FLUOROURACIL: How does it fool your cancer cells?
Fluorouracil is used as an anticancer agent. It belongs to a group of chemotherapeutic drugs known as antimetabolites. As its structure suggests, it looks similar to a nucleic acid base Thymine and Uracil, with slight differences in structure. These slight differences in their structure are used as a means of inhibiting the cancer cells’ vital processes such as DNA repair and replication. As a result, cancer cells are not able to grow and multiply, hence, the chances of cancer cells being able to spread all over the body goes down.
How does Fluorouracil look like in Chemistry?
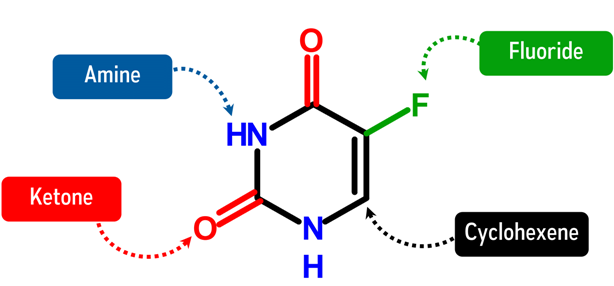
Let’s Get Building!
Using your Student Molecular Set from Duluth Labs let’s create Fluorouracil! You’ll need:
- 4 Carbon atoms
- 2 Oxygen atoms
- 1 Fluorine atom
- 3 Hydrogen atoms
- 2 Nitrogen atoms
- 3 Small Connectors (compact small bonds for hydrogen)
- 6 Medium Connectors
- 6 Long Connectors
- Molecular Tool (for Disassembly)
Put aside all the atoms and connectors needed.
Let’s Start off With Our Carbon 6!
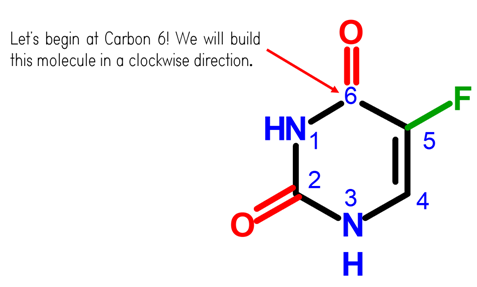
Steps:
-
1

1. Get one Carbon atom (Carbon 6) then, attach an Oxygen atom atom above it using 2 long connectors
-
2

2. Then, get another Carbon atom (Carbon 5) then attach this to the right of Carbon 6 using 1 medium connector.
-
3

3. Attach a fluorine atom to Carbon 5 using a medium connector.
-
4

4. Grab another carbon atom (Carbon 4) then attach this below Carbon 5 using 2 long connectors. Then, place a hydrogen atom on Carbon 4 using a small connector
-
5

5.Attach a nitrogen atom (Nitrogen 3) to the left of Carbon 4 using 1 medium connector. Add a hydrogen atom to Nitrogen 3 using a small connector
-
6

6. Attach a carbon atom (Carbon 2) to the left of Nitrogen 3 using 1 medium connector.
-
7

7. Get one oxygen atom and attached this to Carbon 2 using a medium connector.
-
8

8. Then, attach another nitrogen atom (Nitrogen 1) above Carbon 2 using 1 medium connector. Place 1 hydrogen atom on Nitrogen 1 using a small connector.
-
9

9. Join Nitrogen 1 and Carbon 6 together using 1 medium connector.















https://www.youubbe.me/ – 카지노사이트,바카라사이트,슬롯사이트,온라인카지노,카지노주소
https://youubbe.me/ – 카지노검증사이트
https://youubbe.me/ – 안전한카지노사이트
https://youubbe.me/ – 슬롯카지노
https://youubbe.me/ – 바카라게임
https://youubbe.me/ – 카지노추천
https://youubbe.me/ – 비바카지노
https://youubbe.me/ – 퀸즈슬롯
https://youubbe.me/ – 카지노
https://youubbe.me/ – 바카라
https://youubbe.me/ – 안전한 바카라사이트
https://youubbe.me/ – 온라인슬롯
https://youubbe.me/casinosite/ – 카지노사이트
https://youubbe.me/baccarat/ – 바카라
https://youubbe.me/baccaratsite/ – 바카라사이트
https://youubbe.me/pharaoh-casino/ – 파라오카지노
https://youubbe.me/제왕카지노/ – 제왕카지노
https://youubbe.me/mgm카지노/ – mgm카지노
https://youubbe.me/theking-casino/ – 더킹카지노
https://youubbe.me/coin-casino/ – 코인카지노
https://youubbe.me/solaire-casino/ – 솔레어카지노
https://youubbe.me/casino-game/ – 카지노게임
https://youubbe.me/micro-gaming/ – 마이크로게이밍
https://youubbe.me/asia-gaming/ – 아시아게이밍
https://youubbe.me/taisan-gaming/ – 타이산게이밍
https://youubbe.me/oriental-game/ – 오리엔탈게임
https://youubbe.me/evolution-game/ – 에볼루션게임
https://youubbe.me/dragon-tiger/ – 드래곤타이거
https://youubbe.me/dream-gaming/ – 드림게이밍
https://youubbe.me/vivo-gaming/ – 비보게이밍
youube.me
gamja888.com
instagrme.com
Instagrm.me
Instagrme.net
internetgame.me
instagrme.live
naverom.me
facebokom.me
https://www.youube.me/ – 카지노사이트,바카라사이트,바카라게임사이트,온라인바카라,인터넷카지노
https://youube.me/ – 퀸즈슬롯
https://youube.me/ – 카지노주소
https://youube.me/ – 비바카지노
https://youube.me/ – 카지노추천
https://youube.me/ – 카지노게임
https://youube.me/ – 온라인카지노사이트
https://youube.me/ – 카지노
https://youube.me/ – 바카라
https://youube.me/ – 온라인카지노
https://youube.me/ – 카지노게임사이트
https://youube.me/sandscasinoaddress/ – 카지노검증사이트
https://youube.me/royalcasinoseries/ – 로얄카지노계열
https://youube.me/slotmachinesite/ – 슬롯머신사이트
https://youube.me/maxcasino/ – 맥스카지노
https://youube.me/baccaratgamesite/ – 바카라게임사이트
https://youube.me/casimbakorea-casino/ – 카심바코리아 카지노
https://youube.me/mobilecasino/ – 모바일카지노
https://youube.me/real-timebaccarat/ – 실시간바카라
https://youube.me/livecasino/ – 라이브카지노
https://youube.me/onlineslots/ – 온라인슬롯
https://youube.me/sandscasinoaddress/ – 바카라 이기는방법
https://youube.me/safecasinosite/ – 안전카지노사이트
https://youube.me/ourcasinosite/ – 우리카지노사이트
https://youube.me/sandscasinoaddress/ – 샌즈카지노주소
https://youube.me/baccarat-rulesofthegame/ – 바카라 게임규칙
https://youube.me/baccarat-howtoplay/ – 바카라 게임방법
gamja888.com
instagrme.com
youubbe.me
Instagrm.me
Instagrme.net
internetgame.me
instagrme.live
naverom.me
facebokom.me
temp mail https://tempgmail.email/
temp-mail-email-gmail http://images.google.md/url?sa=t&url=https://tempgmail.email/
temp mail http://www.google.md/url?sa=t&url=https://tempgmail.email/
temp gmail http://images.google.am/url?sa=t&url=https://tempgmail.email/
temp email http://www.google.am/url?sa=t&url=https://tempgmail.email/
tempmail http://maps.google.mn/url?sa=t&url=https://tempgmail.email/
tempemail http://images.google.mn/url?sa=t&url=https://tempgmail.email/
free temp mail http://www.google.mn/url?sa=t&url=https://tempgmail.email/
temp-mail http://www.google.bi/url?sa=t&url=https://tempgmail.email/
temp-gmail http://maps.google.bi/url?sa=t&url=https://tempgmail.email/
temp-mail-email-gmail http://images.google.bi/url?sa=t&url=https://tempgmail.email/
temp mail http://images.google.me/url?sa=t&url=https://tempgmail.email/
temp gmail http://www.google.me/url?sa=t&url=https://tempgmail.email/
temp email http://www.google.fm/url?sa=t&url=https://tempgmail.email/
tempmail http://maps.google.fm/url?sa=t&url=https://tempgmail.email/
tempemail http://images.google.fm/url?sa=t&url=https://tempgmail.email/
free temp mail http://maps.google.je/url?sa=t&url=https://tempgmail.email/
temp-mail http://images.google.je/url?sa=t&url=https://tempgmail.email/
temp-gmail http://www.google.je/url?sa=t&url=https://tempgmail.email/
temp-mail-email-gmail http://maps.google.co.tz/url?sa=t&url=https://tempgmail.email/
temp mail http://images.google.co.tz/url?sa=t&url=https://tempgmail.email/
temp gmail http://www.google.co.tz/url?sa=t&url=https://tempgmail.email/
temp email http://maps.google.la/url?sa=t&url=https://tempgmail.email/
tempmail http://www.google.la/url?sa=t&url=https://tempgmail.email/
tempemail http://images.google.la/url?sa=t&url=https://tempgmail.email/
free temp mail http://www.google.ms/url?sa=t&url=https://tempgmail.email/
temp-mail http://images.google.ms/url?sa=t&url=https://tempgmail.email/
temp-gmail http://maps.google.ms/url?sa=t&url=https://tempgmail.email/
temp-mail-email-gmail http://images.google.kg/url?sa=t&url=https://tempgmail.email/
temp mail http://maps.google.kg/url?sa=t&url=https://tempgmail.email/
temp-mail-email-gmail http://www.google.kg/url?sa=t&url=https://tempgmail.email/
temp mail http://images.google.sh/url?sa=t&url=https://tempgmail.email/
temp gmail http://www.google.sh/url?sa=t&url=https://tempgmail.email/
temp email http://maps.google.sh/url?sa=t&url=https://tempgmail.email/
tempmail http://ziepod.com/addpodcast.php?xml=https://tempgmail.email/
tempemail http://maps.google.cd/url?sa=t&url=https://tempgmail.email/
free temp mail http://www.google.cd/url?sa=t&url=https://tempgmail.email/
temp-mail http://images.google.cd/url?sa=t&url=https://tempgmail.email/
temp-gmail http://maps.google.ht/url?sa=t&url=https://tempgmail.email/
temp-mail-email-gmail http://www.google.ht/url?sa=t&url=https://tempgmail.email/
temp mail http://images.google.ht/url?sa=t&url=https://tempgmail.email/
temp gmail http://www.google.gp/url?sa=t&url=https://tempgmail.email/
temp email http://maps.google.gp/url?sa=t&url=https://tempgmail.email/
tempmail http://images.google.gp/url?sa=t&url=https://tempgmail.email/
tempemail http://www.google.com.et/url?sa=t&url=https://tempgmail.email/
free temp mail http://maps.google.com.et/url?sa=t&url=https://tempgmail.email/
temp-mail http://images.google.com.et/url?sa=t&url=https://tempgmail.email/
temp-gmail http://www.google.co.mz/url?sa=t&url=https://tempgmail.email/
temp-mail-email-gmail http://images.google.co.mz/url?sa=t&url=https://tempgmail.email/
temp mail http://maps.google.co.mz/url?sa=t&url=https://tempgmail.email/
temp gmail http://images.google.com.bn/url?sa=t&url=https://tempgmail.email/
temp email https://images.google.com/url?q=https://tempgmail.email/
tempmail https://images.google.ac/url?q=https://tempgmail.email/
tempemail https://images.google.ad/url?q=https://tempgmail.email/
free temp mail https://images.google.ae/url?q=https://tempgmail.email/
temp-mail https://images.google.com.af/url?q=https://tempgmail.email/
temp-gmail https://images.google.com.ag/url?q=https://tempgmail.email/
temp-mail-email-gmail https://images.google.com.ai/url?q=https://tempgmail.email/
temp mail https://images.google.al/url?q=https://tempgmail.email/
temp-mail-email-gmail https://images.google.am/url?q=https://tempgmail.email/
temp mail https://images.google.co.ao/url?q=https://tempgmail.email/
temp gmail https://images.google.com.ar/url?q=https://tempgmail.email/
temp email https://images.google.as/url?q=https://tempgmail.email/
tempmail https://images.google.at/url?q=https://tempgmail.email/
tempemail https://images.google.com.au/url?q=https://tempgmail.email/
free temp mail https://images.google.az/url?q=https://tempgmail.email/
temp-mail https://images.google.ba/url?q=https://tempgmail.email/
temp-gmail https://images.google.com.bd/url?q=https://tempgmail.email/
temp-mail-email-gmail https://images.google.be/url?q=https://tempgmail.email/
temp mail https://images.google.bf/url?q=https://tempgmail.email/
temp gmail https://images.google.bg/url?q=https://tempgmail.email/
temp email https://images.google.com.bh/url?q=https://tempgmail.email/
tempmail https://images.google.bi/url?q=https://tempgmail.email/
tempemail https://images.google.bj/url?q=https://tempgmail.email/
free temp mail https://images.google.com.bn/url?q=https://tempgmail.email/
temp-mail https://images.google.com.bo/url?q=https://tempgmail.email/
temp-gmail https://images.google.com.br/url?q=https://tempgmail.email/
temp-mail-email-gmail https://images.google.bs/url?q=https://tempgmail.email/
temp mail https://images.google.bt/url?q=https://tempgmail.email/
temp gmail https://images.google.co.bw/url?q=https://tempgmail.email/
temp email https://images.google.by/url?q=https://tempgmail.email/
tempmail https://images.google.com.bz/url?q=https://tempgmail.email/
tempemail https://images.google.ca/url?q=https://tempgmail.email/
free temp mail https://images.google.com.kh/url?q=https://tempgmail.email/
temp-mail https://images.google.cc/url?q=https://tempgmail.email/
temp-gmail https://images.google.cd/url?q=https://tempgmail.email/
temp-mail-email-gmail https://images.google.cf/url?q=https://tempgmail.email/
temp mail https://images.google.cat/url?q=https://tempgmail.email/
temp-mail-email-gmail https://images.google.cg/url?q=https://tempgmail.email/
temp mail https://images.google.ch/url?q=https://tempgmail.email/
temp gmail https://images.google.ci/url?q=https://tempgmail.email/
temp email https://images.google.co.ck/url?q=https://tempgmail.email/
tempmail https://images.google.cl/url?q=https://tempgmail.email/
tempemail https://images.google.cm/url?q=https://tempgmail.email/
free temp mail https://images.google.cn/url?q=https://tempgmail.email/
temp-mail https://images.google.com.co/url?q=https://tempgmail.email/
temp-gmail https://images.google.co.cr/url?q=https://tempgmail.email/
temp-mail-email-gmail https://images.google.com.cu/url?q=https://tempgmail.email/
temp mail https://images.google.cv/url?q=https://tempgmail.email/
temp gmail https://images.google.com.cy/url?q=https://tempgmail.email/
temp email https://www.youtube.com/redirect?q=https://tempgmail.email/
tempmail https://www.google.com/url?sa=t&url=https://tempgmail.email/
tempemail https://maps.google.com/url?sa=t&url=https://tempgmail.email/
free temp mail https://images.google.de/url?sa=t&url=https://tempgmail.email/
temp-mail https://maps.google.de/url?sa=t&url=https://tempgmail.email/
temp-gmail https://www.google.de/url?sa=t&url=https://tempgmail.email/
temp-mail-email-gmail https://m.ok.ru/dk?st.cmd=outLinkWarning&st.rfn=https://tempgmail.email/
temp mail https://images.google.co.uk/url?sa=t&url=https://tempgmail.email/
temp gmail https://maps.google.co.uk/url?sa=t&url=https://tempgmail.email/
temp email https://www.google.co.uk/url?sa=t&url=https://tempgmail.email/
tempmail https://maps.google.co.jp/url?sa=t&url=https://tempgmail.email/
tempemail https://images.google.co.jp/url?sa=t&url=https://tempgmail.email/
free temp mail https://www.google.co.jp/url?sa=t&url=https://tempgmail.email/
temp-mail https://hub.wiley.com/%E2%80%8Eexternal-link.jspa?url=https://tempgmail.email/
temp-gmail https://www.google.fr/url?sa=t&url=https://tempgmail.email/
temp-mail-email-gmail https://images.google.fr/url?sa=t&url=https://tempgmail.email/
temp mail https://maps.google.fr/url?sa=t&url=https://tempgmail.email/
temp-mail-email-gmail https://maps.google.es/url?sa=t&url=https://tempgmail.email/
temp mail https://images.google.es/url?sa=t&url=https://tempgmail.email/
temp gmail https://www.google.es/url?sa=t&url=https://tempgmail.email/
temp email https://images.google.it/url?sa=t&url=https://tempgmail.email/
tempmail https://www.google.it/url?sa=t&url=https://tempgmail.email/
tempemail https://maps.google.it/url?sa=t&url=https://tempgmail.email/
free temp mail https://images.google.com.br/url?sa=t&url=https://tempgmail.email/
temp-mail https://www.google.com.br/url?sa=t&url=https://tempgmail.email/
temp-gmail https://maps.google.com.br/url?sa=t&url=https://tempgmail.email/
temp-mail-email-gmail https://www.linkwithin.com/install?platform=blogger&site_id=2069303&url=https://tempgmail.email/
temp mail https://maps.google.ca/url?sa=t&url=https://tempgmail.email/
temp gmail https://images.google.ca/url?sa=t&url=https://tempgmail.email/
temp email https://www.google.ca/url?sa=t&url=https://tempgmail.email/
tempmail https://images.google.com.hk/url?sa=t&url=https://tempgmail.email/
tempemail https://www.google.com.hk/url?sa=t&url=https://tempgmail.email/
free temp mail https://maps.google.com.hk/url?sa=t&url=https://tempgmail.email/
temp-mail https://www.google.nl/url?sa=t&url=https://tempgmail.email/
temp-gmail https://maps.google.nl/url?sa=t&url=https://tempgmail.email/
temp-mail-email-gmail https://images.google.nl/url?sa=t&url=https://tempgmail.email/
temp mail https://maps.google.co.in/url?sa=t&url=https://tempgmail.email/
temp gmail https://images.google.co.in/url?sa=t&url=https://tempgmail.email/
temp email https://www.google.co.in/url?sa=t&url=https://tempgmail.email/
