com/safe-casino-site/
https://maps.google.com.sg/url?sa=t&url=https://betop24.com/safe-casino-site/
https://maps.google.com.tr/url?sa=t&url=https://betop24.com/safe-casino-site/
https://maps.google.com.tw/url?sa=t&url=https://betop24.com/safe-casino-site/
https://maps.google.com.tw/url?sa=t&url=https://betop24.com/safe-casino-site%2F
https://maps.google.com.ua/url?sa=t&url=https://betop24.com/safe-casino-site/
https://maps.google.com/url?sa=t&url=https://betop24.com/safe-casino-site/
https://maps.google.cz/url?sa=t&url=https://betop24.com/safe-casino-site/
https://maps.google.de/url?sa=t&url=https://betop24.com/safe-casino-site/
https://maps.google.dk/url?sa=t&url=https://betop24.com/safe-casino-site/
https://maps.google.es/url?sa=t&url=https://betop24.com/safe-casino-site/
https://maps.google.fi/url?sa=t&url=https://betop24.com/safe-casino-site/
https://maps.google.fr/url?sa=t&url=https://betop24.com/safe-casino-site/
https://maps.google.gr/url?sa=t&url=https://betop24.com/safe-casino-site/
https://maps.google.hu/url?sa=t&url=https://betop24.com/safe-casino-site/
https://maps.google.ie/url?sa=t&url=https://betop24.com/safe-casino-site/
https://maps.google.nl/url?sa=t&url=https://betop24.com/safe-casino-site/
https://maps.google.pl/url?sa=t&url=https://betop24.com/safe-casino-site/
https://maps.google.pt/url?sa=t&url=https://betop24.com/safe-casino-site/
https://maps.google.ro/url?sa=t&url=https://betop24.com/safe-casino-site/
https://maps.google.ru/url?sa=t&url=https://betop24.com/safe-casino-site/
https://maps.google.sk/url?sa=t&url=https://betop24.com/safe-casino-site/
https://www.google.at/url?q=https://betop24.com/safe-casino-site/
https://www.google.co.id/url?q=https://betop24.com/safe-casino-site/
https://www.google.co.il/url?q=https://betop24.com/safe-casino-site/
https://www.google.co.jp/url?q=https://betop24.com/safe-casino-site/
https://www.google.co.kr/url?q=https://betop24.com/safe-casino-site/
https://www.google.co.uk/url?q=https://betop24.com/safe-casino-site/
https://www.google.co.za/url?q=https://betop24.com/safe-casino-site/
https://www.google.com.ar/url?q=https://betop24.com/safe-casino-site/
https://www.google.com.br/url?q=https://betop24.com/safe-casino-site/
https://www.google.com.mx/url?q=https://betop24.com/safe-casino-site/
https://www.google.com.my/url?q=https://betop24.com/safe-casino-site/
https://www.google.com.pe/url?q=https://betop24.com/safe-casino-site/
https://www.google.com.ph/url?q=https://betop24.com/safe-casino-site/
https://www.google.com.tw/url?q=https://betop24.com/safe-casino-site/
https://www.google.com.ua/url?q=https://betop24.com/safe-casino-site/
https://www.google.com.vn/url?q=https://betop24.com/safe-casino-site/
https://www.google.com/url?q=https://betop24.com/safe-casino-site/
https://www.google.de/url?sa=t&url=https://betop24.com/safe-casino-site/
https://www.google.hu/url?q=https://betop24.com/safe-casino-site/
https://www.google.ie/url?q=https://betop24.com/safe-casino-site/
https://www.google.it/url?q=https://betop24.com/safe-casino-site/
https://www.google.pt/url?q=https://betop24.com/safe-casino-site/
https://www.google.ro/url?q=https://betop24.com/safe-casino-site/
https://www.bing.com/news/apiclick.aspx?ref=FexRss&aid=&tid=9BB77FDA801248A5AD23FDBDD5922800&url=https://betop24.com/safe-casino-site/
https://www.google.ae/url?q=https://betop24.com/safe-casino-site/
https://www.google.az/url?q=https://betop24.com/safe-casino-site/
https://www.google.bs/url?q=https://betop24.com/safe-casino-site/
https://www.google.co.bw/url?q=https://betop24.com/safe-casino-site/
https://www.google.co.ke/url?q=https://betop24.com/safe-casino-site/
https://www.google.co.ve/url?q=https://betop24.com/safe-casino-site/
https://www.google.com.bd/url?q=https://betop24.com/safe-casino-site/
https://www.google.com.bh/url?q=https://betop24.com/safe-casino-site/
https://www.google.com.cy/url?q=https://betop24.com/safe-casino-site/
https://www.google.com.ec/url?q=https://betop24.com/safe-casino-site/
https://www.google.com.ng/url?q=https://betop24.com/safe-casino-site/
https://www.google.com.np/url?q=https://betop24.com/safe-casino-site/
https://www.google.com.pk/url?q=https://betop24.com/safe-casino-site/
https://www.google.com.qa/url?q=https://betop24.com/safe-casino-site/
https://www.google.lu/url?q=https://betop24.com/safe-casino-site/
https://club.panasonic.jp/member/terms/?siteId=B1&returnURL=https://betop24.com/safe-casino-site/
https://keywordspace.com/site-info/betop24.com
https://www.bombstat.com/domain/betop24.com
https://maps.google.com.br/url?sa=t&url=https://betop24.com/safe-casino-site/
https://www.google.jo/url?q=https://betop24.com/safe-casino-site/
https://www.google.com.ly/url?q=https://betop24.com/safe-casino-site/
https://cse.google.mu/url?q=https://betop24.com/safe-casino-site/
https://cse.google.ml/url?q=https://betop24.com/safe-casino-site/
https://www.google.de/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.co.uk/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.fr/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.fr/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.nl/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.ch/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.be/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.com.tr/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.com.mx/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.dk/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.pt/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.hr/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.lv/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.com.pr/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.lu/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.lu/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.co.ma/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.mn/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.com.kh/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.co.ug/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.ly/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
http://tyonabi.sakura.ne.jp/link/cgi-bin/out.cgi?id=dorian362&cg=1&siteurl=https://betop24.com/safe-casino-site/
http://feeds.copyblogger.com/%7E/t/0/0/copyblogger/%7Ehttp%3A%2F%2Fbetop24.com/safe-casino-site/
http://www.scoop.co.nz/link-out?p=blog93&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://cse.google.be/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.bf/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.co.ck/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.com.bz/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.com.om/url?q=https://betop24.com/safe-casino-site/
https://cse.google.com.pk/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.com.sl/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.com.vn/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.dm/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.ge/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.iq/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.pl/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.pn/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.so/url?sa=i&url=https://betop24.com/safe-casino-site/
https://images.google.co.cr/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.pe/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.li/url?q=https://betop24.com/safe-casino-site/
https://lcu.hlcommission.org/lcu/pages/auth/forgotPassword.aspx?Returnurl=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.bo/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.lb/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.np/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.sa/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.de/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.ee/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.ie/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.nl/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.tn/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://t.me/iv?url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://transtats.bts.gov/exit.asp?url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.by/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.es/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.pl/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://www.google.sk/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://a.pr-cy.ru/https://betop24.com/safe-casino-site/
https://client.paltalk.com/client/webapp/client/External.wmt?url=https://betop24.com/safe-casino-site/
https://clients1.google.com.et/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.com.nf/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.com.vc/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.je/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.lk/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://clients1.google.mk/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://cse.google.co.kr/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://cse.google.fr/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://doodle.com/r?url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://galter.northwestern.edu/exit?url=https://betop24.com/safe-casino-site/
https://images.google.bg/url?q=https://betop24.com/safe-casino-site/
https://images.google.bs/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.cl/url?q=https://betop24.com/safe-casino-site/
https://images.google.co.bw/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.in/url?q=https://betop24.com/safe-casino-site/
https://images.google.co.ma/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.th/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.co.uz/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.bd/url?q=https://betop24.com/safe-casino-site/
https://images.google.com.br/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.bz/url?q=https://betop24.com/safe-casino-site/
https://images.google.com.cu/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.ec/url?q=https://betop24.com/safe-casino-site/
https://images.google.com.fj/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.gh/url?q=https://betop24.com/safe-casino-site/
https://images.google.com.gi/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.gi/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.ni/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.com.tw/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.cz/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.gy/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.is/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.kz/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.lt/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.me/url?q=https://betop24.com/safe-casino-site/
https://images.google.ro/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://images.google.rs/url?q=https://betop24.com/safe-casino-site/
https://images.google.sm/url?q=https://betop24.com/safe-casino-site/
https://images.google.to/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://ipv4.google.com/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.ad/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.ae/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.bi/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.cat/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.cd/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.cd/url?q=https://betop24.com/safe-casino-site/
https://maps.google.ci/url?q=https://betop24.com/safe-casino-site/
https://maps.google.cm/url?q=https://betop24.com/safe-casino-site/
https://maps.google.co.id/url?q=https://betop24.com/safe-casino-site/
https://maps.google.co.jp/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.co.tz/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.co.zw/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.ec/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.et/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.mm/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.ng/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.np/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.np/url?q=https://betop24.com/safe-casino-site/
https://maps.google.com.pg/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com.ph/url?q=https://betop24.com/safe-casino-site/
https://maps.google.com.sv/url?q=https://betop24.com/safe-casino-site/
https://maps.google.com.sv/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.com/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.ga/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.gg/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.hn/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.ht/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.ht/url?q=https://betop24.com/safe-casino-site/
https://maps.google.ie/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.kz/url?q=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://maps.google.ms/url?sa=t&url=http%3A%2F%2Fbetop24.com/safe-casino-site/
https://sitereport.netcraft.com/?url=https://betop24.com/safe-casino-site%2F
https://ranking.crawler.com/SiteInfo.aspx?url=https://betop24.com/safe-casino-site/
https://valueanalyze.com/show.php?url=https://betop24.com/safe-casino-site/
http://toolbarqueries.google.com.uy/url?sa=t&url=https://betop24.com/safe-casino-site/
http://toolbarqueries.google.com.tw/url?sa=t&url=https://betop24.com/safe-casino-site/
http://toolbarqueries.google.com.tr/url?sa=t&url=https://betop24.com/safe-casino-site/
http://toolbarqueries.google.com.sa/url?sa=t&url=https://betop24.com/safe-casino-site/
http://toolbarqueries.google.com.py/url?sa=t&url=https://betop24.com/safe-casino-site/
http://toolbarqueries.google.com.pr/url?sa=t&url=https://betop24.com/safe-casino-site/
http://toolbarqueries.google.com.pk/url?sa=t&url=https://betop24.com/safe-casino-site/
http://toolbarqueries.google.com.pe/url?sa=t&url=https://betop24.com/safe-casino-site/
http://toolbarqueries.google.com.my/url?sa=t&url=https://betop24.com/safe-casino-site/
http://toolbarqueries.google.com.hk/url?sa=t&url=https://betop24.com/safe-casino-site/
http://toolbarqueries.google.com.gt/url?sa=t&url=https://betop24.com/safe-casino-site/
http://toolbarqueries.google.com.gh/url?sa=t&url=https://betop24.com/safe-casino-site/
https://clients1.google.com.ar/url?sa=t&url=https://betop24.com/safe-casino-site/
https://clients1.google.com.ag/url?sa=t&url=https://betop24.com/safe-casino-site/
https://clients1.google.co.zm/url?sa=t&url=https://betop24.com/safe-casino-site/
https://clients1.google.co.za/url?sa=t&url=https://betop24.com/safe-casino-site/
https://clients1.google.co.ve/url?sa=t&url=https://betop24.com/safe-casino-site/
https://clients1.google.co.uz/url?sa=t&url=https://betop24.com/safe-casino-site/
https://clients1.google.co.ug/url?sa=t&url=https://betop24.com/safe-casino-site/
https://clients1.google.co.th/url?sa=t&url=https://betop24.com/safe-casino-site/
https://clients1.google.co.nz/url?sa=t&url=https://betop24.com/safe-casino-site/
https://clients1.google.co.kr/url?sa=t&url=https://betop24.com/safe-casino-site/
https://clients1.google.co.ke/url?sa=t&url=https://betop24.com/safe-casino-site/
https://clients1.google.co.il/url?sa=t&url=https://betop24.com/safe-casino-site/
https://clients1.google.co.id/url?sa=t&url=https://betop24.com/safe-casino-site/
https://clients1.google.co.cr/url?sa=t&url=https://betop24.com/safe-casino-site/
https://clients1.google.co.ck/url?sa=t&url=https://betop24.com/safe-casino-site/
https://cse.google.co.ck/url?sa=t&url=https://betop24.com/safe-casino-site/
https://cse.google.co.bw/url?sa=t&url=https://betop24.com/safe-casino-site/
https://cse.google.cm/url?sa=t&url=https://betop24.com/safe-casino-site/
https://cse.google.cl/url?sa=t&url=https://betop24.com/safe-casino-site/
https://cse.google.ci/url?sa=t&url=https://betop24.com/safe-casino-site/
https://cse.google.ch/url?sa=t&url=https://betop24.com/safe-casino-site/
https://cse.google.ch/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.cg/url?sa=t&url=https://betop24.com/safe-casino-site/
https://cse.google.cd/url?sa=t&url=https://betop24.com/safe-casino-site/
https://cse.google.by/url?sa=t&url=https://betop24.com/safe-casino-site/
https://cse.google.bs/url?sa=t&url=https://betop24.com/safe-casino-site/
https://cse.google.bi/url?sa=t&url=https://betop24.com/safe-casino-site/
https://cse.google.bg/url?sa=t&url=https://betop24.com/safe-casino-site/
https://cse.google.be/url?sa=t&url=https://betop24.com/safe-casino-site/
https://cse.google.be/url?sa=i&url=https://betop24.com/safe-casino-site/
https://cse.google.ba/url?sa=t&url=https://betop24.com/safe-casino-site/
https://cse.google.az/url?sa=t&url=https://betop24.com/safe-casino-site/
https://cse.google.at/url?sa=t&url=https://betop24.com/safe-casino-site/
https://images.google.ca/url?sa=t&url=https://betop24.com/safe-casino-site/
https://images.google.by/url?sa=t&url=https://betop24.com/safe-casino-site/
https://images.google.bs/url?sa=t&url=https://betop24.com/safe-casino-site/
https://images.google.bi/url?sa=t&url=https://betop24.com/safe-casino-site/
https://images.google.bg/url?sa=t&url=https://betop24.com/safe-casino-site/
https://images.google.bf/url?sa=t&url=https://betop24.com/safe-casino-site/
https://images.google.be/url?sa=t&url=https://betop24.com/safe-casino-site/
https://images.google.ba/url?sa=t&url=https://betop24.com/safe-casino-site/
https://images.google.ba/url?q=https://betop24.com/safe-casino-site/
https://images.google.az/url?sa=t&url=https://betop24.com/safe-casino-site/
https://images.google.at/url?sa=t&url=https://betop24.com/safe-casino-site/
https://images.google.as/url?sa=t&url=https://betop24.com/safe-casino-site/
https://images.google.am/url?sa=t&url=https://betop24.com/safe-casino-site/
https://images.google.al/url?sa=t&url=https://betop24.com/safe-casino-site/
https://images.google.ae/url?sa=t&url=https://betop24.com/safe-casino-site/
https://images.google.ae/url?q=https://betop24.com/safe-casino-site/
https://images.google.ad/url?sa=t&url=https://betop24.com/safe-casino-site/
https://maps.google.co.ke/url?sa=t&url=https://betop24.com/safe-casino-site/
https://maps.google.co.jp/url?sa=t&url=https://betop24.com/safe-casino-site/
https://maps.google.co.in/url?sa=t&url=https://betop24.com/safe-casino-site/
https://maps.google.co.il/url?sa=t&url=https://betop24.com/safe-casino-site/
https://maps.google.co.id/url?sa=t&url=https://betop24.com/safe-casino-site/
https://maps.google.co.cr/url?sa=t&url=https://betop24.com/safe-casino-site/
https://maps.google.co.bw/url?sa=t&url=https://betop24.com/safe-casino-site/
https://maps.google.cm/url?sa=t&url=https://betop24.com/safe-casino-site/
https://maps.google.cl/url?sa=t&url=https://betop24.com/safe-casino-site/
https://maps.google.ci/url?sa=t&url=https://betop24.com/safe-casino-site/
https://maps.google.ch/url?sa=t&url=https://betop24.com/safe-casino-site/
https://maps.google.cd/url?sa=t&url=https://betop24.com/safe-casino-site/
https://maps.google.cat/url?sa=t&url=https://betop24.com/safe-casino-site/
https://maps.google.ca/url?sa=t&url=https://betop24.com/safe-casino-site/
https://www.google.co.tz/url?sa=t&url=https://betop24.com/safe-casino-site/
https://www.google.co.th/url?sa=t&url=https://betop24.com/safe-casino-site/
https://www.google.co.nz/url?sa=t&url=https://betop24.com/safe-casino-site/
