Styrene: The Building Block to Foam and Plastic!
Styrene, also known as ethenylbenzene, vinylbenzene, and phenylethene. Is an organic compound with the chemical formula C6H5CH=CH2. This derivative of benzene is a colorless oily liquid that evaporates easily and has a sweet smell, although high concentrations have a less pleasant odor. Styrene is the precursor to polystyrene and several copolymers. Polystyrene is the molecule behind plastics and foam. It is clear, hard, and rather brittle.
Styrene, a component of polystyrene is a colorless, toxic liquid with a strong aromatic odor. It is used to make rubbers, polymers and copolymers.
Styrene is found in alcoholic beverages, it is also present in cranberry, bilberry, currants, grapes, vinegar, parsley, milk and dairy products, whisky, cocoa, coffee, tea, roasted filberts and peanuts. Styrene is a flavoring ingredient.
What does Styrene look like in Chemistry?

Let’s Get Building!
Using your Student Molecular Model Set from Duluth Labs let’s create Styrene! You’ll need:
-
8 Carbon Atoms
-
8 Hydrogen atoms
-
8 Small connectors (compact small bonds for hydrogen)
-
4 Medium Connectors
-
8 Long connectors
-
Molecular Tool (for Disassembly)
Put aside all the atoms and connectors needed.
Let’s Start Building With Our Phenol Portion.
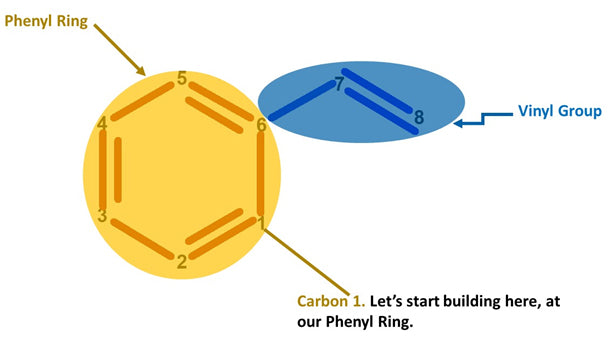
Note: We build this portion in a clockwise direction, starting with Carbon 1.
Let’s start!
Steps:
-
1

1. Get a carbon atom (Carbon 1) then attach another carbon (Carbon 2) to it using 2 long connectors. Add a hydrogen atom to Carbon 1 using a small connector
-
2

2. Attach another carbon atom (Carbon 3) to Carbon 2using a medium connector. Place a hydrogen atom on Carbon 2 using a small connector
-
3

3. Get another carbon atom (Carbon 4)then attach this to Carbon 3 using 2 long connectors. Using a small connector attach a hydrogen atom to carbon 3
-
4

4. Then, attach another carbon atom (Carbon 5) to Carbon 4 using a medium connector, then, attach a hydrogen atom to carbon 5 using a small connector.
-
5

5. Attach one carbon atom (Carbon 6)toCarbon 5 using 2 long connectors and then attach a hydrogen atom to Carbon 6 using a small connector.
-
6

6. Join Carbon 6and Carbon 1together using a medium connector.
-

Yay! We've just built our Phenyl Portion!
Note: Let’s now build the Vinyl portion of our Styrene molecule. We build this portion starting with Carbon 7.

Let’s continue building!
Steps:
-
7

7. Get one carbon atom (Carbon 7) then attach this to Carbon 6 (of the Phenyl portion) using a medium connector. Then, place one hydrogen atom on carbon 7 using a small connector.
-
8

8. Get another carbon atom (Carbon 8) and use 2 long connectors to attach Carbon 8 to Carbon 7. Then, add two hydrogen atoms to carbon 8 using two small connectors.
-

Yay! We've just built our Vinyl Portion!