tempmail https://maps.google.ru/url?sa=t&url=https://tempgmail.email/
tempemail https://www.google.ru/url?sa=t&url=https://tempgmail.email/
free temp mail https://images.google.ru/url?sa=t&url=https://tempgmail.email/
temp-mail https://blogs.rtve.es/libs/getfirma_footer_prod.php?blogurl=https://tempgmail.email/
temp-gmail https://maps.google.pl/url?sa=t&url=https://tempgmail.email/
temp-mail-email-gmail https://www.google.pl/url?sa=t&url=https://tempgmail.email/
temp mail https://images.google.pl/url?sa=t&url=https://tempgmail.email/
temp-mail-email-gmail https://www.google.com.au/url?sa=t&url=https://tempgmail.email/
temp mail https://maps.google.com.au/url?sa=t&url=https://tempgmail.email/
temp gmail https://images.google.com.au/url?sa=t&url=https://tempgmail.email/
temp email https://webfeeds.brookings.edu/%7E/t/0/0/%7Ehttps://tempgmail.email/
tempmail https://images.google.com.tw/url?sa=t&url=https://tempgmail.email/
tempemail https://www.google.com.tw/url?sa=t&url=https://tempgmail.email/
free temp mail https://maps.google.com.tw/url?sa=t&url=https://tempgmail.email/
temp-mail https://www.fhwa.dot.gov/reauthorization/reauexit.cfm?link=https://tempgmail.email/
temp-gmail https://maps.google.co.id/url?sa=t&url=https://tempgmail.email/
temp-mail-email-gmail https://images.google.co.id/url?sa=t&url=https://tempgmail.email/
temp mail https://www.google.co.id/url?sa=t&url=https://tempgmail.email/
temp gmail https://images.google.com/url?sa=t&url=https://tempgmail.email/
temp email https://m.odnoklassniki.ru/dk?st.cmd=outLinkWarning&st.rfn=https://tempgmail.email/
tempmail https://images.google.be/url?sa=t&url=https://tempgmail.email/
tempemail https://maps.google.be/url?sa=t&url=https://tempgmail.email/
free temp mail https://www.google.be/url?sa=t&url=https://tempgmail.email/
temp-mail https://images.google.at/url?sa=t&url=https://tempgmail.email/
temp-gmail https://maps.google.at/url?sa=t&url=https://tempgmail.email/
temp-mail-email-gmail https://www.google.at/url?sa=t&url=https://tempgmail.email/
temp mail https://www.google.cz/url?sa=t&url=https://tempgmail.email/
temp gmail https://maps.google.cz/url?sa=t&url=https://tempgmail.email/
temp email https://images.google.ch/url?sa=t&url=https://tempgmail.email/
tempmail https://www.google.ch/url?sa=t&url=https://tempgmail.email/
tempemail https://maps.google.ch/url?sa=t&url=https://tempgmail.email/
free temp mail https://images.google.com.ua/url?sa=t&url=https://tempgmail.email/
temp-mail https://maps.google.com.ua/url?sa=t&url=https://tempgmail.email/
temp-gmail https://www.google.com.ua/url?sa=t&url=https://tempgmail.email/
temp-mail-email-gmail https://www.google.com.tr/url?sa=t&url=https://tempgmail.email/
temp mail https://maps.google.com.tr/url?sa=t&url=https://tempgmail.email/
temp-mail-email-gmail https://images.google.com.tr/url?sa=t&url=https://tempgmail.email/
temp mail https://www2.ogs.state.ny.us/help/urlstatusgo.html?url=https://tempgmail.email/
temp gmail https://ds.jpn.org/jump.php?url=https://tempgmail.email/
temp email https://maps.google.com.mx/url?sa=t&url=https://tempgmail.email/
tempmail https://www.google.com.mx/url?sa=t&url=https://tempgmail.email/
tempemail https://images.google.com.mx/url?sa=t&url=https://tempgmail.email/
free temp mail https://www.google.dk/url?sa=t&url=https://tempgmail.email/
temp-mail https://images.google.dk/url?sa=t&url=https://tempgmail.email/
temp-gmail https://maps.google.dk/url?sa=t&url=https://tempgmail.email/
temp-mail-email-gmail https://www.google.fi/url?sa=t&url=https://tempgmail.email/
temp mail https://images.google.fi/url?sa=t&url=https://tempgmail.email/
temp gmail https://maps.google.fi/url?sa=t&url=https://tempgmail.email/
temp email https://community.nfpa.org/external-link.jspa?url=https://tempgmail.email/
tempmail https://images.google.co.nz/url?sa=t&url=https://tempgmail.email/
tempemail https://www.google.co.nz/url?sa=t&url=https://tempgmail.email/
free temp mail https://maps.google.co.nz/url?sa=t&url=https://tempgmail.email/
temp-mail https://www.scire.coppe.ufrj.br/atrio_change_url/url-redir.php?url=https://tempgmail.email/
temp-gmail https://community.esri.com/external-link.jspa?url=https://tempgmail.email/
temp-mail-email-gmail https://images.google.co.th/url?sa=t&url=https://tempgmail.email/
temp mail https://maps.google.co.th/url?sa=t&url=https://tempgmail.email/
temp gmail https://www.google.co.th/url?sa=t&url=https://tempgmail.email/
temp email https://images.google.com.vn/url?sa=t&url=https://tempgmail.email/
tempmail https://www.google.com.vn/url?sa=t&url=https://tempgmail.email/
tempemail https://www.google.pt/url?sa=t&url=https://tempgmail.email/
free temp mail https://maps.google.pt/url?sa=t&url=https://tempgmail.email/
temp-mail https://images.google.pt/url?sa=t&url=https://tempgmail.email/
temp-gmail https://jump.2ch.net/?https://tempgmail.email/
temp-mail-email-gmail https://community.macmillan.com/external-link.jspa?url=https://tempgmail.email/
temp mail https://maps.google.ro/url?sa=t&url=https://tempgmail.email/
temp-mail-email-gmail https://www.google.ro/url?sa=t&url=https://tempgmail.email/
temp mail https://images.google.ro/url?sa=t&url=https://tempgmail.email/
temp gmail https://images.google.com.my/url?sa=t&url=https://tempgmail.email/
temp email https://maps.google.com.my/url?sa=t&url=https://tempgmail.email/
tempmail https://www.google.com.my/url?sa=t&url=https://tempgmail.email/
tempemail https://www.google.co.za/url?sa=t&url=https://tempgmail.email/
free temp mail https://images.google.co.za/url?sa=t&url=https://tempgmail.email/
temp-mail https://maps.google.co.za/url?sa=t&url=https://tempgmail.email/
temp-gmail https://www.google.com.sg/url?sa=t&url=https://tempgmail.email/
temp-mail-email-gmail https://maps.google.com.sg/url?sa=t&url=https://tempgmail.email/
temp mail https://images.google.com.sg/url?sa=t&url=https://tempgmail.email/
temp gmail https://club.panasonic.jp/member/terms/?siteId=B1&returnURL=https://tempgmail.email/
temp email https://images.google.gr/url?sa=t&url=https://tempgmail.email/
tempmail https://maps.google.gr/url?sa=t&url=https://tempgmail.email/
tempemail https://www.google.gr/url?sa=t&url=https://tempgmail.email/
free temp mail https://www.google.hu/url?sa=t&url=https://tempgmail.email/
temp-mail https://maps.google.hu/url?sa=t&url=https://tempgmail.email/
temp-gmail https://images.google.hu/url?sa=t&url=https://tempgmail.email/
temp-mail-email-gmail https://maps.google.co.il/url?sa=t&url=https://tempgmail.email/
temp mail https://images.google.co.il/url?sa=t&url=https://tempgmail.email/
temp gmail https://www.google.co.il/url?sa=t&url=https://tempgmail.email/
temp email https://www.google.cl/url?sa=t&url=https://tempgmail.email/
tempmail https://images.google.cl/url?sa=t&url=https://tempgmail.email/
tempemail https://maps.google.cl/url?sa=t&url=https://tempgmail.email/
free temp mail https://maps.google.ie/url?sa=t&url=https://tempgmail.email/
temp-mail https://www.google.ie/url?sa=t&url=https://tempgmail.email/
temp-gmail https://images.google.ie/url?sa=t&url=https://tempgmail.email/
temp-mail-email-gmail https://images.google.bg/url?sa=t&url=https://tempgmail.email/
temp mail https://www.google.bg/url?sa=t&url=https://tempgmail.email/
temp-mail-email-gmail https://maps.google.bg/url?sa=t&url=https://tempgmail.email/
temp mail https://community.nxp.com/external-link.jspa?url=https://tempgmail.email/
temp gmail http://transtats.bts.gov/exit.asp?url=https://tempgmail.email/
temp email http://latinamerica.brother.com/LeavingOurSite.aspx?goto=https://tempgmail.email/
tempmail https://workable.com/nr?l=https://tempgmail.email/
tempemail https://register.scotland.gov.uk/subscribe/widgetsignup?url=https://tempgmail.email/
free temp mail https://images.google.com.co/url?sa=t&url=https://tempgmail.email/
temp-mail https://maps.google.com.co/url?sa=t&url=https://tempgmail.email/
temp-gmail https://www.google.com.co/url?sa=t&url=https://tempgmail.email/
temp-mail-email-gmail https://www.google.sk/url?sa=t&url=https://tempgmail.email/
temp mail https://maps.google.sk/url?sa=t&url=https://tempgmail.email/
temp gmail https://images.google.sk/url?sa=t&url=https://tempgmail.email/
temp email https://forum.solidworks.com/external-link.jspa?url=https://tempgmail.email/
tempmail https://maps.google.lt/url?sa=t&url=https://tempgmail.email/
tempemail https://ref.gamer.com.tw/redir.php?url=https://tempgmail.email/
free temp mail https://images.google.com.sa/url?sa=t&url=https://tempgmail.email/
temp-mail https://maps.google.com.sa/url?sa=t&url=https://tempgmail.email/
temp-gmail https://www.google.com.sa/url?sa=t&url=https://tempgmail.email/
temp-mail-email-gmail https://images.google.hr/url?sa=t&url=https://tempgmail.email/
temp mail https://www.google.hr/url?sa=t&url=https://tempgmail.email/
temp gmail https://maps.google.hr/url?sa=t&url=https://tempgmail.email/
temp email https://images.google.com.pe/url?sa=t&url=https://tempgmail.email/
tempmail https://www.google.com.pe/url?sa=t&url=https://tempgmail.email/
tempemail https://maps.google.ae/url?sa=t&url=https://tempgmail.email/
free temp mail https://images.google.ae/url?sa=t&url=https://tempgmail.email/
temp-mail https://www.google.ae/url?sa=t&url=https://tempgmail.email/
temp-gmail https://www.google.co.ve/url?sa=t&url=https://tempgmail.email/
temp-mail-email-gmail https://maps.google.co.ve/url?sa=t&url=https://tempgmail.email/
temp mail https://images.google.co.ve/url?sa=t&url=https://tempgmail.email/
temp-mail-email-gmail http://onlinemanuals.txdot.gov/help/urlstatusgo.html?url=https://tempgmail.email/
temp mail https://www.google.com.pk/url?sa=t&url=https://tempgmail.email/
temp gmail https://images.google.com.pk/url?sa=t&url=https://tempgmail.email/
temp email https://community.rsa.com/t5/custom/page/page-id/ExternalRedirect?url=https://tempgmail.email/
tempmail https://www.google.com.eg/url?sa=t&url=https://tempgmail.email/
tempemail https://maps.google.com.eg/url?sa=t&url=https://tempgmail.email/
free temp mail https://images.google.com.eg/url?sa=t&url=https://tempgmail.email/
temp-mail https://www.google.si/url?sa=t&url=https://tempgmail.email/
temp-gmail https://maps.google.si/url?sa=t&url=https://tempgmail.email/
temp-mail-email-gmail https://images.google.si/url?sa=t&url=https://tempgmail.email/
temp mail http://www.pickyourown.org/articles.php?NAME=Visit+Us&URL=https://tempgmail.email/
temp gmail https://maps.google.lv/url?sa=t&url=https://tempgmail.email/
temp email https://www.google.lv/url?sa=t&url=https://tempgmail.email/
tempmail https://images.google.lv/url?sa=t&url=https://tempgmail.email/
tempemail https://www.google.ee/url?sa=t&url=https://tempgmail.email/
free temp mail https://maps.google.ee/url?sa=t&url=https://tempgmail.email/
temp-mail https://images.google.ee/url?sa=t&url=https://tempgmail.email/
temp-gmail https://www.google.com.ng/url?sa=t&url=https://tempgmail.email/
temp-mail-email-gmail https://images.google.com.ng/url?sa=t&url=https://tempgmail.email/
temp mail https://www.google.by/url?sa=t&url=https://tempgmail.email/
temp gmail http://bukkit.org/proxy.php?link=https://tempgmail.email/
temp email https://images.google.com.ec/url?sa=t&url=https://tempgmail.email/
tempmail https://www.google.com.ec/url?sa=t&url=https://tempgmail.email/
tempemail http://client.paltalk.com/client/webapp/client/External.wmt?url=https://tempgmail.email/
free temp mail https://maps.google.ba/url?sa=t&url=https://tempgmail.email/
temp-mail https://foro.infojardin.com/proxy.php?link=https://tempgmail.email/
temp-gmail https://www.curseforge.com/linkout?remoteUrl=https://tempgmail.email/
temp-mail-email-gmail https://www.google.com.bd/url?sa=t&url=https://tempgmail.email/
temp mail https://images.google.com.bd/url?sa=t&url=https://tempgmail.email/
temp-mail-email-gmail http://legacy.aom.org/verifymember.asp?nextpage=https://tempgmail.email/
temp mail https://images.google.com.do/url?sa=t&url=https://tempgmail.email/
temp gmail https://maps.google.com.do/url?sa=t&url=https://tempgmail.email/
temp email https://www.google.com.do/url?sa=t&url=https://tempgmail.email/
tempmail http://www.aastocks.com/sc/changelang.aspx?lang=sc&url=https://tempgmail.email/
tempemail https://images.google.lk/url?sa=t&url=https://tempgmail.email/
free temp mail https://www.google.com.pr/url?sa=t&url=https://tempgmail.email/
temp-mail https://images.google.com.gt/url?sa=t&url=https://tempgmail.email/
temp-gmail https://maps.google.com.gt/url?sa=t&url=https://tempgmail.email/
temp-mail-email-gmail https://images.google.co.cr/url?sa=t&url=https://tempgmail.email/
temp mail https://maps.google.co.cr/url?sa=t&url=https://tempgmail.email/
temp gmail https://maps.google.lu/url?sa=t&url=https://tempgmail.email/
temp email https://images.google.lu/url?sa=t&url=https://tempgmail.email/
tempmail https://www.google.lu/url?sa=t&url=https://tempgmail.email/
tempemail https://images.google.com.kw/url?sa=t&url=https://tempgmail.email/
free temp mail https://www.google.com.kw/url?sa=t&url=https://tempgmail.email/
temp-mail https://www.flashback.org/leave.php?u=https://tempgmail.email/
temp-gmail https://www.google.is/url?sa=t&url=https://tempgmail.email/
temp-mail-email-gmail https://www.google.dz/url?sa=t&url=https://tempgmail.email/
temp mail https://anonym.to/?https://tempgmail.email/
temp gmail https://www.google.tn/url?sa=t&url=https://tempgmail.email/
temp email https://maps.google.tn/url?sa=t&url=https://tempgmail.email/
tempmail https://www.talgov.com/Main/exit.aspx?url=https://tempgmail.email/
tempemail https://maps.google.com.np/url?sa=t&url=https://tempgmail.email/
free temp mail https://images.google.com.np/url?sa=t&url=https://tempgmail.email/
temp-mail https://www.google.com.np/url?sa=t&url=https://tempgmail.email/
temp-gmail https://www.google.kz/url?sa=t&url=https://tempgmail.email/
temp-mail-email-gmail https://www.google.co.ma/url?sa=t&url=https://tempgmail.email/
temp mail https://maps.google.ge/url?sa=t&url=https://tempgmail.email/
temp-mail-email-gmail https://maps.google.com.ni/url?sa=t&url=https://tempgmail.email/
temp mail https://www.google.com.jm/url?sa=t&url=https://tempgmail.email/
temp gmail https://images.google.com.cy/url?sa=t&url=https://tempgmail.email/
temp email https://images.google.com.qa/url?sa=t&url=https://tempgmail.email/
tempmail https://www.google.com.sv/url?sa=t&url=https://tempgmail.email/
tempemail https://images.google.com.sv/url?sa=t&url=https://tempgmail.email/
free temp mail https://smccd.edu/disclaimer/redirect.php?url=https://tempgmail.email/
temp-mail https://images.google.ps/url?sa=t&url=https://tempgmail.email/
temp-gmail https://maps.google.com.bo/url?sa=t&url=https://tempgmail.email/