https://www.google.co.ma/url?sa=t&url=https://betop24.com/safe-casino-site/
https://www.google.co.ls/url?sa=t&url=https://betop24.com/safe-casino-site/
https://www.google.co.kr/url?sa=t&url=https://betop24.com/safe-casino-site/
https://www.google.co.ke/url?sa=t&url=https://betop24.com/safe-casino-site/
https://www.google.co.jp/url?sa=t&url=https://betop24.com/safe-casino-site/
https://www.google.co.in/url?sa=t&url=https://betop24.com/safe-casino-site/
https://www.google.co.il/url?sa=t&url=https://betop24.com/safe-casino-site/
https://www.google.co.id/url?sa=t&url=https://betop24.com/safe-casino-site/
https://www.google.co.cr/url?sa=t&url=https://betop24.com/safe-casino-site/
https://www.google.co.bw/url?sa=t&url=https://betop24.com/safe-casino-site/
https://www.google.cm/url?sa=t&url=https://betop24.com/safe-casino-site/
https://www.google.cl/url?sa=t&url=https://betop24.com/safe-casino-site/
https://www.google.ci/url?sa=t&url=https://betop24.com/safe-casino-site/
https://www.google.ch/url?sa=t&url=https://betop24.com/safe-casino-site/
https://www.google.cd/url?sa=t&url=https://betop24.com/safe-casino-site/
https://www.google.cat/url?sa=t&url=https://betop24.com/safe-casino-site/
https://www.google.ca/url?sa=t&url=https://betop24.com/safe-casino-site/
http://toolbarqueries.google.com.uy/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://toolbarqueries.google.com.tw/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://toolbarqueries.google.com.tr/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://toolbarqueries.google.com.sa/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://toolbarqueries.google.com.py/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://toolbarqueries.google.com.pr/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://toolbarqueries.google.com.pk/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://toolbarqueries.google.com.pe/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://toolbarqueries.google.com.my/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://toolbarqueries.google.com.hk/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://toolbarqueries.google.com.gt/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://toolbarqueries.google.com.gh/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.com.ar/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.com.ag/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.co.zm/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.co.za/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.co.ve/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.co.uz/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.co.ug/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.co.th/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.co.nz/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.co.kr/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.co.ke/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.co.il/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.co.id/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.co.cr/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.co.ck/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.co.ck/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.co.bw/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.cm/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.cl/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.ci/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.ch/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.ch/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.cg/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.cd/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.by/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.bs/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.bi/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.bg/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.be/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.be/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.ba/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.az/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.at/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ca/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.by/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.bs/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.bi/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.bg/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.bf/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.be/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ba/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ba/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.az/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.at/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.as/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.am/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.al/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ae/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ae/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ad/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.ke/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.jp/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.in/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.il/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.id/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.cr/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.bw/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.cm/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.cl/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ci/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ch/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.cd/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.cat/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ca/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.co.tz/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.co.th/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.co.nz/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.co.ma/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.co.ls/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.co.kr/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.co.ke/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.co.jp/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.co.in/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.co.il/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.co.id/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.co.cr/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.co.bw/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.cm/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.cl/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.ci/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.ch/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.cd/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.cat/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.ca/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.co.zw/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html%2F
https://www.adminer.org/redirect/?url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://sfwater.org/redirect.aspx?url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://web.archive.org/web/20180804180141/https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://baoviet.com.vn/Redirect.aspx?url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://sc.sie.gov.hk/TuniS/https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.ric.edu/Pages/link_out.aspx?target=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.pe/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.pk/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.np/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.jm/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.li/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.kh/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.co/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.cm/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.gi/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.la/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.tk/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.co.mz/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.sb/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.sh/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.ly/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.fm/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.vu/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.cf/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.tj/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.cv/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.st/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.sr/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.iq/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.tk/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://posts.google.com/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.nu/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.com/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://asia.google.com/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.de/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.de/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.uk/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.co.uk/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.jp/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.jp/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.co.jp/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.fr/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.es/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.es/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.es/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.it/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.it/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.it/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.br/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.br/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.br/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ca/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ca/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.ca/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.hk/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.hk/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.hk/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.nl/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.nl/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.in/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.in/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.co.in/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ru/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.ru/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ru/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.pl/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.pl/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.pl/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.au/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.au/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.au/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.tw/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.tw/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.tw/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.id/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.id/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.co.id/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ch/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.ch/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.be/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.be/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.at/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.at/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.at/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.cz/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.cz/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.th/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.th/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.co.th/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.ua/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.ua/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.ua/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.tr/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.tr/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.mx/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.mx/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.dk/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.dk/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.hu/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.hu/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.hu/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.fi/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.fi/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.fi/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.nz/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.co.nz/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.nz/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.vn/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.vn/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.pt/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.pt/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ro/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.ro/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ro/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.my/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.my/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.my/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.co.za/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.za/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.za/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.sg/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.sg/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.sg/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.gr/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.gr/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.gr/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.il/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.il/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.co.il/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.cl/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.cl/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.cl/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ie/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.ie/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ie/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.sk/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.sk/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.sk/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.bg/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.bg/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.bg/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.pe/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.pe/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ae/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ae/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.ae/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.pk/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.pk/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.co/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.co/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.co/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.eg/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.eg/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.eg/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.lt/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.sa/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.sa/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.sa/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.hr/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.hr/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.co.ve/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.ve/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.ve/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.ee/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ee/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ee/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.si/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.si/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.si/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.by/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.ec/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.ec/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.lv/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.lv/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ba/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.ng/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.ng/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.gt/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.gt/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.cr/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.cr/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.lu/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.kw/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.kw/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.is/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.dz/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.tn/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.tn/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.iq/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.iq/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.cm/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.bd/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.bd/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.do/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.do/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.do/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.kz/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ge/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.lk/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.ni/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.la/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.jm/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.cy/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.qa/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.sv/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.sv/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ps/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.bo/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.li/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.mn/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.bh/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.bh/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.kh/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.lb/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.lb/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.tt/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.tt/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.ci/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.hn/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.hn/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.sn/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.sn/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.mt/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.mt/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.cat/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.bi/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.bi/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.cd/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.fm/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.fm/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.fm/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.as/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.pa/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.pa/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.om/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.om/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.mz/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.co.bw/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.ms/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.kg/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.bz/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.bz/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.bn/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.sh/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.ag/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.ag/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://app.feedblitz.com/f/f.fbz?track=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://archives.midweek.com/?URL=http%253A%252F%252Fhttps://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://fooyoh.com/wcn.php?url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html&keyid=42973
