This Week’s Hints:

-
It is extracted from a plant
-
It can be used as a flavoring agent in food, beverages and pharmaceuticals
-
Very sweet in smell
V____ ____ I ____ L ____ ____ 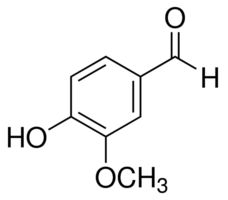
That’s Right! This Week’s Molecule will be Vanillin!
Here’s a bit of information about Vanillin:
Vanillin is a phenolic compound which has a phenolic ring, an aldehyde group, hydroxyl and ether group. It is derived from vanilla bean, as which synthetic vanillin is now used very often as a flavouring agent in foods. This is because natural vanilla tends to be more expensive than vanillin.
So What Does Vanillin Looks Like in Chemistry?


Let’s Get Building!
Using your Student Molecular Set from Duluth Labs, let’s create Vanillin. You’ll need:
- 8 Carbon Atoms
- 8 Hydrogen Atoms
- 3 Oxygen Atoms
- 7 Medium connectors (single covalent bonds)
- 8 Long connectors (double bonds)
- 8 Short connectors (hydrogen bonds)
- Molecular Tool (for Disassembly)
Put aside all the atoms and connectors needed.
We will be building this molecule in a clockwise rotation with the carbon atom in the 6:30 position. Using the chemical structure above as a representative, we will start by building a Benzene Ring.
- First using a carbon atom and 2 long connectors, attach another carbon atom to form a double bonded carbon.

- Then using a medium connector, attach a carbon atom to the double bonded carbon.

- Using 2 long connectors, attach another carbon atom to form another double bonded carbon.

- Using a medium connector, connect a carbon atom to the double bonded carbon.

- Using 2 long connectors, attach another carbon atom to form another double bonded carbon.

- Finally, using a medium connector, connect both carbon atoms to each other.

At this stage, we now have our Benzene Ring! Now lets add all the functional groups to create Vanillin.
Starting with the carbon at the 6:30 position, we are going to create the OCH3 group.
- Using a medium connector attach an oxygen atom, then using another medium connector attach a carbon atom. Thereafter, using 3 short connectors attach 3 hydrogen atoms to the carbon.

- Going in a clockwise position, at the next carbon atom ( for this example: carbon #2) in the ring, use a medium connector to attach an oxygen atom.

- Using a short connector, attach a hydrogen atom to the oxygen atom forming an OH group.

- Going in a clockwise position, at carbon #5, use a medium connector to attach a carbon atom.

- Using 2 long connectors, attach an oxygen atom

- Using a short connector, attach a hydrogen atom to the carbon free electron.

- Now that we have added all the functional groups, it is time to use the remaining small connectors and attach hydrogen atoms to the free electrons on the carbon atoms throughout the molecule.

We Want to Know How you made your Vanilla!
How did your Vanillin turn out?
Comment below how your Vanillin molecule turned out.
Sign up to our mailing list to tune in next week for another molecule of the week!
Download today's blog post here.
See you then xoxo :)
Duluth Labs
Afton Direct LLC

