temp-mail-email-gmail https://images.google.li/url?sa=t&url=https://tempgmail.email/
temp mail https://maps.google.mn/url?sa=t&url=https://tempgmail.email/
temp gmail https://images.google.mn/url?sa=t&url=https://tempgmail.email/
temp email https://www.google.com.bh/url?sa=t&url=https://tempgmail.email/
tempmail https://maps.google.com.bh/url?sa=t&url=https://tempgmail.email/
tempemail https://images.google.com.kh/url?sa=t&url=https://tempgmail.email/
free temp mail https://www.google.com.kh/url?sa=t&url=https://tempgmail.email/
temp-mail https://maps.google.com.lb/url?sa=t&url=https://tempgmail.email/
temp-gmail https://images.google.com.lb/url?sa=t&url=https://tempgmail.email/
temp-mail-email-gmail https://www.google.tt/url?sa=t&url=https://tempgmail.email/
temp mail https://images.google.tt/url?sa=t&url=https://tempgmail.email/
temp gmail https://maps.google.hn/url?sa=t&url=https://tempgmail.email/
temp email https://www.google.hn/url?sa=t&url=https://tempgmail.email/
tempmail https://www.google.sn/url?sa=t&url=https://tempgmail.email/
tempemail https://maps.google.sn/url?sa=t&url=https://tempgmail.email/
free temp mail https://www.google.com.mt/url?sa=t&url=https://tempgmail.email/
temp-mail https://maps.google.com.mt/url?sa=t&url=https://tempgmail.email/
temp-gmail https://maps.google.iq/url?sa=t&url=https://tempgmail.email/
temp-mail-email-gmail https://images.google.iq/url?sa=t&url=https://tempgmail.email/
temp mail https://images.google.cat/url?sa=t&url=https://tempgmail.email/
temp-mail-email-gmail https://images.google.cm/url?sa=t&url=https://tempgmail.email/
temp mail https://maps.google.co.ug/url?sa=t&url=https://tempgmail.email/
temp gmail https://images.google.com.ly/url?sa=t&url=https://tempgmail.email/
temp email https://www.google.as/url?sa=t&url=https://tempgmail.email/
tempmail https://images.google.com.pa/url?sa=t&url=https://tempgmail.email/
tempemail https://maps.google.com.pa/url?sa=t&url=https://tempgmail.email/
free temp mail https://images.google.la/url?sa=t&url=https://tempgmail.email/
temp-mail https://maps.google.com.om/url?sa=t&url=https://tempgmail.email/
temp-gmail https://images.google.com.om/url?sa=t&url=https://tempgmail.email/
temp-mail-email-gmail https://images.google.co.mz/url?sa=t&url=https://tempgmail.email/
temp mail https://www.google.co.bw/url?sa=t&url=https://tempgmail.email/
temp gmail https://www.google.ms/url?sa=t&url=https://tempgmail.email/
temp email https://www.google.ci/url?sa=t&url=https://tempgmail.email/
tempmail https://images.google.kg/url?sa=t&url=https://tempgmail.email/
tempemail https://www.google.com.bz/url?sa=t&url=https://tempgmail.email/
free temp mail https://maps.google.com.bz/url?sa=t&url=https://tempgmail.email/
temp-mail https://images.google.com.bn/url?sa=t&url=https://tempgmail.email/
temp-gmail https://images.google.sh/url?sa=t&url=https://tempgmail.email/
temp-mail-email-gmail https://maps.google.bi/url?sa=t&url=https://tempgmail.email/
temp mail https://images.google.bi/url?sa=t&url=https://tempgmail.email/
temp gmail https://images.google.com.ag/url?sa=t&url=https://tempgmail.email/
temp email https://www.google.com.ag/url?sa=t&url=https://tempgmail.email/
tempmail https://maps.google.cd/url?sa=t&url=https://tempgmail.email/
tempemail https://www.google.fm/url?sa=t&url=https://tempgmail.email/
free temp mail https://maps.google.fm/url?sa=t&url=https://tempgmail.email/
temp-mail https://images.google.fm/url?sa=t&url=https://tempgmail.email/
temp-gmail http://www.prairiestateoutdoors.com/?URL=https://tempgmail.email/
temp-mail-email-gmail https://live.warthunder.com/away/?to=https://tempgmail.email/
temp mail http://sinp.msu.ru/ru/ext_link?url=https://tempgmail.email/
temp-mail-email-gmail https://chofu.keizai.biz/https://tempgmail.email/
temp mail http://go.infomine.com/?re=121&tg=https://tempgmail.email/
temp gmail http://novacat.nova.edu/showres?url=https://tempgmail.email/
temp email https://cineuropa.org/el.aspx?el=https://tempgmail.email/
tempmail http://1gr.cz/log/redir.aspx?r=rajce_like&url=https://tempgmail.email/
tempemail http://busy.org/exit?url=https://tempgmail.email/
free temp mail http://www.fito.nnov.ru/go.php?url=https://tempgmail.email/
temp-mail https://via-midgard.com/index.php?do=away&url=https://tempgmail.email/
temp-gmail https://t.me/iv?url=https://tempgmail.email/
temp-mail-email-gmail http://web.stanford.edu/cgi-bin/redirect?dest=https://tempgmail.email/
temp mail http://wasearch.loc.gov/e2k//https://tempgmail.email/
temp gmail https://ogs.state.ny.us/help/urlstatusgo.html?url=https://tempgmail.email/
temp email https://optimize.viglink.com/page/pmv?url=https://tempgmail.email/
tempmail https://li137-108.members.linode.com/redirect/?url=https://tempgmail.email/
tempemail http://georgewbushlibrary.smu.edu/exit.aspx?url=https://tempgmail.email/
free temp mail https://es.chaturbate.com/external_link/?url=https://tempgmail.email/
temp-mail https://galter.northwestern.edu/exit?url=https://tempgmail.email/
temp-gmail https://addons.mozilla.org/en-US/firefox/search/?q=https://tempgmail.email/
temp-mail-email-gmail https://medium.com/search?q=https://tempgmail.email/
temp mail https://www.slideshare.net/search/slideshow?searchfrom=header&q=https://tempgmail.email/
temp gmail https://archive.org/search.php?query=https://tempgmail.email/
temp email https://issuu.com/search?q=https://tempgmail.email/
tempmail https://www.scribd.com/search?content_type=tops&page=1&query=https://tempgmail.email/
tempemail https://themeforest.net/search/https://tempgmail.email/
free temp mail https://www.4shared.com/web/q/#query=https://tempgmail.email/
temp-mail https://www.buzzfeed.com/search?q=https://tempgmail.email/
temp-gmail https://www.goodreads.com/search?utf8=%E2%9C%93&query=https://tempgmail.email/
temp-mail-email-gmail https://www.tripadvisor.com/Search?q=https://tempgmail.email/
temp mail https://www.wattpad.com/search/https://tempgmail.email/
temp-mail-email-gmail https://www.crunchyroll.com/search?from=&q=https://tempgmail.email/
temp mail https://www.coursera.org/search?query=https://tempgmail.email/
temp gmail https://www.discogs.com/search/?q=https://tempgmail.email/
temp email https://www.livejournal.com/rsearch?q=https://tempgmail.email/
tempmail https://www.reverbnation.com/main/search?q=https://tempgmail.email/
tempemail https://www.flickr.com/search/?text=https://tempgmail.email/
free temp mail https://discover.hubpages.com/search?query=https://tempgmail.email/
temp-mail https://audioboom.com/search?q=https://tempgmail.email/
temp-gmail https://seekingalpha.com/search?q=https://tempgmail.email/
temp-mail-email-gmail https://www.dpreview.com/search?query=https://tempgmail.email/
temp mail https://www.minds.com/discovery/search?q=https://tempgmail.email/
temp gmail https://www.producthunt.com/search?q=https://tempgmail.email/
temp email https://www.codecademy.com/search?query=https://tempgmail.email/
tempmail http://www.authorstream.com/tag/https://tempgmail.email/
tempemail https://500px.com/search?q=https://tempgmail.email/
free temp mail https://www.pbase.com/search?q=https://tempgmail.email/
temp-mail https://www.deviantart.com/search?q=https://tempgmail.email/
temp-gmail https://coub.com/search?q=https://tempgmail.email/
temp-mail-email-gmail https://www.magcloud.com/browse/search?q=https://tempgmail.email/
temp mail https://www.mediapost.com/publications/search/?q=https://tempgmail.email/
temp gmail https://www.bloglovin.com/search?q=https://tempgmail.email/&search_term=https://tempgmail.email/
temp email https://casa.abril.com.br/?s=https://tempgmail.email/
tempmail https://www.diigo.com/profile/artisanmarket?query=https://tempgmail.email/
tempemail https://trove.nla.gov.au/search/category/websites?keyword=https://tempgmail.email/
free temp mail https://www.selfgrowth.com/search/google?query=https://tempgmail.email/
temp-mail https://brandyourself.com/blog/?s=https://tempgmail.email/
temp-gmail https://bilalarticles.com/?s=https://tempgmail.email/
temp-mail-email-gmail https://globalgenes.happyfox.com/kb/search/?q=https://tempgmail.email/
temp mail https://ao.23video.com/search/perform?search=https://tempgmail.email/
temp-mail-email-gmail http://ttlink.com/search/notice?q=https://tempgmail.email/
temp mail https://faucre.com/?s=https://tempgmail.email/
temp gmail https://xyloyl.com/?s=https://tempgmail.email/
temp email https://www.onfeetnation.com/main/search/search?q=https://tempgmail.email/
tempmail https://farleu.com/?s=https://tempgmail.email/
tempemail https://www.worldwidetopsite.link/search?q=https://tempgmail.email/
free temp mail https://www.zupyak.com/search?q=https://tempgmail.email/
temp-mail https://leoville.withknown.com/content/all/?q=https://tempgmail.email/
temp-gmail https://lauramarie204.withknown.com/content/all/?q=https://tempgmail.email/
temp-mail-email-gmail https://drikkes.withknown.com/content/all/?q=https://tempgmail.email/
temp mail https://technologyenhancingedu.withknown.com/content/all/?q=https://tempgmail.email/
temp gmail https://kinlane.withknown.com/content/all/?q=https://tempgmail.email/
temp email https://simon.withknown.com/content/all/?q=https://tempgmail.email/
tempmail https://blogging.withknown.com/content/all/?q=https://tempgmail.email/
tempemail https://gilrg18.withknown.com/content/all/?q=https://tempgmail.email/
free temp mail https://ga.videomarketingplatform.co/search/perform?search=https://tempgmail.email/
temp-mail https://jbf4093j.videomarketingplatform.co/search/perform?search=https://tempgmail.email/
temp-gmail https://washerrange07.werite.net/?q=https://tempgmail.email/
temp-mail-email-gmail https://altobaby3.werite.net/?q=https://tempgmail.email/
temp mail https://hempletter8.werite.net/?q=https://tempgmail.email/
temp gmail https://dollarturret20.bravejournal.net/?q=https://tempgmail.email/
temp email https://rifleheaven0.bravejournal.net/?q=https://tempgmail.email/
tempmail https://layertown6.bravejournal.net/?q=https://tempgmail.email/
tempemail https://handlebail05.bravejournal.net/?q=https://tempgmail.email/
free temp mail https://www.coupondunia.in/blog/?s=https://tempgmail.email/
temp-mail https://www.australiantraveller.com/search/?search_term=https://tempgmail.email/
temp-gmail https://www.sitelike.org/similar/ce-top10.com/
temp-mail-email-gmail https://webhitlist.com/main/search/search?q=https://tempgmail.email/
temp mail https://articlescad.com/article_search.php?keyword=https://tempgmail.email/
temp-mail-email-gmail https://yercum.com/?s=66666
temp mail https://statvoo.com/search/66666
temp gmail https://zoacum.com/?s=https://tempgmail.email/
temp email https://www.sitelinks.info/ce-top10.com//
tempmail https://absmho.com/?s=https://tempgmail.email/
tempemail https://www.party.biz/search?query=https://tempgmail.email/
free temp mail http://mathuncle9.blogdigy.com/?s=https://tempgmail.email/
temp-mail https://www.lifeofdad.com/?s=https://tempgmail.email/
temp-gmail http://www.fxstat.com/search?s=https://tempgmail.email/
temp-mail-email-gmail http://blowbrazil3.blogzet.com/?s=https://tempgmail.email/
temp mail https://edukite.org/?s=https://tempgmail.email/
temp gmail https://whatsondigest.com/?s=https://tempgmail.email/
temp email https://www.epubzone.org/?s=https://tempgmail.email/
tempmail https://journalistopia.com/?s=https://tempgmail.email/
tempemail http://juteeast5.blogminds.com/?s=https://tempgmail.email/
free temp mail https://dirtragdirtfest.com/?s=https://tempgmail.email/
temp-mail https://www.websiteperu.com/search/https://tempgmail.email/
temp-gmail https://growing-minds.org/?s=https://tempgmail.email/
temp-mail-email-gmail https://www.sitesimilar.net/ce-top10.com/
temp mail https://www.business2community.com/?s=https://tempgmail.email/
temp gmail https://medborgarbudget.lundby.goteborg.se/search?utf8=%E2%9C%93&term=https://tempgmail.email/
temp email http://www.rn-tp.com/search/node?keys=https://tempgmail.email/
tempmail https://bagogames.com/?s=https://tempgmail.email/
tempemail https://www.bunity.com/search?q=https://tempgmail.email/
free temp mail https://e-shopy.org/?s=https://tempgmail.email/
temp-mail http://jevois.org/qa/index.php?qa=search&q=https://tempgmail.email/
temp-gmail http://forum.fruct.org/search/node/https://tempgmail.email/
temp-mail-email-gmail https://www.desmaakvanitalie.nl/?s=https://tempgmail.email/
temp mail https://www.au-e.com/search/https://tempgmail.email/
temp-mail-email-gmail http://sites2.jf.ifsudestemg.edu.br/search/node/https://tempgmail.email/
temp mail https://webranksdirectory.com/?s=https://tempgmail.email/
temp gmail https://www.realgabinete.com.br/Resultados-Pesquisa?Search=https://tempgmail.email/
temp email http://www.kauaifoodbank.org/Search-Results?Search=https://tempgmail.email/
tempmail http://community.runanempire.com/index.php?p=%2Fsearch&Search=https://tempgmail.email/
tempemail https://bloggerbabes.com/?s=https://tempgmail.email/
free temp mail https://unitedagainsttorture.org/?s=https://tempgmail.email/
temp-mail https://www.merricksart.com/?s=https://tempgmail.email/&submit=search
temp-gmail https://shalee01.podspot.de/?s=https://tempgmail.email/
temp-mail-email-gmail https://web.colby.edu/ru127-fa2015/?s=https://tempgmail.email/
temp mail https://chrisguillebeau.com/?s=https://tempgmail.email/
temp gmail http://ajiie.lecture.ub.ac.id/?s=https://tempgmail.email/
temp email https://www.airfrov.com/?s=https://tempgmail.email/
tempmail https://corporacion.mx/?s=https://tempgmail.email/
tempemail https://italianamericanlife.com/?s=https://tempgmail.email/
free temp mail https://www.hiddenamericans.com/?s=https://tempgmail.email/
temp-mail https://editricezona.it/?s=https://tempgmail.email/
temp-gmail https://jaguda.com/?s=https://tempgmail.email/
temp-mail-email-gmail https://topwebdirectoy.com/?s=https://tempgmail.email/
temp mail https://www.updownsite.com/search/https://tempgmail.email/
temp gmail https://cpawebtrust.org/?s=https://tempgmail.email/
temp email https://ourpotluckfamily.com/?s=https://tempgmail.email/
tempmail https://webjunctiondirectory.com/?s=https://tempgmail.email/
tempemail http://paulsohn.org/?s=https://tempgmail.email/
free temp mail https://jordanseasyentertaining.com/?s=https://tempgmail.email/
temp-mail https://irakyat.my/search?query=https://tempgmail.email/
temp-gmail https://www.topdomadirectory.com/search?q=https://tempgmail.email/
temp-mail-email-gmail https://webhubdirectory.com/?s=https://tempgmail.email/
temp mail https://99sitedirectory.com/?s=https://tempgmail.email/
temp-mail-email-gmail https://topmillionwebdirectory.com/?s=https://tempgmail.email/
temp mail https://seotopdirectory.com/?s=https://tempgmail.email/
temp gmail https://sciohost.org/?s=https://tempgmail.email/