http://m.odnoklassniki.ru/dk?st.cmd=outLinkWarning&st.rfn=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://onlinemanuals.txdot.gov/help/urlstatusgo.html?url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://onlinemanuals.txdot.gov/help/urlstatusgo.html?url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://sc.sie.gov.hk/TuniS/https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://www.aa.org/pages/en_US/disclaimer?u=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://www.aastocks.com/sc/changelang.aspx?lang=sc&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://www.blueletterbible.org/tools/redirect.cfm?Site=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://www.cineuropa.org/el.aspx?el=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://www.drinksmixer.com/redirect.php?url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://www.justjared.com/flagcomment.php?el=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://www.kaskus.co.id/redirect?url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://www.meetme.com/apps/redirect/?url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://www.office.xerox.com/perl-bin/reseller_exit.pl?url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://www.popcouncil.org/scripts/leaving.asp?URL=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://www.ric.edu/Pages/link_out.aspx?target=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://www.sinp.msu.ru/ru/ext_link?url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://www.webclap.com/php/jump.php?url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://www2.sandbox.google.co.jp/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://www2.sandbox.google.co.kr/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://cr.naver.com/rd?u=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://devicedoctor.com/driver-feedback.php?device=PCI%20bus&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://domain.opendns.com/https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://my.dek-d.com/username/link/link.php?out=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://panchodeaonori.sakura.ne.jp/feed/aonori/feed2js.php?src=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://refer.ash1.ccbill.com/cgi-bin/clicks.cgi?CA=933914&PA=1785830&HTML=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://refer.ccbill.com/cgi-bin/clicks.cgi/http:/?CA=928498&PA=1458253&HTML=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://sc.youth.gov.hk/TuniS/https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://wasearch.loc.gov/e2k/*/https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://wikimapia.org/external_link?url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://www.astro.wisc.edu/?URL=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://www.astro.wisc.edu/?URL=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://www.cssdrive.com/?URL=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://www.earth-policy.org/?URL=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://www.fmvz.unam.mx/fmvz/biblioteca/revistas_electronicas/control/envioClics.php?cnH57w=1118&kqJm89=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://www.ric.edu/Pages/link_out.aspx?target=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://www.shinobi.jp/etc/goto.html?http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.htmlkuttymovies-2020-kuttymovies-hd-tamil-movies-download
http://www.spiritfanfiction.com/link?l=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://www.webclap.com/php/jump.php?url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://www.zerocarts.com/demo/index.php?url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://advisor.wmtransfer.com/SiteDetails.aspx?url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://blog.ss-blog.jp/pages/mobile/step/index?u=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://blogs.rtve.es/libs/getfirma_footer_prod.php?blogurl=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://bukkit.org/proxy.php?link=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://client.paltalk.com/client/webapp/client/External.wmt?url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://club.panasonic.jp/member/terms/?siteId=B1&returnURL=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://community.cypress.com/external-link.jspa?url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://community.esri.com/external-link.jspa?url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://community.rsa.com/external-link.jspa?url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.ac/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.ad/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.ad/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.ae/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.al/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.am/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.as/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.at/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.az/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.ba/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.bg/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.bi/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.bi/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.bj/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.bs/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.bt/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.bt/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.by/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.by/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.ca/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.cat/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.cd/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.cd/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.cf/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.cg/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.ch/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.ci/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.cl/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.cm/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.co.ao/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.co.bw/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.co.ck/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.co.cr/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.co.id/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.co.in/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.co.jp/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.co.ke/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.co.kr/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.co.ls/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.co.ma/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.co.mz/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.co.nz/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.co.th/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.co.tz/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.co.ug/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.co.uz/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.co.uz/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.co.ve/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.co.vi/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.co.za/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.co.zm/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.co.zw/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.af/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.ag/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.ai/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.ai/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.au/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.bd/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.bh/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.bn/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.bo/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.co/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.co/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.cu/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.cy/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.ec/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.eg/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.et/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.et/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.fj/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.gh/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.gi/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.gt/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.hk/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.jm/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.kh/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.kw/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.ly/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.mm/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.mt/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.mt/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.mx/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.my/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.na/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.nf/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.ng/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.ni/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.np/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.om/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.pa/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.pe/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.pg/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.ph/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.pr/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.sa/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.sa/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.sg/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.sv/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.tj/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.tr/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.tw/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.ua/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.uy/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.vc/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.cz/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.de/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.dk/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.dz/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.dz/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.ee/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.es/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.fi/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.fr/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.ga/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.ga/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.gg/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.gl/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.gm/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.gp/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.gr/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.gy/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.hn/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.hr/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.ht/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.hu/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.ie/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.im/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.iq/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.is/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.it/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.je/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.jo/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.kg/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.kg/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.ki/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.ki/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.kz/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.la/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.li/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.lk/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.lt/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.lu/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.lv/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.me/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.mg/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.mk/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.mn/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.mn/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.ms/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.mu/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.mv/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.mw/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.ne/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.nl/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.no/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.nr/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.nu/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.nu/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.ps/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.ro/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.rs/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.ru/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.rw/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.sc/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.se/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.sh/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.si/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.sk/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.sm/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.sn/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.so/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.sr/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.td/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.tg/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.tg/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.tl/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.tm/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.tm/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.tn/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.to/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.tt/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.vg/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.vu/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.ws/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.ws/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://ditu.google.com/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://foro.infojardin.com/proxy.php?link=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://foro.infojardin.com/proxy.php?link=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://forum.solidworks.com/external-link.jspa?url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://galter.northwestern.edu/exit?url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://id.telstra.com.au/register/crowdsupport?gotoURL=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ad/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ad/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ae/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.at/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.be/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.bg/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.bs/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.bt/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.bt/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.by/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.by/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ca/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.cat/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ch/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.cl/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.bw/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.id/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.il/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.in/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.jp/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.kr/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.nz/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.th/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.ug/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.ug/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.uk/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.uz/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.uz/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.ve/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.za/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.au/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.bd/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.br/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.co/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.do/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.ec/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.eg/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.et/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.et/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.gt/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.hk/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.jm/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.jm/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.kh/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.lb/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.mx/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.my/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.ng/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.np/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.om/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.om/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.pe/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.pe/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.ph/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.pk/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.pk/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.sa/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.sa/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.sa/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.sg/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.sv/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.tj/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.tr/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.tw/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.ua/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.uy/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.vn/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.cz/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.de/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.dk/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.dm/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.dz/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.dz/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ee/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.es/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.fi/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.fr/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.gr/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.hr/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.hu/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ie/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.iq/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.iq/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.it/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.kg/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.kg/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.lk/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.lt/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.lu/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.lv/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.mn/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.mn/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.nl/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.pl/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.pt/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ro/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ru/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.sc/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.se/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.si/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.sk/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.tm/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.tm/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.tn/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.tn/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.tt/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ws/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ws/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://legacy.aom.org/verifymember.asp?nextpage=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://m.ok.ru/dk?st.cmd=outLinkWarning&st.rfn=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ae/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.at/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.be/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.bg/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.bs/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.bt/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.bt/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.by/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ca/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ch/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.cl/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.cr/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.id/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.il/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.in/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.jp/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.ke/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.kr/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.nz/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.th/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.ug/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.uk/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.ve/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.za/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.au/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.br/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.co/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.do/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.ec/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.eg/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.et/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.et/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.gt/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.hk/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.jm/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.mx/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.my/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.ng/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.ni/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.om/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.om/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.pe/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.ph/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.sg/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.tr/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.tw/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.cz/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.dk/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.dz/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.dz/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.es/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.fi/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.fr/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ge/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.gr/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.hn/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.hr/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.iq/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.iq/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.it/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.kg/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.kg/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.li/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.li/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.lk/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.lt/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.lu/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.lv/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.mn/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.mn/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.pl/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.pt/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ro/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ru/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.sc/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.se/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.si/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.sk/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.tn/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.tn/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.vu/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.vu/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ws/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ws/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://optimize.viglink.com/page/pmv?url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://plus.google.com/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://pw.mail.ru/forums/fredirect.php?url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://ref.gamer.com.tw/redir.php?url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://rspcb.safety.fhwa.dot.gov/pageRedirect.aspx?RedirectedURL=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://sfwater.org/redirect.aspx?url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://sfwater.org/redirect.aspx?url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://wfc2.wiredforchange.com/dia/track.jsp?v=2&c=hdorrh%2BHcDlQ%2BzUEnZU5qlfKZ1Cl53X6&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.adminer.org/redirect/?url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.ae/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.as/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.at/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.be/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.bg/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.by/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.ca/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.ch/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.cl/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.co.bw/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.co.id/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.co.il/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.co.in/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.co.jp/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.co.ke/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.co.kr/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.co.ma/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.co.nz/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.co.th/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.co.uk/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.co.ve/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.co.za/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.au/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.bd/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.br/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.co/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.do/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.ec/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.eg/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.hk/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.mx/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.my/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.ng/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.np/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.pe/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.ph/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.pk/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.pr/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.sa/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.sg/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.sv/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.tr/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.tw/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.ua/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.uy/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.vn/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.cz/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.de/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.dk/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.dz/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.ee/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.fi/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.fr/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.gr/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.hn/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.hr/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.ie/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.is/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.it/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.kg/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.kg/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.kz/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.lu/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.lv/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.nl/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.pt/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.ro/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.ru/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.se/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.si/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.tn/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.tt/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.transtats.bts.gov/exit.asp?url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.transtats.bts.gov/exit.asp?url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.youtube.com/redirect?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://youtube.com/redirect?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://advisor.wmtransfer.com/SiteDetails.aspx?url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://advisor.wmtransfer.com/SiteDetails.aspx?url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html&tab=feedback
https://advisor.wmtransfer.com/SiteDetails.aspx?url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html&tab=rating
https://advisor.wmtransfer.com/SiteDetails.aspx?url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html&tab=wminfo
https://advisor.wmtransfer.com/SiteDetails.aspx?url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://anonym.to/?https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://anonym.to/?http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://blogs.rtve.es/libs/getfirma_footer_prod.php?blogurl=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://blogs.rtve.es/libs/getfirma_footer_prod.php?blogurl=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://bukkit.org/proxy.php?link=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://bukkit.org/proxy.php?link=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cdrinfo.com/Sections/Ads/ReviewsAroundTheWebRedirector.aspx?TargetUrl=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://ceskapozice.lidovky.cz/redir.aspx?url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cgl.ethz.ch/disclaimer.php?dlurl=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://chofu.keizai.biz/banner.php?type=text_banner&position=right&id=3&uri=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.ac/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.ad/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.ae/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.al/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.am/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.as/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.az/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.bf/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.bi/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.bj/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.bs/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.bt/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.by/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.cd/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.cf/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.cg/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.ci/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.cm/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.co.ao/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.co.ck/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.co.il/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.co.in/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.co.jp/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.co.kr/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.co.ls/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.co.ma/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.co.mz/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.co.nz/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.co.th/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.co.tz/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.co.uz/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.co.ve/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.co.vi/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.co.zm/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.co.zw/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.com.af/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.com.ag/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.com.ai/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.com.ar/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.com.bh/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.com.bn/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.com.bo/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.com.bz/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.com.cu/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.com.cy/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.com.eg/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.com.fj/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.com.gh/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.com.gi/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.com.hk/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.com.jm/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