temp email https://rootwholebody.com/?s=https://tempgmail.email/
tempmail http://www.bcsnerie.com/?s=https://tempgmail.email/
tempemail https://webworthdirectory.com/?s=https://tempgmail.email/
free temp mail https://infusionsoft.makingexperience.com/?s=https://tempgmail.email/
temp-mail https://thegibraltarmagazine.com/?s=https://tempgmail.email/
temp-gmail https://raretopsitesdirectory.com/?s=https://tempgmail.email/
temp-mail-email-gmail https://www.flodaforsfarare.se/Lasta-sidor/Sok/?q=https://tempgmail.email/
temp mail https://lebanon.in.gov/?s=https://tempgmail.email/
temp gmail https://www.fashionata.com/search/node?keys=https://tempgmail.email/
temp email http://lkfeb.komunitas.uksw.edu/search/?q=https://tempgmail.email/
tempmail https://donetsk-dr.ru/?s=https://tempgmail.email/
tempemail https://www.researchgate.net/search?q=https://tempgmail.email/
free temp mail https://baefikre.blogspot.com/search?q=https://tempgmail.email/
temp-mail http://www.rn-tp.com/index.php/search/node?keys=https://tempgmail.email/
temp-gmail http://sites.gsu.edu/jsalters2/?s=https://tempgmail.email/
temp-mail-email-gmail http://www.journeysaremydiary.com/?s=https://tempgmail.email/
temp mail https://seotech2.com/?s=https://tempgmail.email/
temp gmail https://amidov.com/?s=https://tempgmail.email/
temp email https://wrencommunity.org/?s=https://tempgmail.email/
tempmail https://checkseo.in/seoranking.aspx?url=https://tempgmail.email/
tempemail https://talvinsingh.com/?s=https://tempgmail.email/
free temp mail https://sports.cwmalls.com/?s=https://tempgmail.email/
temp-mail http://www.craveeatery.com/?s=https://tempgmail.email/
temp-gmail https://dinoruss.com/?s=https://tempgmail.email/
temp-mail-email-gmail https://worldclimatecommunity.com/?s=https://tempgmail.email/
temp mail https://kolocsolidaire.org/?s=https://tempgmail.email/
temp-mail-email-gmail https://www.thaifeedmill.com/?s=https://tempgmail.email/
temp mail https://www.gcobb.com/?s=https://tempgmail.email/
temp gmail http://seotechnology.in/search.php?search=httpswwwce-top10com
temp email https://voyage-first.com/?s=https://tempgmail.email/
tempmail https://albarays.com/?s=https://tempgmail.email/
tempemail https://theredwarriorsfc.com/?s=https://tempgmail.email/
free temp mail https://www.linkedbd.com/search/https://tempgmail.email/
temp-mail http://beta.lekhafoods.com/search.aspx?search=https://tempgmail.email/
temp-gmail https://allurewebsolutions.com/?s=https://tempgmail.email/
temp-mail-email-gmail https://okuu.shop/?keyword=https://tempgmail.email/&action=search
temp mail https://shralpin.com/?s=https://tempgmail.email/
temp gmail https://jointoken.com/?s=https://tempgmail.email/
temp email https://www.mast-king.com/?s=https://tempgmail.email/
tempmail https://reinaldobessa.com.br/?s=https://tempgmail.email/
tempemail https://music-education.org/?s=https://tempgmail.email/
free temp mail https://sacemaquarterly.com/?s=https://tempgmail.email/
temp-mail https://dialang.org/?s=https://tempgmail.email/
temp-gmail https://www.microlinkinc.com/search/https://tempgmail.email/
temp-mail-email-gmail http://ricelakewis.com/?s=https://tempgmail.email/
temp mail https://bchea.org/?s=https://tempgmail.email/
temp gmail https://ngoinabox.org/?s=https://tempgmail.email/
temp email https://bosmol.com/?s=https://tempgmail.email/
tempmail https://wscsd.org/?s=https://tempgmail.email/
tempemail https://unibasq.org/?s=https://tempgmail.email/
free temp mail http://www.backpacksandbunkbeds.co.uk/?s=https://tempgmail.email/
temp-mail http://bestmitzvahs.com/?s=https://tempgmail.email/
temp-gmail http://www.audiomelody.com/?s=https://tempgmail.email/
temp-mail-email-gmail http://propick.com.au/?s=https://tempgmail.email/
temp mail https://rankmakerdirectory.com/?s=https://tempgmail.email/
temp-mail-email-gmail http://mhalc.org/?s=https://tempgmail.email/
temp mail https://bestsitesdirectory.com/?s=https://tempgmail.email/
temp gmail https://www.factfiend.com/?s=https://tempgmail.email/
temp email https://rankwebdirectory.com/?s=https://tempgmail.email/
tempmail https://clicktoselldirectoy.com/?s=https://tempgmail.email/
tempemail https://seohelperdirectory.com/?s=https://tempgmail.email/
free temp mail https://busniessdirectory.com/?s=https://tempgmail.email/
temp-mail https://webrankdirectory.com/?s=https://tempgmail.email/
temp-gmail https://listodirectory.com/?s=https://tempgmail.email/
temp-mail-email-gmail http://splen.sakura.ne.jp/project/trac.cgi/search?q=https://tempgmail.email/
temp mail http://www.dailymagazine.news/?action=newsSearch&q=https://tempgmail.email/
temp gmail https://thebigbubblemiami.com/?s=https://tempgmail.email/
temp email https://sites.gsu.edu/jsalters2/?s=https://tempgmail.email/
tempmail https://you.stonybrook.edu/brianneledda/?s=https://tempgmail.email/
tempemail https://www.learnwithplayathome.com/search?q=https://tempgmail.email/
free temp mail https://artvoice.com/?s=https://tempgmail.email/
temp-mail https://newsroom.mastercard.com/?s=https://tempgmail.email/
temp-gmail https://seekingalpha.com/search/?q=https://tempgmail.email/
temp-mail-email-gmail https://mgt.sjp.ac.lk/emv/?s=https://tempgmail.email/
temp mail http://foodformyfamily.com/?s=https://tempgmail.email/
temp gmail https://www.adpushup.com/blog/?s=https://tempgmail.email/
temp email https://www.bruceclay.com/?s=https://tempgmail.email/
tempmail https://www.sci.ku.ac.th/alumni/?s=https://tempgmail.email/
tempemail https://vierkur.blogspot.com/search?q=https://tempgmail.email/
free temp mail https://adz.ro/shortcuts/suche?q=https://tempgmail.email/
temp-mail https://www.lovehappensmag.com/blog/?s=https://tempgmail.email/
temp-gmail https://arthive.com/search/https:/www.ce-top10.com
temp-mail-email-gmail https://rainnews.com/?s=https://tempgmail.email/
temp mail https://www.bestdial.in/?s=https://tempgmail.email/
temp-mail-email-gmail https://madridsalud.es/?s=https://tempgmail.email/
temp mail https://www.historicalclimatology.com/apps/search?q=https://tempgmail.email/
temp gmail https://revistaindustrias.com/?s=https://tempgmail.email/
temp email https://www.vvhelvoirt.nl/154/uitgebreid-zoeken/?q=https://tempgmail.email/
tempmail https://www.tor.com/search-page/?s=https://tempgmail.email/
tempemail https://www.plala.or.jp/search/?keyword=https://tempgmail.email/
free temp mail https://engawa.kakaku.com/tagsearch/?Tag=https://tempgmail.email/
temp-mail https://ph.indeed.com/jobs?q=https://tempgmail.email/
temp-gmail https://tvtropes.org/pmwiki/search_result.php?q=https://tempgmail.email/
temp-mail-email-gmail https://www.panasonic.com/jp/search.html?q=https://tempgmail.email/
temp mail https://www.xerox.com/en-us/search-results#q=https://tempgmail.email/
temp gmail https://www.shinystat.com/en/home/SearchForm?Search=https://tempgmail.email/
temp email https://blog.suny.edu/?s=https://tempgmail.email/
tempmail https://www.michaellinenberger.com/blog/?s=https://tempgmail.email/
tempemail https://www.erpsoftwareblog.com/?s=https://tempgmail.email/
free temp mail https://blog.justinablakeney.com/?s=https://tempgmail.email/
temp-mail https://lifeimitatesdoodles.blogspot.com/search?q=https://tempgmail.email/
temp-gmail https://hk.seekweb.com/?q=https://tempgmail.email/
temp-mail-email-gmail https://www.ludvikaok.se/Lasta-sidor/Sok/?q=https://tempgmail.email/
temp mail https://www.norbergsfk.se/Lasta-sidor/Sok/?q=https://tempgmail.email/
temp gmail https://www.ifksunnefriidrott.com/lastasidor/Sok/?q=https://tempgmail.email/
temp email https://www.logeion.nl/zoeken?q=https://tempgmail.email/
tempmail https://www.eslovjsk.com/Lastasidor/Sok/?q=https://tempgmail.email/
tempemail https://www.radabmk.se/lastasidor/Sok/?q=https://tempgmail.email/
free temp mail https://www.azaleabk.se/varaevenemang/Azaleadagen/lastasidor/sok/?q=https://tempgmail.email/
temp-mail https://publichistory.humanities.uva.nl/?s=https://tempgmail.email/
temp-gmail https://www.nibd.edu.pk/?s=https://tempgmail.email/
temp-mail-email-gmail http://www.eatingisntcheating.co.uk/search?s=https://tempgmail.email/
temp mail https://www.korpenstorsjon.se/Lagidrotter/Fotboll/Lasta-sidor/Sok/?q=https://tempgmail.email/
temp-mail-email-gmail http://www.almacenamientoabierto.com/?s=https://tempgmail.email/
temp mail http://brokeassgourmet.com/articles?q=https://tempgmail.email/
temp gmail http://oer.moeys.gov.kh/search?q=https://tempgmail.email/
temp email https://www.ellatinoamerican.com/busqueda?s=https://tempgmail.email/
tempmail https://bigtimestrength.libsyn.com/size/5/?search=https://tempgmail.email/
tempemail https://eezee.sg/search-v2?q=https://tempgmail.email/
free temp mail https://dmv.ny.gov/forms?query=https://tempgmail.email/
temp-mail https://faq.whatsapp.com/search?query=https://tempgmail.email/
temp-gmail https://tkmk.biz/?s=https://tempgmail.email/
temp-mail-email-gmail https://www.izito.co.uk/?q=https://tempgmail.email/
temp mail https://www.cheric.org/research/search/index.php?query=https://tempgmail.email/
temp gmail http://www.pickthebrain.com/blog/?s=https://tempgmail.email/
temp email https://www.nespapool.org/Search-Results?Search=https://tempgmail.email/
tempmail https://stockholmbasket.se/sok-funktion/?ss360Query=https://tempgmail.email/
tempemail https://rumahliterasiindonesia.org/?s=https://tempgmail.email/
free temp mail http://k-mit.com/bbs/search?sfl=wr_subject%7C%7Cwr_content&sop=and&stx=https://tempgmail.email/
temp-mail http://www.omahapython.org/blog/?s=https://tempgmail.email/
temp-gmail https://search.visymo.com/?q=https://tempgmail.email/
temp-mail-email-gmail act=Search&CODE=show&searchid=9c8390834bd37da35114f5ce088166d3&search_in=posts&result_type=topics&highlite=https://tempgmail.email/
temp mail https://neilpatel.com/?s=https://tempgmail.email/
temp gmail https://www.timeshighereducation.com/search?search=https://tempgmail.email/
temp email https://yoast.com/?s=https://tempgmail.email/
tempmail https://uk.pcmag.com/se/?q=https://tempgmail.email/
tempemail https://www.tableau.com/search#q=https://tempgmail.email/
free temp mail https://www.androidcentral.com/search/visor_search/www.ce-top10.com/
temp-mail https://www.nature.com/search?q=https://tempgmail.email/
temp-gmail https://www.apple.com/us/search/https%3A-www.ce-top10.com/
temp-mail-email-gmail https://www.apa.org/search?query=https://tempgmail.email/
temp mail https://www.codeinwp.com/?s=https://tempgmail.email/
temp-mail-email-gmail https://www.techradar.com/uk/search?searchTerm=https://tempgmail.email/
temp mail https://biz30.timedoctor.com/?s=https://tempgmail.email/
temp gmail https://www.wired.com/search/?q=https://tempgmail.email/
temp email https://www.bloomberg.com/search?query=https://tempgmail.email/
tempmail https://www.iso.org/search.html?q=https://tempgmail.email/
tempemail https://ec.europa.eu/search/index.do?QueryText=https://tempgmail.email/
free temp mail https://www.investopedia.com/search?q=https://tempgmail.email/
temp-mail https://www.aspca.org/search/node?search=https://tempgmail.email/
temp-gmail https://www.ipcc.ch/search/?search=https://tempgmail.email/
temp-mail-email-gmail https://en.wikipedia.org/w/index.php?search=https://tempgmail.email/
temp mail https://www.rei.com/search?q=https://tempgmail.email/
temp gmail https://350.org/?s=https://tempgmail.email/
temp email https://www.rollingstone.com/results/#?q=https://tempgmail.email/
tempmail https://www.benefiber.com/search-results/?charset_=UTF-8&q=https://tempgmail.email/
tempemail https://www.tomsguide.com/search?searchTerm=https://tempgmail.email/
free temp mail https://www.pewresearch.org/search/https://tempgmail.email/
temp-mail https://www.hrw.org/sitesearch?search=https://tempgmail.email/
temp-gmail https://www.itprotoday.com/search/node/https://tempgmail.email/
temp-mail-email-gmail https://www.anodoslivestage.gr/?s=https://tempgmail.email/
temp mail https://www.complex.com/search?q=https://tempgmail.email/
temp gmail https://primeroab.se/?s=https://tempgmail.email/
temp email https://www.amazon.co.uk/s?k=https://tempgmail.email/
tempmail https://www.optometrytimes.com/search?searchTerm=https://tempgmail.email/
tempemail %2Ccpttestimonials&s=https://tempgmail.email/
free temp mail https://horoscopodia.virgula.com.br/?s=https://tempgmail.email/
temp-mail https://www.parroquiesterbrugent.cat/?s=https://tempgmail.email/
temp-gmail https://www.urologytimes.com/search?searchTerm=https://tempgmail.email/
temp-mail-email-gmail https://www.dermatologytimes.com/search?searchTerm=https://tempgmail.email/
temp mail http://www.hsan.com.br/?s=https://tempgmail.email/
temp-mail-email-gmail https://www.writenaregiven.com/search?q=https://tempgmail.email/
temp mail https://www.dlys-couleurs.com/blog/?s=https://tempgmail.email/
temp gmail https://birthrightforall.com/?s=https://tempgmail.email/
temp email http://fab.ait.ac.th/?s=https://tempgmail.email/
tempmail http://blogs.harvard.edu/stoptorture/?s=https://tempgmail.email/
tempemail https://www.dailymagazine.news/?action=newsSearch&q=https://tempgmail.email/
free temp mail https://www.bly.com/blog/?s=https://tempgmail.email/
temp-mail https://hawksites.newpaltz.edu/nysmtpmidhudson/?s=https://tempgmail.email/
temp-gmail http://www.seewhatshow.com/?s=https://tempgmail.email/
temp-mail-email-gmail http://iyfbodn.com/display.cfm?s=https://tempgmail.email/
temp mail https://u.osu.edu/zagorsky.1/?s=https://tempgmail.email/
temp gmail http://ncboyd.blogs.wm.edu/?s=https://tempgmail.email/
temp email https://makanaibio.com/?s=https://tempgmail.email/
tempmail http://cse.oauife.edu.ng/?s=https://tempgmail.email/
tempemail https://blogs.geelongcollege.vic.edu.au/researchproject/?s=https://tempgmail.email/
free temp mail https://www.salfordcvs.co.uk/search/node/=https://tempgmail.email/
temp-mail lovehappensmag.com/blog/?s=https://tempgmail.email/
temp-gmail https://www.spaziotennis.com/?s=https://tempgmail.email/
temp-mail-email-gmail https://ethanol.nebraska.gov/?s=https://tempgmail.email/
temp mail http://google.com/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
temp gmail http://google.ac/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
temp email http://google.ad/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
tempmail http://google.ae/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
tempemail http://google.com.af/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
free temp mail http://google.com.ag/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
temp-mail http://google.com.ai/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
temp-gmail http://google.al/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
temp-mail-email-gmail http://google.am/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