.com.kh/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.com.kw/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.com.ly/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.com.mm/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.com.mt/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.com.my/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.com.na/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.com.np/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.com.om/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.com.pe/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.com.pg/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.com.pk/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.com.pr/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.com.qa/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.com.sa/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.com.sl/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.com.tj/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.com.tr/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.com.tw/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.com.ua/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.com.uy/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.com.vn/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.cv/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.de/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.dj/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.dm/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.fm/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.ga/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.gg/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.gl/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.gm/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.gp/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.gy/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.ht/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.ie/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.im/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.is/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.it/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.jo/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.kg/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.ki/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.la/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.li/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.md/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.me/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.mg/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.mn/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.ms/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.mv/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.mw/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.ne/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.nr/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.nu/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.pn/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.ro/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.rw/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.sc/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.sh/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.sm/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.so/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.sr/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.st/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.td/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.tg/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.tl/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.tm/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.to/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.tt/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.vg/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.vu/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.ws/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://club.panasonic.jp/member/terms/?siteId=B1&returnURL=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://community.cypress.com/external-link.jspa?url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://community.cypress.com/external-link.jspa?url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://community.esri.com/external-link.jspa?url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://community.esri.com/external-link.jspa?url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://community.rsa.com/external-link.jspa?url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://contacts.google.com/url?sa=t&source=web&rct=j&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html&ved=2ahUKEwip75vMsq7mAhUv06YKHYSzAN44rAIQFjAYegQIKRAB
https://cse.google.ba/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.by/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.cm/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.co.id/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.co.in/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.co.jp/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.co.ke/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.br/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.hk/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.mx/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.nf/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.ng/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.pr/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.tr/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.tw/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.vn/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.de/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.es/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.it/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.ru/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.se/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.tt/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://demo.html5xcss3.com/demo.php?url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://dereferer.org/?https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://drive.sandbox.google.com.br/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://engawa.kakaku.com/jump/?url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://eventlog.centrum.cz/redir?data=aclick1c68565-349178t12&s=najistong&v=1&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://foro.infojardin.com/proxy.php?link=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html%2F
https://forum.solidworks.com/external-link.jspa?url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://forum.solidworks.com/external-link.jspa?url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://forums-archive.eveonline.com/default.aspx?g=warning&l=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html&domain=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://galter.northwestern.edu/exit?url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://galter.northwestern.edu/exit?url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://gleam.io/zyxKd-INoWr2EMzH?l=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://hobby.idnes.cz/peruanske-palive-papricky-rocoto-dlz-/redir.aspx?url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://id.telstra.com.au/register/crowdsupport?gotoURL=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ac/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ac/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ad/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ae/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ae/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.al/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.al/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.al/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.am/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.am/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.as/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.as/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.as/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.at/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.at/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.az/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.az/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.az/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ba/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ba/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ba/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.be/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.be/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.bf/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.bf/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.bg/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.bi/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.bi/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.bj/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.bj/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.bj/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.bs/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.bt/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.by/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ca/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.cat/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.cat/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.cd/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.cd/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.cd/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.cf/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.cf/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.cg/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.cg/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.cg/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ch/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ch/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ci/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ci/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ci/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.cl/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.cm/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.cm/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.ao/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.ao/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.ao/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.bw/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.ck/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.ck/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.ck/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.cr/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.cr/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.id/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.id/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.il/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.in/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.jp/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.ke/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.ke/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.ke/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.kr/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.kr/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.ls/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.ls/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.ls/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.ma/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.ma/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.mz/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.mz/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.mz/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.nz/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.th/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.tz/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.tz/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.tz/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.ug/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.uk/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.ve/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.ve/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.vi/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.vi/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.vi/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.za/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.za/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.zm/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.zm/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.zm/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.zw/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.zw/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.zw/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.af/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.af/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.af/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.ag/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.ag/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.ag/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.ai/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.ai/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.ai/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.ar/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.ar/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.ar/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.au/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.bd/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.bh/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.bh/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.bh/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.bn/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.bn/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.bn/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.bo/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.bo/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.bo/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.bz/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.bz/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.co/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.co/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.cy/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.cy/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.cy/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.do/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.do/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.ec/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.eg/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.eg/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.et/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.fj/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.fj/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.gh/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.gh/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.gi/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.gt/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.gt/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.hk/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.hk/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.jm/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.kh/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.kh/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.kw/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.kw/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.kw/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.lb/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.ly/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.ly/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.ly/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.mm/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.mm/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.mm/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.mt/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.mt/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.mt/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.mx/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.mx/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.my/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.my/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.na/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.na/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.na/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.nf/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.ng/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.ni/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.ni/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.np/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.np/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.om/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.pa/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.pa/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.pg/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.pg/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.ph/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.ph/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.pk/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.pr/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.pr/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.pr/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.py/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.py/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.qa/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.qa/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.sg/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.sg/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.sl/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.sl/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.sv/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.tj/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.tj/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.tr/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.tw/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.ua/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.ua/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.uy/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.uy/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.vc/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.vc/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.vn/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.vn/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.cv/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.cv/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.cz/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.de/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.de/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.dj/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.dj/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.dk/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.dk/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.dm/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.dm/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.dz/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ee/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ee/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.es/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.es/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.fi/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.fi/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.fm/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.fm/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.fr/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.fr/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ga/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ga/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ga/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ge/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ge/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ge/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.gg/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.gg/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.gg/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.gl/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.gl/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.gm/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.gm/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.gm/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.gp/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.gp/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.gr/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.gr/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.gy/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.hn/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.hn/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.hn/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.hr/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.hr/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ht/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ht/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ht/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.hu/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.hu/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ie/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ie/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.im/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.im/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.im/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.iq/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.is/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.is/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.it/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.it/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.je/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.je/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.jo/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.jo/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.jo/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.kg/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ki/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ki/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ki/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.kz/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.kz/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.la/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.la/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.la/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.li/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.li/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.lk/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.lk/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.lu/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.lu/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.lv/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.lv/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.md/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.md/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.md/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.me/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.me/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.mg/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.mg/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.mg/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.mk/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.mk/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.mk/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.mn/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ms/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ms/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ms/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.mu/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.mu/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.mu/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.mv/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.mv/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.mv/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.mw/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.mw/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.mw/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ne/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ne/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.nl/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.no/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.no/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.no/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.nr/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.nr/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.nr/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.nu/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.nu/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.nu/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.pl/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.pn/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.pn/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.pn/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ps/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ps/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ps/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.pt/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ro/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.rs/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.rs/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ru/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ru/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.rw/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.rw/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.sc/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.sc/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.se/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.se/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.sh/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.sh/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.sh/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.si/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.si/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.sk/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.sk/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.sm/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.sm/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.sn/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.sn/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.sn/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.so/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.so/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.sr/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.sr/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.sr/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.st/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.st/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.st/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.td/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.td/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.tg/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.tg/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.tg/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.tl/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.tl/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.tm/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.tn/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.to/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.tt/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.vg/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.vg/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.vg/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.vu/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.vu/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.vu/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ws/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://inquiry.princetonreview.com/away/?value=cconntwit&category=FS&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://ipv4.google.com/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://it.paltalk.com/client/webapp/client/External.wmt?url=http://https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://jump.2ch.net/?https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://lcu.hlcommission.org/lcu/pages/auth/forgotPassword.aspx?Returnurl=http://https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://legacy.aom.org/verifymember.asp?nextpage=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://lexsrv2.nlm.nih.gov/fdse/search/search.pl?Match=0&Realm=All&Terms=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://m.addthis.com/live/redirect/?url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://m.odnoklassniki.ru/dk?st.cmd=outLinkWarning&st.rfn=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://m.odnoklassniki.ru/dk?st.cmd=outLinkWarning&st.rfn=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://m.ok.ru/dk?st.cmd=outLinkWarning&st.rfn=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://m.ok.ru/dk?st.cmd=outLinkWarning&st.rfn=http://https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ad/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ad/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ae/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.as/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.as/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.as/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.at/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.at/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ba/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ba/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.be/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.be/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.bf/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.bf/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.bg/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.bg/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.bi/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.bi/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.bj/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.bj/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.bj/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.bs/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.bs/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.bt/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.by/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.by/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ca/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.cat/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.cat/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.cd/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.cf/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.cf/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.cg/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.cg/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.cg/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ch/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ch/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ci/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ci/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.cl/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.cl/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.cm/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.cm/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.ao/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.ao/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.ao/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.bw/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.bw/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.bw/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.ck/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.ck/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.ck/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.cr/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.cr/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.id/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.il/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.in/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.in/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.ke/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.kr/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.kr/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.ls/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.ls/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.ls/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.mz/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.mz/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.mz/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.nz/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.th/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.th/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.tz/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.tz/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.ug/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.ug/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.uk/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.ve/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.ve/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.vi/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.vi/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.vi/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.za/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.za/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.zm/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.zm/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.zm/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.zw/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.zw/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.ag/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.ag/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.ag/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.ai/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.ai/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.ai/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.ar/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.ar/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.ar/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.au/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.bd/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.bd/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.bd/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.bh/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.bh/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.bh/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.bn/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.bn/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.bn/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.bo/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.br/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.bz/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.bz/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.bz/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.co/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.co/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.cu/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.cu/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.cu/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.do/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.do/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.ec/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.eg/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.eg/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.fj/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.fj/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.fj/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.gh/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.gh/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.gh/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.gi/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.gi/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.gi/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.gt/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.gt/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.hk/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.hk/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.jm/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.jm/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.kh/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.kh/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.kh/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.kw/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.kw/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.kw/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.lb/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.ly/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.ly/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.ly/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.mm/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.mm/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.mt/