temp mail http://google.co.ao/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
temp-mail-email-gmail http://google.com.ar/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
temp mail http://google.as/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
temp gmail http://google.at/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
temp email http://google.com.au/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
tempmail http://google.az/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
tempemail http://google.ba/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
free temp mail http://google.com.bd/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
temp-mail http://google.be/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
temp-gmail http://google.bf/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
temp-mail-email-gmail http://google.bg/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
temp mail http://google.com.bh/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
temp gmail http://google.bi/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
temp email http://google.bj/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
tempmail http://google.com.bn/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
tempemail http://google.com.bo/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
free temp mail http://google.com.br/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
temp-mail http://google.bs/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
temp-gmail http://google.bt/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
temp-mail-email-gmail http://google.co.bw/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
temp mail http://google.by/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
temp gmail http://google.com.bz/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
temp email http://google.ca/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
tempmail http://google.com.kh/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
tempemail http://google.cc/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
free temp mail http://google.cd/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
temp-mail http://google.cf/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
temp-gmail http://google.cat/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
temp-mail-email-gmail http://google.cg/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
temp mail http://google.ch/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
temp-mail-email-gmail http://google.ci/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
temp mail http://google.co.ck/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
temp gmail http://google.cl/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
temp email http://google.cm/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
tempmail http://google.cn/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
tempemail http://google.com.co/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
free temp mail http://google.co.cr/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
temp-mail http://google.com.cu/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
temp-gmail http://google.cv/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
temp-mail-email-gmail http://google.com.cy/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
temp mail http://google.cz/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
temp gmail http://google.de/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
temp email http://google.dj/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
tempmail http://google.dk/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
tempemail http://google.dm/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
free temp mail http://google.com.do/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
temp-mail http://google.dz/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
temp-gmail http://google.com.ec/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
temp-mail-email-gmail http://google.ee/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
temp mail http://google.com.eg/url?sr=1&ct2=jp/0_0_s_0_1_a&sa=t&usg=AFQjCNHJ_EDQ-P32EiJs6GJXly0yVYLfVg&cid=52779144202765&url=https://tempgmail.email/
temp gmail https… https://www.google.com.tw/url?sa=t&url=https://tempgmail.email/
temp email http://wikimapia.org/external_link?url=https://tempgmail.email/
tempmail https://www.kaskus.co.id/redirect?url=https://tempgmail.email/
tempemail https://community.esri.com/?url=https://tempgmail.email/
free temp mail http://rosreestr.ru/bitrix/redirect.php?goto=https://tempgmail.email/
temp-mail https://rssfeeds.wkyc.com/%7E/t/0/0/wkyc/news/%7Ehttps://tempgmail.email/
temp-gmail https://rssfeeds.wfaa.com/%7E/t/0/0/wfaa/local/%7Ehttps://tempgmail.email/
temp-mail-email-gmail https://getpocket.com/redirect?url=https://tempgmail.email/
temp mail https://pw.mail.ru/forums/fredirect.php?url=https://tempgmail.email/
temp-mail-email-gmail http://web.stanfor… http://search.bt.com/result?p=https://tempgmail.email/
temp mail http://feeds.osce.org/%7E/t/0/0/oscelatestnews/%7Ehttps://tempgmail.email/
temp gmail http://m.ok.ru/dk?st.cmd=outLinkWarning&st.rfn=https://tempgmail.email/
temp email
tempmail http://www.blog-directory.org/BlogList.aspx?q=https://tempgmail.email/
tempemail https://www.familiesonline.co.uk/search-result?indexCatalogue=search&searchQuery=https://tempgmail.email/
free temp mail https://yercum.com/?s=https://tempgmail.email/
temp-mail https://statvoo.com/search/ce-top10.com
temp-gmail http://www.rn-tp.com/search/node?keys=https%3A//www.ce-top10.com/
temp-mail-email-gmail https://mt-superman.com/bbs/search.php?srows=10&gr_id=&sfl=wr_subject%7C%7Cwr_content&stx=https://tempgmail.email/
temp mail https://usedheaven.com/bbs/search.php?sfl=wr_subject%7C%7Cwr_content&sop=and&stx=httpswww.ce-top10.com/
temp gmail http://forum.fruct.org/search/node/https%3A//www.ce-top10.com/
temp email https://www.economieetsociete.com/search/https%3A++www.ce-top10.com/
tempmail https://www.merricksart.com/?s=https://tempgmail.email/
tempemail https://www.aha-now.com/?s=https://tempgmail.email/
free temp mail https://www.logeion.nl/zoeken?q=https:%2F%2Fwww.ce-top10.com%2F
temp-mail https://www.malardalensdistansryttare.se/Lastasidor/Sok/?q=https://tempgmail.email/
temp-gmail https://www.vernis-halal.eu/en/search?controller=search&orderby=position&orderway=desc&search_query=https://tempgmail.email/
temp-mail-email-gmail https://www.basket.se/forbundet/Distrikt-BDF/Distrikten/stockholmsbasketbolldistriktsforbund/tavling/3×3/lastasidor/sok/?q=https://tempgmail.email/
temp mail https://freediving.cetmacomposites.it/it/ricerca?controller=search&orderby=position&orderway=desc&search_query=https://tempgmail.email/
temp gmail https://www.svenskbordtennis.com/forbundet/Distrikten/goteborgsbordtennisforbund/Spelarutveckling/samtraning/Lastasidor/Sok/?q=https://tempgmail.email/
temp email https://ce-top10.com.siteindices.com/
tempmail http://fedorasrv.com/link3/link3.cgi?mode=cnt&hp=https://tempgmail.email/
tempemail http://www.rss.geodles.com/fwd.php?url=https://tempgmail.email/
free temp mail http://imperialoptical.com/news-redirect.aspx?url=https://tempgmail.email/
temp-mail https://www.interecm.com/interecm/tracker?op=click&id=5204.db2&url=https://tempgmail.email/
temp-gmail http://kenchow.keensdesign.com/out.php?url=https://tempgmail.email/
temp-mail-email-gmail http://www.kompassdiamonds.com/WebKompassDiamonds/en/redirect?url=https://tempgmail.email/
temp mail http://landbidz.com/redirect.asp?url=https://tempgmail.email/
temp-mail-email-gmail http://www.leefleming.com/neurotwitch/index.php?URL=https://tempgmail.email/
temp mail http://sns.lilyenglish.com/link.php?url=https://tempgmail.email/
temp gmail http://lolataboo.com/out.php?https://tempgmail.email/
temp email http://www.mietenundkaufen.com/cgi-bin/linklist/links.pl?action=redirect&id=44828&URL=https://tempgmail.email/
tempmail http://sp.ojrz.com/out.html?id=tometuma&go=https://tempgmail.email/
tempemail http://forum.pokercollectif.com/redirect-to/?redirect=https://tempgmail.email/
free temp mail http://www.ra2d.com/directory/redirect.asp?id=596&url=https://tempgmail.email/
temp-mail https://relationshiphq.com/french.php?u=https://tempgmail.email/
temp-gmail http://www.reliablesoftware.com/DasBlog/ct.ashx?id=4ff24f38-3e75-4e01-9755-92e5a585806c&url=https://tempgmail.email/
temp-mail-email-gmail http://m.shopinelpaso.com/redirect.aspx?url=https://tempgmail.email/
temp mail http://m.shopinsanantonio.com/redirect.aspx?url=https://tempgmail.email/
temp gmail http://m.shopinsanjose.com/redirect.aspx?url=https://tempgmail.email/
temp email http://suek.com/bitrix/rk.php?goto=https://tempgmail.email/
tempmail https://throttlecrm.com/resources/webcomponents/link.php?realm=aftermarket&dealergroup=A5002T&link=https://tempgmail.email/
tempemail http://tstz.com/link.php?url=https://tempgmail.email/
free temp mail https://www.turnerdrake.com/blog/ct.ashx?id=3791bd86-2a35-4466-92ac-551acb587cea&url=https://tempgmail.email/
temp-mail http://hui.zuanshi.com/link.php?url=https://tempgmail.email/
temp-gmail https://www.weingut-villa.de/k2/System/Language/en/?goto=https://tempgmail.email/
temp-mail-email-gmail http://gamlihandil.fo/url.asp?url=https://tempgmail.email/
temp mail http://www.xata.co.il/redirect.php?url=https://tempgmail.email/
temp gmail http://www.resort-planning.co.jp/link/cutlinks/rank.php?url=https://tempgmail.email/
temp email http://gonzo.kz/banner/redirect?url=https://tempgmail.email/
tempmail http://redirect.me/?https://tempgmail.email/
tempemail http://light.anatoto.net/out.cgi?id=01178&url=https://tempgmail.email/
free temp mail http://tv.e-area.net/sm/out.cgi?id=10682&url=https://tempgmail.email/
temp-mail http://www.h-paradise.net/mkr1/out.cgi?id=01010&go=https://tempgmail.email/
temp-gmail http://www.highpoint.net/ASP/adredir.asp?url=https://tempgmail.email/
temp-mail-email-gmail http://www.mastertgp.net/tgp/click.php?id=353693&u=https://tempgmail.email/
temp mail http://dir.portokal-bg.net/counter.php?redirect=https://tempgmail.email/
temp-mail-email-gmail http://www.slfeed.net/jump.php?jump=https://tempgmail.email/
temp mail http://vlatkovic.net/ct.ashx?url=https://tempgmail.email/
temp gmail http://sm.zn7.net/out.cgi?id=00018&url=https://tempgmail.email/
temp email http://smyw.org/cgi-bin/atc/out.cgi?id=190&u=https://tempgmail.email/
tempmail https://www.izbudujemy.pl/redir.php?cid=78&unum=1&url=https://tempgmail.email/
tempemail http://catalog.grad-nk.ru/click/?id=130002197&id_town=0&www=https://tempgmail.email/
free temp mail http://www.paladiny.ru/go.php?url=https://tempgmail.email/
temp-mail https://www.tral.ru/images/get.php?go=https://tempgmail.email/
temp-gmail https://vrazvedka.ru/forum/go.php?https://tempgmail.email/
temp-mail-email-gmail http://dsxm.caa.se/links.do?c=138&t=3282&h=utskick.html&g=0&link=https://tempgmail.email/
temp mail http://www.artisansduchangement.tv/blog/wp-content/plugins/translator/translator.php?l=is&u=https://tempgmail.email/
temp gmail http://www.macro.ua/out.php?link=https://tempgmail.email/
temp email http://cms.nam.org.uk/Aggregator.ashx?url=https://tempgmail.email/
tempmail https://www.delano.mn.us/statistics/logClick.php?statType=banner&pageId=0&Id=56&redirect=https://tempgmail.email/
tempemail http://halongcity.gov.vn/c/document_library/find_file_entry?p_l_id=5202671&noSuchEntryRedirect=https://tempgmail.email/
free temp mail https://www.viecngay.vn/go?to=https://tempgmail.email/
temp-mail http://smile.wjp.am/link-free/link3.cgi?mode=cnt&no=8&hpurl=https://tempgmail.email/
temp-gmail http://www.kgdenoordzee.be/gbook/go.php?url=https://tempgmail.email/
temp-mail-email-gmail https://dakke.co/redirect/?url=https://tempgmail.email/
temp mail http://www.aurki.com/jarioa/redirect?id_feed=510&url=https://tempgmail.email/
temp gmail http://aussieearners.com/members/classifieds/go.php?url=https://tempgmail.email/
temp email http://www.bdsmandfetish.com/cgi-bin/sites/out.cgi?id=mandymon&url=https://tempgmail.email/
tempmail https://www.bettnet.com/blog/?URL=https://tempgmail.email/
tempemail https://bigboobsprivate.com/cgi/out.cgi?id=117&l=top_top&u=https://tempgmail.email/
free temp mail http://www.chungshingelectronic.com/redirect.asp?url=https://tempgmail.email/
temp-mail http://www.dansmovies.com/tp/out.php?link=tubeindex&p=95&url=https://tempgmail.email/
temp-gmail http://www.dddso.com/go.php?url=https://tempgmail.email/
temp-mail-email-gmail http://dirtyboundaries.com/cgi-bin/top/out.cgi?ses=GNA2RKxERH&id=251&url=https://tempgmail.email/
temp mail http://dommeteens.com/out.cgi?ses=kYgqhtVvzL&id=37&url=https://tempgmail.email/
temp-mail-email-gmail http://m.ee17.com/go.php?url=https://tempgmail.email/
temp mail http://www.ffatsearch.com/search/rank.cgi?mode=link&id=9636&url=https://tempgmail.email/
temp gmail http://floridafilmofficeinc.com/?goto=https://tempgmail.email/
temp email https://www.freemusic123.com/karaoke/cgi-bin/out.cgi?id=castillo&url=https://tempgmail.email/
tempmail http://www.glorioustronics.com/redirect.php?link=https://tempgmail.email/
tempemail http://hellothai.com/wwwlink/wwwredirect.asp?hp_id=1242&url=https://tempgmail.email/
free temp mail http://www.islulu.com/link.php?url=https://tempgmail.email/
temp-mail http://www.johnvorhees.com/gbook/go.php?url=https://tempgmail.email/
temp-gmail http://www.kanazawa-navi.com/navi/rank.cgi?mode=link&id=700&url=https://tempgmail.email/
temp-mail-email-gmail https://www.kushima.com/cgi-bin/kikusui/link.cgi?d=https://tempgmail.email/
temp mail http://lilnymph.com/out.php?https://tempgmail.email/
temp gmail http://www.lissakay.com/institches/index.php?URL=https://tempgmail.email/
temp email http://www.listenyuan.com/home/link.php?url=https://tempgmail.email/
tempmail https://www.mauihealthguide.com/ads/adclick.php?bannerid=25&zoneid=16&source=&dest=https://tempgmail.email/
tempemail http://www.mistress-and-slave.com/cgi-bin/out.cgi?id=123crush&url=https://tempgmail.email/
free temp mail http://www.momshere.com/cgi-bin/atx/out.cgi?id=212&tag=top12&trade=https://tempgmail.email/
temp-mail http://www.myhottiewife.com/cgi-bin/arpro/out.cgi?id=Jojo&url=https://tempgmail.email/
temp-gmail https://app.newsatme.com/emt/ses/814/33cfb749dac0cb4d05f2f1c78d3486607231be54/click?url=https://tempgmail.email/
temp-mail-email-gmail http://www.omatgp.com/cgi-bin/atc/out.cgi?id=17&u=https://tempgmail.email/
temp mail http://www.orth-haus.com/peters_empfehlungen/jump.php?site=https://tempgmail.email/
temp gmail http://www.sermemole.com/public/serbook/redirect.php?url=https://tempgmail.email/
temp email http://shop-navi.com/link.php?mode=link&id=192&url=https://tempgmail.email/
tempmail http://www.superiorfemdom.com/cgi-bin/sites/out.cgi?id=dominell&url=https://tempgmail.email/
tempemail https://flypoet.toptenticketing.com/index.php?url=https://tempgmail.email/
free temp mail http://whatsthecost.com/linktrack.aspx?url=https://tempgmail.email/
temp-mail http://models.world-collections.com/cgi-bin/df/out.cgi?ses=jVEuX3QqC0&id=831&url=https://tempgmail.email/
temp-gmail http://www.horesga.de/click/advertisement/195?url=https://tempgmail.email/
temp-mail-email-gmail http://kennel-makalali.de/gbook/go.php?url=https://tempgmail.email/
temp mail https://www.d-e-a.eu/newsletter/redirect.php?link=https://tempgmail.email/
temp-mail-email-gmail http://bulletformyvalentine.info/go.php?url=https://tempgmail.email/
temp mail http://ibmp.ir/link/redirect?url=https://tempgmail.email/
temp gmail http://www.cnainterpreta.it/redirect.asp?url=https://tempgmail.email/
temp email http://cgi.mediamix.ne.jp/~k2012/link3/link3.cgi?mode=cnt&no=3&hpurl=https://tempgmail.email/
tempmail http://www.eroticlinks.net/cgi-bin/atx/out.cgi?id=25&tag=topz&trade=https://tempgmail.email/
tempemail http://infoholix.net/redirect.php?mId=4263&mWeb=https://tempgmail.email/
free temp mail http://www.matatabix.net/out/click3.cgi?cnt=eroshocker&url=https://tempgmail.email/
temp-mail http://www.ratsoff.net/blog/nav?to=https://tempgmail.email/
temp-gmail http://www.site-navi.net/sponavi/rank.cgi?mode=link&id=890&url=https://tempgmail.email/
temp-mail-email-gmail http://vidoz.net/go/?url=https://tempgmail.email/
temp mail https://faector.nl/f/communication/email-redirect/99c56779?uri=https://tempgmail.email/
temp gmail https://timesofnepal.com.np/redirect?url=https://tempgmail.email/
temp email https://www.mattias.nu/cgi-bin/redirect.cgi?https://tempgmail.email/
tempmail http://people4success.co.nz/ra.asp?url=https://tempgmail.email/
tempemail http://www.frasergroup.org/peninsula/guestbook/go.php?url=https://tempgmail.email/
free temp mail https://www.swarganga.org/redirect.php?url=https://tempgmail.email/
temp-mail http://69dom.ru/redirect?url=https://tempgmail.email/
temp-gmail http://www.domodedovo-city.ru/plug.php?e=go&url=https://tempgmail.email/
temp-mail-email-gmail http://forum.ink-system.ru/go.php?https://tempgmail.email/
temp mail http://yubik.net.ru/go?https://tempgmail.email/
temp gmail http://staldver.ru/go.php?go=https://tempgmail.email/
temp email http://www.survivalbook.ru/forum/go.php?https://tempgmail.email/
tempmail https://unicom.ru/links.php?go=https://tempgmail.email/
tempemail https://bjorkstadensaikido.se/gbook15/go.php?url=https://tempgmail.email/
free temp mail http://tesay.com.tr/en?go=https://tempgmail.email/
temp-mail https://kombi-nation.co.uk/execs/trackit.php?user=guest_IuSyD&page=https://tempgmail.email/
temp-gmail http://teenstgp.us/cgi-bin/out.cgi?u=https://tempgmail.email/
temp-mail-email-gmail http://matongthiennhien.vn/url.aspx?id=https://tempgmail.email/
temp mail http://thucphamnhapkhau.vn/redirect?url=https://tempgmail.email/
temp-mail-email-gmail https://compromat.ws/goto?https://tempgmail.email/
temp mail http://beautynet.co.za/www/RotBannerStatic/redirect.asp?url=https://tempgmail.email/
temp gmail http://newellpalmer.com.au/?goto=https://tempgmail.email/
temp email http://www.enter-web.biz/Redirect.asp?AccID=-17745&AdCampaignID=2708&AdCampaignType=2&AffDuration=30&url=https://tempgmail.email/
tempmail http://www.zhenghe.biz/urlredirect.php?go=https://tempgmail.email/
tempemail http://hammel.ch/includes/asp/gettarget.asp?type=e&id=https://tempgmail.email/
free temp mail http://ab-search.com/rank.cgi?mode=link&id=107&url=https://tempgmail.email/
temp-mail http://alga-dom.com/scripts/banner.php?id=285&type=top&url=https://tempgmail.email/
temp-gmail http://www.campeggitalia.com/redirect/redirect.asp?sito=https://tempgmail.email/
temp-mail-email-gmail https://www.chinaleatheroid.com/redirect.php?url=https://tempgmail.email/
temp mail http://www.cloud-campaign.com/Redirect.aspx?companyid=15&scenarioid=4523&type=click&recordid=81a4b988-f110-4a83-8310-07af52db7ce8&&url=https://tempgmail.email/
temp gmail http://congovibes.com/index.php?thememode=full;redirect=https://tempgmail.email/
temp email http://www.cureya.com/kinbaku/out.cgi?id=13854&url=https://tempgmail.email/
tempmail http://www.dcgreeks.com/ad_redirect.asp?url=https://tempgmail.email/
tempemail http://www.eliseerotic.com/cgi-bin/autorank/out.cgi?id=pansuto&url=https://tempgmail.email/
free temp mail http://www.extreme-thumbz.com/cgi-bin/atc/out.cgi?id=33&u=https://tempgmail.email/
temp-mail http://www.findingfarm.com/redir?url=https://tempgmail.email/
temp-gmail http://freenudegranny.com/cgi-bin/atc/out.cgi?id=74&u=https://tempgmail.email/
temp-mail-email-gmail http://www.fuckk.com/cgi-bin/atx/out.cgi?id=163&tag=top2&trade=https://tempgmail.email/
temp mail http://www.hentaicrack.com/cgi-bin/atx/out.cgi?s=95&u=https://tempgmail.email/
temp gmail http://www.humaniplex.com/jscs.html?hj=y&ru=https://tempgmail.email/
temp email https://www.hyiphistory.com/visit.php?url=https://tempgmail.email/
tempmail http://www.imxyd.com/urlredirect.php?go=https://tempgmail.email/
tempemail http://iam.ittot.com/urlredirect.php?go=https://tempgmail.email/
free temp mail http://kite-rider.com/0link/rank.cgi?mode=link&id=178&url=https://tempgmail.email/
temp-mail http://ladyboysurprises.com/cgi-bin/at3/out.cgi?trade=https://tempgmail.email/
temp-gmail http://www.letras1.com/out.php?url=https://tempgmail.email/
temp-mail-email-gmail https://www.miratucarta.com/es/redir?url=https://tempgmail.email/
temp mail http://www.nafta-him.com/bitrix/redirect.php?event1=&event2=&event3=&goto=https://tempgmail.email/
temp-mail-email-gmail https://www.prairieoutdoors.com/lt.php?lt=https://tempgmail.email/
temp mail http://www.prismotri.com/url.php?url=https://tempgmail.email/
temp gmail http://prosticks.com/lang.asp?lang=en&url=https://tempgmail.email/
temp email http://lnk.pwwq.com/sm/out.cgi?id=00686&url=https://tempgmail.email/
tempmail http://pcr.richgroupusa.com/pcrbin/message.exe?action=REDIRECT&url=https://tempgmail.email/
tempemail http://www.scouttools.com/eu.aspx?url=https://tempgmail.email/
free temp mail https://www.sicakhaber.com/SicakHaberMonitoru/Redirect/?url=https://tempgmail.email/
temp-mail https://ullafyr.com/gjestebok/go.php?url=https://tempgmail.email/
temp-gmail http://vividvideoclips.com/cgi-bin/at3/out.cgi?s=80&c=3&u=https://tempgmail.email/
temp-mail-email-gmail http://www.qlt-online.de/cgi-bin/click/clicknlog.pl?link=https://tempgmail.email/
temp mail https://www.schwarzburg-tourismus.de/de/page/mod/url/url.php?eid=44&urlpf=https://tempgmail.email/
temp gmail http://slipknot1.info/go.php?url=https://tempgmail.email/
temp email https://www.donbosco.it/common/mod_30_conta.asp?id=6&link=https://tempgmail.email/
tempmail http://www.ndwa.com.jo/goto.php?url=https://tempgmail.email/
tempemail http://www.flowertour.jp/redirect.php?id=mi45f604ec34a55&url=https://tempgmail.email/
free temp mail https://magicode.me/affiliate/go?url=https://tempgmail.email/
temp-mail http://count.f-av.net/cgi/out.cgi?cd=fav&id=ranking_306&go=https://tempgmail.email/
temp-gmail https://tracker.onrecruit.net/api/v1/redirect/?redirect_to=https://tempgmail.email/
temp-mail-email-gmail http://ads.tcshopping.net/usb007/bin/redir.phtml?rid=GOO_AUTORUN&q=https://tempgmail.email/
temp mail http://race.warmd.net/engine.php?do=redirect&url=https://tempgmail.email/
temp gmail http://www.pta.gov.np/index.php/site/language/swaplang/1/?redirect=https://tempgmail.email/
temp email http://www.logopro.co.nz/ra.asp?url=https://tempgmail.email/
tempmail https://uniline.co.nz/Document/Url/?url=https://tempgmail.email/
tempemail http://warnerdisplays.co.nz/ra.asp?url=https://tempgmail.email/
free temp mail http://www.communityweb.org/ASP2/adredir.asp?url=https://tempgmail.email/
temp-mail http://www.morozo.org/exec/out.cgi?id=sioman&url=https://tempgmail.email/
temp-gmail http://old.roofnet.org/external.php?link=https://tempgmail.email/
temp-mail-email-gmail http://www.forum-wodociagi.pl/system/links/3a337d509d017c7ca398d1623dfedf85.html?link=https://tempgmail.email/
temp mail http://www.bucatareasa.ro/link.php?url=https://tempgmail.email/
temp-mail-email-gmail http://maximov-design.ru/link.php?go=https://tempgmail.email/
temp mail http://mosprogulka.ru/go?https://tempgmail.email/
temp gmail http://www.mukhin.ru/go.php?https://tempgmail.email/
temp email http://ranger66.ru/redir.php?url=https://tempgmail.email/
tempmail http://ww.sdam-snimu.ru/redirect.php?url=https://tempgmail.email/
tempemail http://t-s-c.org.tw/modules/links/redirect.php?url=https://tempgmail.email/
free temp mail https://fid.com.ua/redirect/?go=https://tempgmail.email/
temp-mail http://www.uktrademarkregistration.co.uk/JumpTo.aspx?url=https://tempgmail.email/
temp-gmail http://fs.co.za/redirect.aspx?id=55&url=https://tempgmail.email/
temp-mail-email-gmail http://www.amateurinterracial.biz/cgi-bin/atc/out.cgi?id=34&u=https://tempgmail.email/
temp mail http://e-okinet.biz/rank.php?mode=link&id=1599&url=https://tempgmail.email/
temp gmail http://kinnyuu.biz/rank/out.cgi?url=https://tempgmail.email/
temp email http://d-click.fmcovas.org.br/u/20636/11/16715/41_0/0c8eb/?url=https://tempgmail.email/
tempmail http://www.18to19.com/cgi-bin/atx/out.cgi?s=60&c=1&l=&u=https://tempgmail.email/
tempemail http://t.agrantsem.com/tt.aspx?cus=216&eid=1&p=216-2-71016b553a1fa2c9.3b14d1d7ea8d5f86&d=https://tempgmail.email/
free temp mail https://prod1.airage.com/cirrata/www/delivery/ck.php?ct=1&oaparams=2__bannerid=150__zoneid=50__cb=27f996991c__oadest=https://tempgmail.email/
temp-mail https://autoexplosion.com/link-exchange/results.php?url=https://tempgmail.email/
temp-gmail http://districtaustin.com/wp-content/themes/eatery/nav.php?-Menu-=https://tempgmail.email/
temp-mail-email-gmail http://dresscircle-net.com/psr/rank.cgi?mode=link&id=14&url=https://tempgmail.email/
temp mail http://bares.blog.idnes.cz/redir.aspx?url=https://tempgmail.email/
temp gmail http://es.chaturbate.com/external_link/?url=https://tempgmail.email/
temp email http://ipv4.google.com/url?q=https://tempgmail.email/
tempmail http://decisoes.fazenda.gov.br/netacgi/nph-brs?d=DECW&f=S&l=20&n=-DTPE&p=10&r=3&s1=COANA&u=https://tempgmail.email/
tempemail http://s5.histats.com/stats/r.php?869637&100&47794&urlr=&https://tempgmail.email/
free temp mail http://optimize.viglink.com/page/pmv?url=https://tempgmail.email/
temp-mail http://www.astro.wisc.edu/?URL=https://tempgmail.email/
temp-gmail http://blogs.rtve.es/libs/getfirma_footer_prod.php?blogurl=https://tempgmail.email/
temp-mail-email-gmail http://m.odnoklassniki.ru/dk?st.cmd=outLinkWarning&st.rfn=https://tempgmail.email/
temp mail http://sound2sense.archiveweb.mus.cam.ac.uk/?URL=https://tempgmail.email/
temp-mail-email-gmail http://search.bt.com/result?p=https://tempgmail.email/poi=&y=Web+Search&rd=r2
temp mail http://cc.naver.jp/cc?a=dtl.topic&r=&i=&bw=1024&px=0&py=0&sx=-1&sy=-1&m=1&nsc=knews.viewpage&u=https://tempgmail.email/
temp gmail http://channel.pixnet.net/newdirect.php?blog=cr19h3id&serial=0&url=https://tempgmail.email/
temp email http://analytics.tradedoubler.com/r.php?u=https://tempgmail.email/
tempmail http://community.acer.com/en/home/leaving/https://tempgmail.email/
tempemail http://dgi.mos.ru/bitrix/rk.php?goto=https://tempgmail.email/
free temp mail http://webfeeds.brookings.edu/~/t/0/0/~https://tempgmail.email/
temp-mail http://logp4.xiti.com/go.click?xts=130831&s2=1&p=armee-de-terre&clic=a&type=click&url=https://tempgmail.email/rdt=on
temp-gmail http://www.fhwa.dot.gov/reauthorization/reauexit.cfm?link=https://tempgmail.email/
temp-mail-email-gmail http://tvtropes.org/pmwiki/no_outbounds.php?o=https://tempgmail.email/
temp mail http://blog.ss-blog.jp/pages/mobile/step/index?u=https://tempgmail.email/
temp gmail http://my.apa.org/apa/idm/logout.seam?ERIGHTS_TARGET=https://tempgmail.email/
temp email http://notoprinting.xsrv.jp/feed2js/feed2js.php?src=https://tempgmail.email/
tempmail http://www.canada.com/scripts/go.aspx?g=vancouversun&n=subscribe_link_header&u=https://tempgmail.email/
tempemail http://en.gigazine.net/?URL=https://tempgmail.email/
free temp mail http://guru.sanook.com/?URL=https://tempgmail.email/
temp-mail http://rsc.org/rsc-id/account/generateauthorizationtoken?returnUrl=https://tempgmail.email/
temp-gmail http://ds.jpn.org/jump.php?url=https://tempgmail.email/
temp-mail-email-gmail http://accounts.cancer.org/login?redirectURL=https://tempgmail.email/theme=RFL
temp mail http://jump.2ch.net/?https://tempgmail.email/
temp gmail http://new.creativecommons.org/choose/results-one?q_1=2&q_1=1&field_commercial=n&field_derivatives=sa&field_jurisdiction=&field_format=Text&field_worktitle=Blog&field_attribute_to_name=TempMail&field_attribute_to_url=https://tempgmail.email/
temp email http://creativecommons.org/choose/results-one?q_1=2&q_1=1&field_commercial=n&field_derivatives=sa&field_jurisdiction=&field_format=Text&field_worktitle=Blog&field_attribute_to_name=TempMail&field_attribute_to_url=https://tempgmail.email/
tempmail http://cse.google.de/url?sa=t&url=https://tempgmail.email/
tempemail http://images.google.de/url?sa=t&url=https://tempgmail.email/
free temp mail http://maps.google.de/url?sa=t&url=https://tempgmail.email/
temp-mail http://clients1.google.de/url?sa=t&url=https://tempgmail.email/
temp-gmail http://t.me/iv?url=https://tempgmail.email/
temp-mail-email-gmail http://www.t.me/iv?url=https://tempgmail.email/
temp mail http://pw.mail.ru/forums/fredirect.php?url=https://tempgmail.email/
temp-mail-email-gmail http://legal.un.org/docs/doc_top.asp?path=../ilc/documentation/english/a_cn4_13.pd&Lang=Ef&referer=https://tempgmail.email/
temp mail http://www.bing.com/news/apiclick.aspx?ref=FexRss&aid=&tid=9BB77FDA801248A5AD23FDBDD5922800&url=https://tempgmail.email/
temp gmail http://wasearch.loc.gov/e2k/*/https://tempgmail.email/
temp email http://cr.naver.com/redirect-notification?u=https://tempgmail.email/
tempmail http://nutritiondata.self.com/facts/recipe/1304991/2?mbid=HDFD&trackback=https://tempgmail.email/
tempemail http://www.justjared.com/flagcomment.php?el=https://tempgmail.email/
free temp mail http://old.post-gazette.com/pets/redir.asp?url=https://tempgmail.email/
temp-mail http://www.etis.ford.com/externalURL.do?url=https://tempgmail.email/
temp-gmail http://daemon.indapass.hu/http/session_request?redirect_to=https://tempgmail.email/partner_id=bloghu
temp-mail-email-gmail http://fcaw.library.umass.edu/goto/https:/https://tempgmail.email/
temp mail http://www.etracker.com/lnkcnt.php?et=qPKGYV&url=https://tempgmail.email/