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.mt/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.mt/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.mx/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.mx/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.my/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.my/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.na/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.na/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.na/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.ng/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.ni/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.ni/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.om/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.pa/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.pa/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.pe/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.pe/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.pg/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.ph/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.pr/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.pr/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.pr/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.py/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.py/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.qa/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.qa/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.sa/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.sa/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.sg/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.sg/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.sl/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.sl/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.sv/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.tr/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.tr/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.tw/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.tw/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.ua/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.ua/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.ua/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.uy/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.uy/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.uy/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.vc/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.vc/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.cv/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.cv/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.cz/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.cz/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.de/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.de/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.dj/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.dj/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.dk/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.dk/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.dm/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.dm/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.dm/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.dz/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ee/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ee/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.es/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.es/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.fi/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.fi/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.fm/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.fm/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.fr/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.fr/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ga/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ge/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ge/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.gg/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.gg/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.gl/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.gl/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.gm/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.gm/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.gm/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.gp/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.gp/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.gr/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.gr/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.gy/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.gy/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.hn/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.hr/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.hr/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ht/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.hu/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.hu/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.hu/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ie/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.im/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.im/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.im/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.iq/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.is/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.is/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.is/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.it/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.it/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.je/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.je/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.je/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.jo/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.jo/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.jo/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.kg/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ki/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ki/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ki/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.kz/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.kz/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.la/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.la/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.la/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.li/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.lk/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.lk/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.lt/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.lu/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.lu/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.lv/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.lv/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.mg/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.mg/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.mg/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.mk/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.mk/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.mk/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.mn/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ms/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ms/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://www.fhwa.dot.gov/reauthorization/reauexit.cfm?link=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.fhwa.dot.gov/reauthorization/reauexit.cfm?link=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.ladan.com.ua/link/go.php?url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html%2F
https://www.webclap.com/php/jump.php?url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html%2F
https://schwarzes-bw.de/wbb231/redir.php?url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.webclap.com/php/jump.php?url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.cu/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://www2.ogs.state.ny.us/help/urlstatusgo.html?url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://go.infomine.com/?re=121&tg=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://link.harikonotora.net/?http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://text.usg.edu/tt/https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://uniportal.huaweidevice.com/uniportal/jsp/clearCrossDomainCookie.jsp?redirectUrl=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://www.fito.nnov.ru/go.php?url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://www2.ogs.state.ny.us/help/urlstatusgo.html?url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://affiliates.bookdepository.com/scripts/click.php?a_aid=Alexa1202&a_bid=9abb5269&desturl=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://s5.histats.com/stats/r.php?869637&100&47794&urlr=&https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://15.pro.tok2.com/%7Ekoro/cutlinks/rank.php?url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://affiliates.bookdepository.com/scripts/click.php?a_aid=Alexa1202&a_bid=9abb5269&desturl=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://affiliates.bookdepository.com/scripts/click.php?a_aid=Alexa1202&abid=9abb5269&desturl=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://channel.pixnet.net/newdirect.php?blog=cr19h3id&serial=0&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.cu/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://janeyleegrace.worldsecuresystems.com/redirect.aspx?destination=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.ba/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.be/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.bg/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.bs/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.ca/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.cg/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.ch/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.cl/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.co.bw/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.co.cr/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.co.id/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.co.il/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.co.il/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.co.in/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.co.jp/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.co.ke/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.co.kr/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.co.ma/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.co.nz/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.co.th/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.co.ug/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.co.uk/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.co.uk/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.co.ve/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.co.za/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.af/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.au/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.bd/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.br/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.do/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.py/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.qa/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.sg/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.sv/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.tr/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.tw/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.ua/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.uy/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.vn/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.pt/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.rs/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.si/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.sk/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.tn/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.vg/url?q=https%3A%2F%2Fhttps://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.at/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.be/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.bg/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ca/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ca/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ch/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.cl/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.id/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.il/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.in/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.jp/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.nz/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.th/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.uk/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.za/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.au/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.br/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.br/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.cu/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.hk/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.mx/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.my/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.sg/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.tr/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.tw/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.ua/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.vn/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.vn/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.cz/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.de/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.dk/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.es/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.fi/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.fr/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.gr/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.hu/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ie/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.nl/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.pl/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.pt/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ro/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ru/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.sk/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.be/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ca/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ch/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.cl/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.cl/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.id/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.id/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html%2F
https://maps.google.co.il/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.in/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.jp/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.nz/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.th/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.uk/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.za/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.au/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.br/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.hk/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.mx/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.my/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.sg/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.tr/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.tw/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.tw/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html%2F
https://maps.google.com.ua/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.cz/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.de/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.dk/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.es/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.fi/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.fr/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.gr/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.hu/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ie/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.nl/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.pl/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.pt/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ro/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ru/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.sk/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.at/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.co.id/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.co.il/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.co.jp/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.co.kr/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.co.uk/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.co.za/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.ar/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.br/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.mx/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.my/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.pe/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.ph/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.tw/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.ua/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.vn/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.de/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.hu/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.ie/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.it/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.pt/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.ro/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.bing.com/news/apiclick.aspx?ref=FexRss&aid=&tid=9BB77FDA801248A5AD23FDBDD5922800&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.ae/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.az/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.bs/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.co.bw/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.co.ke/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.co.ve/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.bd/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.bh/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.cy/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.ec/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.ng/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.np/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.pk/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.qa/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.lu/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://club.panasonic.jp/member/terms/?siteId=B1&returnURL=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://keywordspace.com/site-info/betop24.com