temp gmail http://contacts.google.com/url?sa=t&url=https://tempgmail.email/
temp email http://club.panasonic.jp/member/terms/?siteId=B1&returnURL=https://tempgmail.email/
tempmail http://sitereport.netcraft.com/?url=https://tempgmail.email/
tempemail http://id.telstra.com.au/register/crowdsupport?gotoURL=https://tempgmail.email/
free temp mail http://gleam.io/TempMail?l=https://tempgmail.email/
temp-mail http://www.kaskus.co.id/redirect?url=https://tempgmail.email/
temp-gmail http://branch.app.link/?$deeplink_path=article/jan/123&$fallback_url=https://tempgmail.email/
temp-mail-email-gmail http://translate.itsc.cuhk.edu.hk/gb/https://tempgmail.email/
temp mail http://domain.opendns.com/https://tempgmail.email/
temp gmail http://bbs.pku.edu.cn/v2/jump-to.php?url=https://tempgmail.email/
temp email http://www.vz.ru/redir/?source=vz_teasers_main2&id=917154&exturl=https://tempgmail.email/
tempmail http://eventlog.centrum.cz/redir?data=aclick1c68565-349178t12&s=najistong&v=1&url=https://tempgmail.email/
tempemail http://community.esri.com/external-link.jspa?url=https://tempgmail.email/
free temp mail http://profiles.newsmax.com/sso/signup.aspx?ReturnURL=https://tempgmail.email/
temp-mail http://learninginmotion.uvic.ca/emailStat.htm?action=redirect&email=187&emailAddress=anthtwo@uvic.ca&url=https://tempgmail.email/
temp-gmail http://www.curseforge.com/linkout?remoteUrl=https://tempgmail.email/
temp-mail-email-gmail http://forums.thesims.com/en_uS/home/leaving/https://tempgmail.email/
temp mail http://www2.ogs.state.ny.us/help/urlstatusgo.html?url=https://tempgmail.email/
temp-mail-email-gmail http://scanmail.trustwave.com/?c=8510&d=4qa02KqxZJadHuhFUvy7ZCUfI_2L10yeH0EeBz7FGQ&u=https://tempgmail.email/
temp mail http://tools.folha.com.br/print?url=https://tempgmail.email/site=blogfolha
temp gmail http://slides.virtualpathology.leeds.ac.uk/research_4/slide_library/r_bishop_collection/card_index_set/soft_tissue/33959.svs/view.apml?returnurl=https://tempgmail.email/
temp email http://www.etracker.de/lnkcnt.php?et=aSVPbb&url=https://tempgmail.email/
tempmail http://mix.com/search/https://tempgmail.email/domain
tempemail http://talgov.com/Main/exit.aspx?url=https://tempgmail.email/
free temp mail http://www.bad.org.uk/for-the-public/patient-information-leaflets/androgenetic-alopecia/?showmore=1&returnlink=https://tempgmail.email/
temp-mail http://kupiauto.zr.ru/bitrix/rk.php?goto=https://tempgmail.email/
temp-gmail http://amp.shinystat.com/cgi-bin/redir.cgi?URL=https://tempgmail.email/
temp-mail-email-gmail http://sc.hkexnews.hk/TuniS/https://tempgmail.email/
temp mail http://feeds.hanselman.com/~/t/0/0/scotthanselman/~https://tempgmail.email/
temp gmail http://trackings.oxfamintermon.org/click.php?campanya=SYD1612_S0310_03126_0001&data_enviament=20161207&desti=https://tempgmail.email/
temp email http://weheartit.com/from/https://tempgmail.email/
tempmail http://www.spiritfanfiction.com/link?l=https://tempgmail.email/
tempemail http://www.kichink.com/home/issafari?uri=https://tempgmail.email/
free temp mail http://www.dotdata.ct.gov/media/mainpsa.aspx?url=https://tempgmail.email/
temp-mail http://isci.med.miami.edu/?URL=https://tempgmail.email/
temp-gmail http://www.london.umb.edu/?URL=https://tempgmail.email/
temp-mail-email-gmail http://do.expertreviews.co.uk/shop-rest/api/track/long/product/ex/awYaFF9tLidA6PqQ9KsJn1xXgp_luIIW1C2zzpy5QbmacF1pR6puZza5IriLVTEV?target=https://tempgmail.email/
temp mail http://es.catholic.net/ligas/ligasframe.phtml?liga=https://tempgmail.email/
temp gmail http://edu.glogster.com/library/proxy?url=https://tempgmail.email/
temp email http://www.worldlingo.com/S4698.0/translation?wl_url=https://tempgmail.email/
tempmail http://forum.solidworks.com/external-link.jspa?url=https://tempgmail.email/
tempemail http://advisor.wmtransfer.com/SiteDetails.aspx?url=https://tempgmail.email/
free temp mail http://www.winnipegfreepress.com/s?action=doLogout&rurl=https://tempgmail.email/
temp-mail http://ssomgmt.ascd.org/profile/createsso/createsso.aspx?returnURL=https://tempgmail.email/
temp-gmail http://ipx.bcove.me/?url=https://tempgmail.email/
temp-mail-email-gmail http://www.interempresas.net/estadisticas/r.asp?idsector=129&e=221083&c=195&d=https://tempgmail.email/
temp mail http://sherlock.scribblelive.com/r?u=https://tempgmail.email/
temp-mail-email-gmail http://qatar.vcu.edu/?URL=https://tempgmail.email/
temp mail http://www.sinp.msu.ru/ru/ext_link?url=https://tempgmail.email/
temp gmail http://esiiab.uclm.es/phpqrcode/creaqr.php?url=https://tempgmail.email/
temp email http://fishbiz.seagrant.uaf.edu/click-thru.html?url=https://tempgmail.email/
tempmail http://blog.sina.com.tw/url_verify.php?url=https://tempgmail.email/
tempemail http://www.unifrance.org/newsletter-click/6763261?url=https://tempgmail.email/
free temp mail http://xtpanel.xtgem.com/templates/logo.php?site=https://tempgmail.email/
temp-mail http://www.med.lublin.pl/S_baner/adclick.php?bannerid=16&zoneid=0&source=&dest=https://tempgmail.email/
temp-gmail http://ceskapozice.lidovky.cz/redir.aspx?url=https://tempgmail.email/
temp-mail-email-gmail http://mitsui-shopping-park.com/lalaport/ebina/redirect.html?url=https://tempgmail.email/
temp mail http://community.nxp.com/t5/custom/page/page-id/ExternalRedirect?url=https://tempgmail.email/
temp gmail http://innuityweb.myregisteredsite.com/admin/membership_agreement.php?partnerID=3185&domain=https://tempgmail.email/
temp email http://register.scotland.gov.uk/subscribe/widgetsignup?url=https://tempgmail.email/
tempmail http://openforbusinessmagazine01.businesscatalyst.com/Redirect.aspx?destination=https://tempgmail.email/
tempemail http://slack-redir.net/link?url=https://tempgmail.email/
free temp mail http://www.bonanza.com/home/redirect_warning?url=https://tempgmail.email/
temp-mail http://foro.infojardin.com/proxy.php?link=https://tempgmail.email/
temp-gmail http://board-en.drakensang.com/proxy.php?link=https://tempgmail.email/
temp-mail-email-gmail http://bugcrowd.com/external_redirect?site=https://tempgmail.email/
temp mail http://www.sitejabber.com/reviews/https://tempgmail.email/
temp gmail http://rosreestr.gov.ru/bitrix/redirect.php?goto=https://tempgmail.email/
temp email http://hostedmovieupdates.aebn.net/feed/?urlstub=https://tempgmail.email/
tempmail http://rcs-acs-prod-us.sandbox.google.com/url?sa=i&url=https://tempgmail.email/
tempemail http://account.vfw.org/Authenticate.aspx?ReturnURL=https://tempgmail.email/
free temp mail http://na.eventscloud.com/emarketing/go.php?i=409508&e=&l=https://tempgmail.email/
temp-mail http://search.aol.com/aol/search?query=https://tempgmail.email/
temp-gmail http://search.yahoo.com/search?p=https://tempgmail.email/fr=yfp-t&ei=UTF-8&fp=1
temp-mail-email-gmail http://search.lycos.com/web/?q=https://tempgmail.email/
temp mail http://jour425.media.illinois.edu/cgi-bin/425.cgi?PG=&GO=https://tempgmail.email/
temp-mail-email-gmail http://redirects.tradedoubler.com/utm/td_redirect.php?td_keep_old_utm_value=1&url=https://tempgmail.email/
temp mail http://ugc.kn3.net/s/https:/https://tempgmail.email/
temp gmail http://yandex.com/search/?text=https://tempgmail.email/lr=104830&redircnt=1617465358.1
temp email http://duckduckgo.com/?q=Gazeteler+Gazete+oku&t=h_&ia=web
tempmail http://www.ask.com/web?o=0&l=dir&qo=homepageSearchBox&q=https://tempgmail.email/
tempemail http://www.semrush.com/analytics/overview/?q=https://tempgmail.email/searchType=domain
free temp mail https://www.qwant.com/?q=https://tempgmail.email/t=web
temp-mail http://www.ecosia.org/search?q=sitehttps://tempgmail.email/
temp-gmail http://ref.gamer.com.tw/redir.php?url=https://tempgmail.email/
temp-mail-email-gmail http://affiliates.bookdepository.com/scripts/click.php?a_aid=Alexa1202&a_bid=9abb5269&desturl=https://tempgmail.email/
temp mail http://results.excite.com/serp?q=https://tempgmail.email/
temp gmail http://rd.tiscali.it/?u=https://tempgmail.email/
temp email http://www.zive.cz/textredirect.aspx?qu=https://tempgmail.email/
tempmail http://community.rsa.com/external-link.jspa?url=https://tempgmail.email/
tempemail http://f5.glitch.me/proxy/https://tempgmail.email/
free temp mail http://community.keysight.com/external-link.jspa?url=https://tempgmail.email/
temp-mail http://ville.montreal.qc.ca/portal/page?pageid=5798,42657625&dad=portal&schema=PORTAL&id=12879&ret=https://tempgmail.email/
temp-gmail http://drive.sandbox.google.com/url?sa=i&url=https://tempgmail.email/
temp-mail-email-gmail http://api.whatsapp.com/send?text=https://tempgmail.email/
temp mail http://trello.com/add-card?source=mode=popup&name=click+here&desc=https://tempgmail.email/
temp gmail http://www.baidu.com/s?ie=utf-8&f=8&rsv_bp=1&rsv_idx=1&tn=baidu&wd=https://tempgmail.email/
temp email http://justpaste.it/redirect/172fy/https://tempgmail.email/
tempmail http://www.au.kddi.com/wau/manual/site_out.php?model=W54S&url=https://tempgmail.email/
tempemail http://search.seznam.cz/?count=10&q=https://tempgmail.email/gazete-mansetleri-oku&offset=10&co=0&where=world
free temp mail http://www.justjaredjr.com/flagcomment.php?cl=10842755&el=https://tempgmail.email/
temp-mail http://mywords.cle.ust.hk/resources/dictionary_loading.php?link=https://tempgmail.email/
temp-gmail http://media.blubrry.com/jepcast/https://tempgmail.email/?d=RRO2T2U7
temp-mail-email-gmail http://mypage.syosetu.com/?jumplink=https://tempgmail.email/
temp mail http://shop.myfico.com/order/checkout.php?LANG=en&SHOPURL=https://tempgmail.email/ORDERS
temp-mail-email-gmail http://jobzone.ny.gov/views/jobzone/leaving_site.jsf?id=304&url=https://tempgmail.email/
temp mail http://xat.com/web_gear/chat/linkvalidator.php?link=https://tempgmail.email/
temp gmail http://feeds.gizmodo.com.au/~/t/0/0/gizmodoaustralia/~/https://tempgmail.email/
temp email http://thediplomat.com/ads/books/ad.php?i=4&r=https://tempgmail.email/
tempmail http://feeds.kotaku.com.au/~/t/0/0/kotakuaustralia/~/https://tempgmail.email/
tempemail http://feeds.lifehacker.com.au/~/t/0/0/lifehackeraustralia/~/https://tempgmail.email/
free temp mail http://businesscatalyst.com/Redirect.aspx?destination=https://tempgmail.email/
temp-mail http://mitsui-shopping-park.com/lalaport/iwata/redirect.html?url=https://tempgmail.email/
temp-gmail http://feeds.osce.org/~/t/0/0/osceofficetajikistan/~/https://tempgmail.email/
temp-mail-email-gmail http://publishing.brookes.ac.uk/?URL=https://tempgmail.email/
temp mail http://inquiry.princetonreview.com/away/?value=cconntwit&category=FS&url=https://tempgmail.email/
temp gmail http://law.spbu.ru/aboutfaculty/teachers/teacherdetails/a7fb1dbb-e9f3-4fe9-91e9-d77a53b8312c.aspx?returnurl=https://tempgmail.email/
temp email http://marketplace.salisburypost.com/AdHunter/salisburypost/Home/EmailFriend?url=https://tempgmail.email/
tempmail http://nou-rau.uem.br/nou-rau/zeus/auth.php?back=https://tempgmail.email/go=x&code=x&unit=x
tempemail http://janeyleegrace.worldsecuresystems.com/redirect.aspx?destination=https://tempgmail.email/
free temp mail http://webmail.unige.it/horde/util/go.php?url=https://tempgmail.email/
temp-mail http://sc.hkex.com.hk/TuniS/https://tempgmail.email/
temp-gmail http://ams.ceu.edu/optimal/optimal.php?url=https://tempgmail.email/
temp-mail-email-gmail http://www.migrantcinema.net.gridhosted.co.uk/?URL=https://tempgmail.email/
temp mail http://www.wittstock.chemie.uni-oldenburg.de/agef/link_extern.html?link=https://tempgmail.email/
temp gmail http://www.aaronsw.com/2002/display.cgi?t=%3Ca+href%3Dhttps://tempgmail.email/
temp email http://www.earth-policy.org/?URL=https://tempgmail.email/
tempmail http://inginformatica.uniroma2.it/?URL=https://tempgmail.email/
tempemail http://wfc2.wiredforchange.com/dia/track.jsp?v=2&c=hdorrh%2BHcDlQ%2BzUEnZU5qlfKZ1Cl53X6&url=https://tempgmail.email/
free temp mail http://www.cheapassgamer.com/redirect.php?url=https://tempgmail.email/
temp-mail http://www.faseb.org/pdfviewer?loadthis=https://tempgmail.email/
temp-gmail http://redirect.camfrog.com/redirect/?url=https://tempgmail.email/
temp-mail-email-gmail http://www.pfizer.es/redirect.aspx?uri=https://tempgmail.email/
temp mail http://feeds.ligonier.org/~/t/0/0/ligonierministriesblog/~/https://tempgmail.email/
temp-mail-email-gmail http://may2012.archive.ensembl.org/Help/Permalink?url=https://tempgmail.email/
temp mail http://sfwater.org/redirect.aspx?url=https://tempgmail.email/
temp gmail http://feeds.gty.org/~/t/0/0/gtyblog/~/https://tempgmail.email/
temp email http://biology.africamuseum.be/BiocaseProvider_2.4.2/www/utilities/queryforms/qf_manual.cgi?url=https://tempgmail.email/
tempmail http://feeds.marinsoftware.com/~/t/0/0/marininsights/~/https://tempgmail.email/
tempemail http://cse.google.com.gh/url?sa=i&url=https://tempgmail.email/
free temp mail https://search.aol.com/aol/search?query=https://tempgmail.email/
temp-mail https://search.yahoo.com/search?p=https://tempgmail.email/fr=yfp-t&ei=UTF-8&fp=1
temp-gmail http://search.lycos.com/b.php?u=xm7E-gNCFy194ImDkNX1awriwiKxGDz7RlLsGbJtsI.::dqiBl6jH-vcAFTG4cEiAw..::BFSH&s=unknown&p=1&rd=search.lycos.com&as=https://tempgmail.email/
temp-mail-email-gmail https://search14.lycos.com/web/?q=https://tempgmail.email/keyvol=00de98f5fb04c6c760fa
temp mail http://plus.google.com/url?q=https://tempgmail.email/
temp gmail http://www.youtube.com/redirect?q=https://tempgmail.email/
temp email http://web.archive.org/web/20180804180141/https://tempgmail.email/
tempmail http://www.ric.edu/Pages/linkout.aspx?target=https://tempgmail.email/
tempemail http://tyonabi.sakura.ne.jp/link/cgi-bin/out.cgi?id=dorian362&cg=1&siteurl=https://tempgmail.email/
free temp mail http://galter.northwestern.edu/exit?url=https://tempgmail.email/
temp-mail https://rspcb.safety.fhwa.dot.gov/pageRedirect.aspx?RedirectedURL=https://tempgmail.email/
temp-gmail https://sitereport.netcraft.com/?url=https://tempgmail.email/
temp-mail-email-gmail http://community.nfpa.org/external-link.jspa?url=https://tempgmail.email/
temp mail https://transtats.bts.gov/exit.asp?url=https://tempgmail.email/
temp gmail http://www.google.com/url?sa=t&url=https://tempgmail.email/
temp email http://maps.google.com/url?q=https://tempgmail.email/
tempmail https://board-en.drakensang.com/proxy.php?link=https://tempgmail.email/
tempemail http://yp.ocregister.com/_media__/js/netsoltrademark.php?d=https://tempgmail.email/
free temp mail http://yellowpages.staradvertiser.com/media/js/netsoltrademark.php?d=https://tempgmail.email/
temp-mail http://local.lex18.com/media/js/netsoltrademark.php?d=https://tempgmail.email/
temp-gmail http://yellowpages.sfexaminer.com/media/js/netsoltrademark.php?d=https://tempgmail.email/
temp-mail-email-gmail http://localbusiness.starnewsonline.com/media/js/netsoltrademark.php?d=https://tempgmail.email/