https://www.bombstat.com/domain/betop24.com
https://maps.google.com.br/url?sa=t&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.jo/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.ly/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.mu/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.ml/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.de/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.uk/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.fr/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.fr/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.nl/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ch/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.be/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.tr/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.mx/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.dk/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.pt/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.hr/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.lv/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.pr/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.lu/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.lu/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.co.ma/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.mn/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.com.kh/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.ug/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.ly/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://tyonabi.sakura.ne.jp/link/cgi-bin/out.cgi?id=dorian362&cg=1&siteurl=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://feeds.copyblogger.com/%7E/t/0/0/copyblogger/%7Ehttp%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
http://www.scoop.co.nz/link-out?p=blog93&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.be/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.bf/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.co.ck/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.bz/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.om/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.pk/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.sl/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.com.vn/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.dm/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.ge/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.iq/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.pl/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.pn/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.so/url?sa=i&url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.cr/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.pe/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.li/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://lcu.hlcommission.org/lcu/pages/auth/forgotPassword.aspx?Returnurl=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.bo/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.lb/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.np/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.sa/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.de/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ee/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ie/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.nl/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.tn/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://t.me/iv?url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://transtats.bts.gov/exit.asp?url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.by/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.es/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.pl/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://www.google.sk/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://a.pr-cy.ru/https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://client.paltalk.com/client/webapp/client/External.wmt?url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.com.et/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.com.nf/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.com.vc/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.je/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.lk/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://clients1.google.mk/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.co.kr/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://cse.google.fr/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://doodle.com/r?url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://galter.northwestern.edu/exit?url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.bg/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.bs/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.cl/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.bw/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.in/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.ma/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.th/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.co.uz/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.bd/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.br/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.bz/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.cu/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.ec/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.fj/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.gh/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.gi/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.gi/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.ni/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.com.tw/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.cz/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.gy/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.is/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.kz/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.lt/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.me/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.ro/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.rs/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.sm/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://images.google.to/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://ipv4.google.com/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ad/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ae/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.bi/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.cat/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.cd/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.cd/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ci/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.cm/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.id/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.jp/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.tz/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.co.zw/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.ec/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.et/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.mm/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.ng/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.np/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.np/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.pg/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.ph/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.sv/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com.sv/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.com/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ga/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.gg/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.hn/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ht/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ht/url?q=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ie/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.kz/url?q=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://maps.google.ms/url?sa=t&url=http%3A%2F%2F1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://sitereport.netcraft.com/?url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html%2F
https://ranking.crawler.com/SiteInfo.aspx?url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
https://valueanalyze.com/show.php?url=https://1arctic.blogspot.com/2023/03/online-club-invite-reward-guarantee-now.html
terry36sao@gmail.com 1/13/23
https://cutt.ly/u2jE5Vl
카지노사이트 https://ce-top10.com/
바카라사이트 https://ce-top10.com/
온라인카지노 https://ce-top10.com/
온라인바카라 https://ce-top10.com/
온라인슬롯사이트 https://ce-top10.com/
카지노사이트게임 https://ce-top10.com/
카지노사이트검증 https://ce-top10.com/
카지노사이트추천 https://ce-top10.com/
안전카지노사이트 https://ce-top10.com/
안전카지노사이트도메인 https://ce-top10.com/
안전한 카지노사이트 추천 https://ce-top10.com/
바카라사이트게임 https://ce-top10.com/
바카라사이트검증 https://ce-top10.com/
바카라사이트추천 https://ce-top10.com/
안전바카라사이트 https://ce-top10.com/
안전바카라사이트도메인 https://ce-top10.com/
안전한 바카라사이트 추천 https://ce-top10.com/
http://toolbarqueries.google.com.uy/url?sa=t&url=https://ce-top10.com/
http://toolbarqueries.google.com.tw/url?sa=t&url=https://ce-top10.com/
http://toolbarqueries.google.com.tr/url?sa=t&url=https://ce-top10.com/
http://toolbarqueries.google.com.sa/url?sa=t&url=https://ce-top10.com/
http://toolbarqueries.google.com.py/url?sa=t&url=https://ce-top10.com/
http://toolbarqueries.google.com.pr/url?sa=t&url=https://ce-top10.com/
http://toolbarqueries.google.com.pk/url?sa=t&url=https://ce-top10.com/
http://toolbarqueries.google.com.pe/url?sa=t&url=https://ce-top10.com/
http://toolbarqueries.google.com.my/url?sa=t&url=https://ce-top10.com/
http://toolbarqueries.google.com.hk/url?sa=t&url=https://ce-top10.com/
http://toolbarqueries.google.com.gt/url?sa=t&url=https://ce-top10.com/
http://toolbarqueries.google.com.gh/url?sa=t&url=https://ce-top10.com/
https://clients1.google.com.ar/url?sa=t&url=https://ce-top10.com/
https://clients1.google.com.ag/url?sa=t&url=https://ce-top10.com/
https://clients1.google.co.zm/url?sa=t&url=https://ce-top10.com/
https://clients1.google.co.za/url?sa=t&url=https://ce-top10.com/
https://clients1.google.co.ve/url?sa=t&url=https://ce-top10.com/
https://clients1.google.co.uz/url?sa=t&url=https://ce-top10.com/
https://clients1.google.co.ug/url?sa=t&url=https://ce-top10.com/
https://clients1.google.co.th/url?sa=t&url=https://ce-top10.com/
https://clients1.google.co.nz/url?sa=t&url=https://ce-top10.com/
https://clients1.google.co.kr/url?sa=t&url=https://ce-top10.com/
https://clients1.google.co.ke/url?sa=t&url=https://ce-top10.com/
https://clients1.google.co.il/url?sa=t&url=https://ce-top10.com/
https://clients1.google.co.id/url?sa=t&url=https://ce-top10.com/
https://clients1.google.co.cr/url?sa=t&url=https://ce-top10.com/
https://clients1.google.co.ck/url?sa=t&url=https://ce-top10.com/
https://cse.google.co.ck/url?sa=t&url=https://ce-top10.com/
https://cse.google.co.bw/url?sa=t&url=https://ce-top10.com/
https://cse.google.cm/url?sa=t&url=https://ce-top10.com/
https://cse.google.cl/url?sa=t&url=https://ce-top10.com/
https://cse.google.ci/url?sa=t&url=https://ce-top10.com/
https://cse.google.ch/url?sa=t&url=https://ce-top10.com/
https://cse.google.ch/url?sa=i&url=https://ce-top10.com/
https://cse.google.cg/url?sa=t&url=https://ce-top10.com/
https://cse.google.cd/url?sa=t&url=https://ce-top10.com/
https://cse.google.by/url?sa=t&url=https://ce-top10.com/
https://cse.google.bs/url?sa=t&url=https://ce-top10.com/
https://cse.google.bi/url?sa=t&url=https://ce-top10.com/
https://cse.google.bg/url?sa=t&url=https://ce-top10.com/
https://cse.google.be/url?sa=t&url=https://ce-top10.com/
https://cse.google.be/url?sa=i&url=https://ce-top10.com/
https://cse.google.ba/url?sa=t&url=https://ce-top10.com/
https://cse.google.az/url?sa=t&url=https://ce-top10.com/
https://cse.google.at/url?sa=t&url=https://ce-top10.com/
https://images.google.ca/url?sa=t&url=http://casino1top.xyz/index.php/2023/01/12/instructions-to-win-large-on-web-based-openings/
https://images.google.by/url?sa=t&url=http://casino1top.xyz/index.php/2023/01/12/instructions-to-win-large-on-web-based-openings/
https://images.google.bs/url?sa=t&url=http://casino1top.xyz/index.php/2023/01/12/instructions-to-win-large-on-web-based-openings/
https://images.google.bi/url?sa=t&url=http://casino1top.xyz/index.php/2023/01/12/instructions-to-win-large-on-web-based-openings/
https://images.google.bg/url?sa=t&url=http://casino1top.xyz/index.php/2023/01/12/instructions-to-win-large-on-web-based-openings/
https://images.google.bf/url?sa=t&url=http://casino1top.xyz/index.php/2023/01/12/instructions-to-win-large-on-web-based-openings/
https://images.google.be/url?sa=t&url=http://casino1top.xyz/index.php/2023/01/12/instructions-to-win-large-on-web-based-openings/
https://images.google.ba/url?sa=t&url=http://casino1top.xyz/index.php/2023/01/12/instructions-to-win-large-on-web-based-openings/
https://images.google.ba/url?q=http://casino1top.xyz/index.php/2023/01/12/instructions-to-win-large-on-web-based-openings/
https://images.google.az/url?sa=t&url=http://casino1top.xyz/index.php/2023/01/12/instructions-to-win-large-on-web-based-openings/
https://images.google.at/url?sa=t&url=http://casino1top.xyz/index.php/2023/01/12/instructions-to-win-large-on-web-based-openings/
https://images.google.as/url?sa=t&url=http://casino1top.xyz/index.php/2023/01/12/instructions-to-win-large-on-web-based-openings/
https://images.google.am/url?sa=t&url=http://casino1top.xyz/index.php/2023/01/12/instructions-to-win-large-on-web-based-openings/
https://images.google.al/url?sa=t&url=http://casino1top.xyz/index.php/2023/01/12/instructions-to-win-large-on-web-based-openings/
https://images.google.ae/url?sa=t&url=http://casino1top.xyz/index.php/2023/01/12/instructions-to-win-large-on-web-based-openings/
https://images.google.ae/url?q=http://casino1top.xyz/index.php/2023/01/12/instructions-to-win-large-on-web-based-openings/
https://images.google.ad/url?sa=t&url=http://casino1top.xyz/index.php/2023/01/12/instructions-to-win-large-on-web-based-openings/
https://maps.google.co.ke/url?sa=t&url=http://casino1top.xyz/index.php/2023/01/12/instructions-to-win-large-on-web-based-openings/
https://maps.google.co.jp/url?sa=t&url=http://casino1top.xyz/index.php/2023/01/12/instructions-to-win-large-on-web-based-openings/
https://maps.google.co.in/url?sa=t&url=http://casino1top.xyz/index.php/2023/01/12/instructions-to-win-large-on-web-based-openings/
https://maps.google.co.il/url?sa=t&url=http://casino1top.xyz/index.php/2023/01/12/instructions-to-win-large-on-web-based-openings/
https://maps.google.co.id/url?sa=t&url=http://casino1top.xyz/index.php/2023/01/12/instructions-to-win-large-on-web-based-openings/
https://maps.google.co.cr/url?sa=t&url=http://casino1top.xyz/index.php/2023/01/12/instructions-to-win-large-on-web-based-openings/
https://maps.google.co.bw/url?sa=t&url=http://casino1top.xyz/index.php/2023/01/12/instructions-to-win-large-on-web-based-openings/
https://maps.google.cm/url?sa=t&url=http://casino1top.xyz/index.php/2023/01/12/instructions-to-win-large-on-web-based-openings/
https://maps.google.cl/url?sa=t&url=http://casino1top.xyz/index.php/2023/01/12/instructions-to-win-large-on-web-based-openings/
https://maps.google.ci/url?sa=t&url=http://casino1top.xyz/index.php/2023/01/12/instructions-to-win-large-on-web-based-openings/
https://maps.google.ch/url?sa=t&url=http://casino1top.xyz/index.php/2023/01/12/instructions-to-win-large-on-web-based-openings/
https://maps.google.cd/url?sa=t&url=http://casino1top.xyz/index.php/2023/01/12/instructions-to-win-large-on-web-based-openings/
https://maps.google.cat/url?sa=t&url=http://casino1top.xyz/index.php/2023/01/12/instructions-to-win-large-on-web-based-openings/
https://maps.google.ca/url?sa=t&url=http://casino1top.xyz/index.php/2023/01/12/instructions-to-win-large-on-web-based-openings/
https://www.google.co.tz/url?sa=t&url=http://casino1top.xyz/index.php/2023/01/12/instructions-to-win-large-on-web-based-openings/
https://www.google.co.th/url?sa=t&url=http://casino1top.xyz/index.php/2023/01/12/instructions-to-win-large-on-web-based-openings/
https://www.google.co.nz/url?sa=t&url=http://casino1top.xyz/index.php/2023/01/12/instructions-to-win-large-on-web-based-openings/
https://www.google.co.ma/url?sa=t&url=http://casino1top.xyz/index.php/2023/01/12/instructions-to-win-large-on-web-based-openings/
https://www.google.co.ls/url?sa=t&url=http://casino1top.xyz/index.php/2023/01/12/instructions-to-win-large-on-web-based-openings/
https://www.google.co.kr/url?sa=t&url=http://casino1top.xyz/index.php/2023/01/12/instructions-to-win-large-on-web-based-openings/
https://www.google.co.ke/url?sa=t&url=http://casino1top.xyz/index.php/2023/01/12/instructions-to-win-large-on-web-based-openings/
https://www.google.co.jp/url?sa=t&url=http://casino1top.xyz/index.php/2023/01/12/instructions-to-win-large-on-web-based-openings/
https://www.google.co.in/url?sa=t&url=http://casino1top.xyz/index.php/2023/01/12/instructions-to-win-large-on-web-based-openings/
https://www.google.co.il/url?sa=t&url=http://casino1top.xyz/index.php/2023/01/12/instructions-to-win-large-on-web-based-openings/
https://www.google.co.id/url?sa=t&url=http://casino1top.xyz/index.php/2023/01/12/instructions-to-win-large-on-web-based-openings/
https://www.google.co.cr/url?sa=t&url=http://casino1top.xyz/index.php/2023/01/12/instructions-to-win-large-on-web-based-openings/
https://www.google.co.bw/url?sa=t&url=http://casino1top.xyz/index.php/2023/01/12/instructions-to-win-large-on-web-based-openings/
https://www.google.cm/url?sa=t&url=http://casino1top.xyz/index.php/2023/01/12/instructions-to-win-large-on-web-based-openings/
https://www.google.cl/url?sa=t&url=http://casino1top.xyz/index.php/2023/01/12/instructions-to-win-large-on-web-based-openings/
https://www.google.ci/url?sa=t&url=http://casino1top.xyz/index.php/2023/01/12/instructions-to-win-large-on-web-based-openings/
https://www.google.ch/url?sa=t&url=http://casino1top.xyz/index.php/2023/01/12/instructions-to-win-large-on-web-based-openings/
https://www.google.cd/url?sa=t&url=http://casino1top.xyz/index.php/2023/01/12/instructions-to-win-large-on-web-based-openings/
https://www.google.cat/url?sa=t&url=http://casino1top.xyz/index.php/2023/01/12/instructions-to-win-large-on-web-based-openings/
https://www.google.ca/url?sa=t&url=http://casino1top.xyz/index.php/2023/01/12/instructions-to-win-large-on-web-based-openings/
바카라사이트
카지노사이트
카지노게임사이트
온라인카지노
퀸즈슬롯
맥스카지노
비바카지노
카지노주소
바카라추천
온라인바카라게임
안전한 바카라사이트
바카라
카지노
퀸즈슬롯 카지노
바카라게임사이트
온라인바카라
밀리언클럽카지노
안전카지노사이트
바카라사이트추천
우리카지노계열
슬롯머신777
로얄카지노사이트
크레이지슬롯
온라인블랙잭
인터넷룰렛
카지노검증사이트
안전바카라사이트
모바일바카라
바카라 필승법
메리트카지노
바카라 노하우
바카라사이트
온라인카지노
카지노사이트
카지노게임사이트
퀸즈슬롯
맥스카지노
비바카지노
카지노주소
바카라추천
온라인바카라게임
안전한 바카라사이트
바카라
카지노
퀸즈슬롯 카지노
바카라게임사이트
온라인바카라
밀리언클럽카지노
안전카지노사이트
바카라사이트추천
우리카지노계열
슬롯머신777
로얄카지노사이트
크레이지슬롯
온라인블랙잭
인터넷룰렛
카지노검증사이트
안전바카라사이트
모바일바카라
바카라 필승법
메리트카지노
바카라 노하우
https://youube.me/
https://instagrme.com/
https://youubbe.me/
https://Instagrm.me/
https://Instagrme.net/
https://internetgame.me/
https://instagrme.live/
https://naverom.me
https://facebokom.me