temp mail http://www.youtube.com/redirect?event=channel_description&q=https://tempgmail.email/
temp-mail-email-gmail http://toolbarqueries.google.no/url?sa=t&url=https://tempgmail.email/
temp mail http://toolbarqueries.google.pl/url?sa=t&url=https://tempgmail.email/
temp gmail http://toolbarqueries.google.ro/url?sa=t&url=https://tempgmail.email/
temp email http://toolbarqueries.google.rs/url?sa=t&url=https://tempgmail.email/
tempmail http://toolbarqueries.google.se/url?sa=t&url=https://tempgmail.email/
tempemail http://toolbarqueries.google.si/url?sa=t&url=https://tempgmail.email/
free temp mail http://toolbarqueries.google.sk/url?sa=t&url=https://tempgmail.email/
temp-mail http://tools.folha.com.br/print?url=https://tempgmail.email/F&site=blogfolha
temp-gmail https://trello.com/add-card?source=mode=popup&name=click+here&desc=http%3A%2F%2Ftempgmail.email
temp-mail-email-gmail http://wasearch.loc.gov/e2k/*/tempgmail.email
temp mail https://webmail.unige.it/horde/util/go.php?url=https://tempgmail.email/F
temp gmail http://www.aaronsw.com/2002/display.cgi?t=%3Ca+href%3Dhttps://tempgmail.email/F
temp email https://www.bing.com/news/apiclick.aspx?ref=FexRss&aid=&tid=9BB77FDA801248A5AD23FDBDD5922800&url=https://tempgmail.email/
tempmail https://www.bonanza.com/home/redirect_warning?url=tempgmail.email/
tempemail http://www.canada.com/scripts/go.aspx?g=vancouversun&n=subscribe_link_header&u=https://tempgmail.email/F
free temp mail https://www.cheapassgamer.com/redirect.php?url=https://tempgmail.email/F
temp-mail http://www.earth-policy.org/?URL=https://tempgmail.email/F
temp-gmail https://www.etis.ford.com/externalURL.do?url=https://tempgmail.email/F
temp mail https://sitereport.netcraft.com/?url=https://tempgmail.email/F
temp gmail http://scanmail.trustwave.com/?c=8510&d=4qa02KqxZJadHuhFUvy7ZCUfI_2L10yeH0EeBz7FGQ&u=https://tempgmail.email/F
temp email http://sc.hkexnews.hk/TuniS/tempgmail.email
tempmail https://redirect.camfrog.com/redirect/?url=https://tempgmail.email/F
tempemail http://rcs-acs-prod-us.sandbox.google.com/url?sa=i&url=https://tempgmail.email/F
free temp mail https://www.oxfordpublish.org/?URL=https://tempgmail.email/F
temp-mail https://profiles.newsmax.com/sso/signup.aspx?ReturnURL=https://tempgmail.email/F
temp-gmail http://nou-rau.uem.br/nou-rau/zeus/auth.php?back=https://tempgmail.email/F&go=x&code=x&unit=x
temp-mail-email-gmail https://mypage.syosetu.com/?jumplink=http%3A%2F%2Ftempgmail.email
temp mail https://mix.com/search/tempgmail.email/domain
temp-mail-email-gmail https://mitsui-shopping-park.com/lalaport/ebina/redirect.html?url=https://tempgmail.email/
temp mail https://www.georgewbushlibrary.gov/exit.aspx?url=https://tempgmail.email/F
temp gmail https://forums.thesims.com/en_uS/home/leaving/tempgmail.email
tempmail http://client.paltalk.com/client/webapp/client/External.wmt?url=http%3A%2F%2Ftempgmail.email
tempemail https://ceskapozice.lidovky.cz/redir.aspx?URL=https://tempgmail.email/F
blairebble@gmail.com
SPORTSTOTO7.COM
카지노사이트 https://sportstoto7.com/
바카라사이트 https://sportstoto7.com/
온라인카지노 https://sportstoto7.com/
온라인바카라 https://sportstoto7.com/
온라인슬롯사이트 https://sportstoto7.com/
카지노사이트게임 https://sportstoto7.com/
카지노사이트검증 https://sportstoto7.com/
카지노사이트추천 https://sportstoto7.com/
안전카지노사이트 https://sportstoto7.com/
안전카지노사이트도메인 https://sportstoto7.com/
안전한 카지노사이트 추천 https://sportstoto7.com/
바카라사이트게임 https://sportstoto7.com/
바카라사이트검증 https://sportstoto7.com/
바카라사이트추천 https://sportstoto7.com/
안전바카라사이트 https://sportstoto7.com/
안전바카라사이트도메인 https://sportstoto7.com/
안전한 바카라사이트 추천 https://sportstoto7.com/
http://www.erotikplatz.at/redirect.php?id=939&mode=fuhrer&url=https://sportstoto7.com/
http://www.imsnet.at/LangChange.aspx?uri=https://sportstoto7.com/
https://www.kath-kirche-kaernten.at/pfarren/pfarre/C3014?URL=https://sportstoto7.com/
http://gs.matzendorf.at/includes/linkaufruf.asp?art=kapitel&link=https://sportstoto7.com/
http://www.nuttenzone.at/jump.php?url=https://sportstoto7.com/
https://cms.oeav-events.at/wGlobal/nessyEDVapps/layout/fancybox.php?link=https://sportstoto7.com/
https://www.oebb.at/nightjet_newsletter/tc/xxxx?url=https://sportstoto7.com/
https://www.gardensonline.com.au/Global/Players/YouTube.aspx?VideoURL=https://sportstoto7.com/
http://www2.golflink.com.au/out.aspx?frm=gglcmicrosite&target=https://sportstoto7.com/
http://www2.golflink.com.au/out.aspx?frm=logo&target=https://sportstoto7.com/
https://www.golfselect.com.au/redirect?activityType_cd=WEB-LINK&course_id=2568&tgturl=https://sportstoto7.com/
https://www.malcolmturnbull.com.au/?URL=https://sportstoto7.com/
http://march-hare.com.au/library/default.asp?PP=/library/toc/lib-12.xml&tocPath=&URL=https://https://sportstoto7.com/
https://www.oliverhume.com.au/enquiry/thank-you/?redirectTo=https://sportstoto7.com/
http://www.parents-guide-illawarra.com.au/Redirect.aspx?destination=https://https://sportstoto7.com/
https://ramset.com.au/Document/Url/?url=https://sportstoto7.com/
https://ramset.com.au/document/url/?url=https://sportstoto7.com/
http://rubyconnection.com.au/umbraco/newsletterstudio/tracking/trackclick.aspx?url=https://sportstoto7.com/
http://southburnett.com.au/movies/movie.php?url=https://sportstoto7.com/
https://www.vicsport.com.au/analytics/outbound?url=https://sportstoto7.com/
https://www.vwwatercooled.com.au/forums/redirect-to/?redirect=https://https://sportstoto7.com/
http://clients3.weblink.com.au/clients/aluminalimited/priceframe1.aspx?link=https://sportstoto7.com/
http://fedorasrv.com/link3/link3.cgi?mode=cnt&hp=https://sportstoto7.com/
http://www.rss.geodles.com/fwd.php?url=https://sportstoto7.com/
http://imperialoptical.com/news-redirect.aspx?url=https://sportstoto7.com/
https://www.interecm.com/interecm/tracker?op=click&id=5204.db2&url=https://sportstoto7.com/
http://kenchow.keensdesign.com/out.php?url=https://sportstoto7.com/
http://www.kompassdiamonds.com/WebKompassDiamonds/en/redirect?url=https://sportstoto7.com/
http://landbidz.com/redirect.asp?url=https://sportstoto7.com/
http://www.leefleming.com/neurotwitch/index.php?URL=https://sportstoto7.com/
http://sns.lilyenglish.com/link.php?url=https://sportstoto7.com/
http://lolataboo.com/out.php?https://sportstoto7.com/
http://www.mietenundkaufen.com/cgi-bin/linklist/links.pl?action=redirect&id=44828&URL=https://sportstoto7.com/
http://sp.ojrz.com/out.html?id=tometuma&go=https://sportstoto7.com/
http://forum.pokercollectif.com/redirect-to/?redirect=https://sportstoto7.com/
http://www.ra2d.com/directory/redirect.asp?id=596&url=https://sportstoto7.com/
https://relationshiphq.com/french.php?u=https://sportstoto7.com/
http://www.reliablesoftware.com/DasBlog/ct.ashx?id=4ff24f38-3e75-4e01-9755-92e5a585806c&url=https://sportstoto7.com/
http://m.shopinelpaso.com/redirect.aspx?url=https://sportstoto7.com/
http://m.shopinsanantonio.com/redirect.aspx?url=https://sportstoto7.com/
http://m.shopinsanjose.com/redirect.aspx?url=https://sportstoto7.com/
http://suek.com/bitrix/rk.php?goto=https://sportstoto7.com/
https://throttlecrm.com/resources/webcomponents/link.php?realm=aftermarket&dealergroup=A5002T&link=https://sportstoto7.com/
http://tstz.com/link.php?url=https://sportstoto7.com/
https://www.turnerdrake.com/blog/ct.ashx?id=3791bd86-2a35-4466-92ac-551acb587cea&url=https://sportstoto7.com/
http://hui.zuanshi.com/link.php?url=https://sportstoto7.com/
https://www.weingut-villa.de/k2/System/Language/en/?goto=https://sportstoto7.com/
http://gamlihandil.fo/url.asp?url=https://sportstoto7.com/
http://www.xata.co.il/redirect.php?url=https://sportstoto7.com/
http://www.resort-planning.co.jp/link/cutlinks/rank.php?url=https://sportstoto7.com/
http://gonzo.kz/banner/redirect?url=https://sportstoto7.com/
http://redirect.me/?https://sportstoto7.com/
http://light.anatoto.net/out.cgi?id=01178&url=https://sportstoto7.com/
http://tv.e-area.net/sm/out.cgi?id=10682&url=https://sportstoto7.com/
http://www.h-paradise.net/mkr1/out.cgi?id=01010&go=https://sportstoto7.com/
http://www.highpoint.net/ASP/adredir.asp?url=https://sportstoto7.com/
http://www.mastertgp.net/tgp/click.php?id=353693&u=https://sportstoto7.com/
http://dir.portokal-bg.net/counter.php?redirect=https://sportstoto7.com/
http://www.slfeed.net/jump.php?jump=https://sportstoto7.com/
http://vlatkovic.net/ct.ashx?url=https://sportstoto7.com/
http://sm.zn7.net/out.cgi?id=00018&url=https://sportstoto7.com/
http://smyw.org/cgi-bin/atc/out.cgi?id=190&u=https://sportstoto7.com/
https://www.izbudujemy.pl/redir.php?cid=78&unum=1&url=https://sportstoto7.com/
http://catalog.grad-nk.ru/click/?id=130002197&id_town=0&www=https://sportstoto7.com/
http://www.paladiny.ru/go.php?url=https://sportstoto7.com/
https://www.tral.ru/images/get.php?go=https://sportstoto7.com/
https://vrazvedka.ru/forum/go.php?https://sportstoto7.com/
http://dsxm.caa.se/links.do?c=138&t=3282&h=utskick.html&g=0&link=https://sportstoto7.com/
http://www.artisansduchangement.tv/blog/wp-content/plugins/translator/translator.php?l=is&u=https://sportstoto7.com/
http://www.macro.ua/out.php?link=https://sportstoto7.com/
http://cms.nam.org.uk/Aggregator.ashx?url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
https://www.delano.mn.us/statistics/logClick.php?statType=banner&pageId=0&Id=56&redirect=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://halongcity.gov.vn/c/document_library/find_file_entry?p_l_id=5202671&noSuchEntryRedirect=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
https://www.viecngay.vn/go?to=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://smile.wjp.am/link-free/link3.cgi?mode=cnt&no=8&hpurl=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.kgdenoordzee.be/gbook/go.php?url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
https://dakke.co/redirect/?url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.aurki.com/jarioa/redirect?id_feed=510&url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://aussieearners.com/members/classifieds/go.php?url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.bdsmandfetish.com/cgi-bin/sites/out.cgi?id=mandymon&url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
https://www.bettnet.com/blog/?URL=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
https://bigboobsprivate.com/cgi/out.cgi?id=117&l=top_top&u=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.chungshingelectronic.com/redirect.asp?url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.dansmovies.com/tp/out.php?link=tubeindex&p=95&url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.dddso.com/go.php?url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://dirtyboundaries.com/cgi-bin/top/out.cgi?ses=GNA2RKxERH&id=251&url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://dommeteens.com/out.cgi?ses=kYgqhtVvzL&id=37&url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://m.ee17.com/go.php?url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.ffatsearch.com/search/rank.cgi?mode=link&id=9636&url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://floridafilmofficeinc.com/?goto=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
https://www.freemusic123.com/karaoke/cgi-bin/out.cgi?id=castillo&url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.glorioustronics.com/redirect.php?link=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://hellothai.com/wwwlink/wwwredirect.asp?hp_id=1242&url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.islulu.com/link.php?url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.johnvorhees.com/gbook/go.php?url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.kanazawa-navi.com/navi/rank.cgi?mode=link&id=700&url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
https://www.kushima.com/cgi-bin/kikusui/link.cgi?d=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://lilnymph.com/out.php?https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.lissakay.com/institches/index.php?URL=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.listenyuan.com/home/link.php?url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
https://www.mauihealthguide.com/ads/adclick.php?bannerid=25&zoneid=16&source=&dest=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.mistress-and-slave.com/cgi-bin/out.cgi?id=123crush&url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.momshere.com/cgi-bin/atx/out.cgi?id=212&tag=top12&trade=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.myhottiewife.com/cgi-bin/arpro/out.cgi?id=Jojo&url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
https://app.newsatme.com/emt/ses/814/33cfb749dac0cb4d05f2f1c78d3486607231be54/click?url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.omatgp.com/cgi-bin/atc/out.cgi?id=17&u=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.orth-haus.com/peters_empfehlungen/jump.php?site=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.sermemole.com/public/serbook/redirect.php?url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://shop-navi.com/link.php?mode=link&id=192&url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.superiorfemdom.com/cgi-bin/sites/out.cgi?id=dominell&url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
https://flypoet.toptenticketing.com/index.php?url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://whatsthecost.com/linktrack.aspx?url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://models.world-collections.com/cgi-bin/df/out.cgi?ses=jVEuX3QqC0&id=831&url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.horesga.de/click/advertisement/195?url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://kennel-makalali.de/gbook/go.php?url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
https://www.d-e-a.eu/newsletter/redirect.php?link=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://bulletformyvalentine.info/go.php?url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://ibmp.ir/link/redirect?url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.cnainterpreta.it/redirect.asp?url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://cgi.mediamix.ne.jp/~k2012/link3/link3.cgi?mode=cnt&no=3&hpurl=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.eroticlinks.net/cgi-bin/atx/out.cgi?id=25&tag=topz&trade=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://infoholix.net/redirect.php?mId=4263&mWeb=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.matatabix.net/out/click3.cgi?cnt=eroshocker&url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.ratsoff.net/blog/nav?to=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.site-navi.net/sponavi/rank.cgi?mode=link&id=890&url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://vidoz.net/go/?url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
https://faector.nl/f/communication/email-redirect/99c56779?uri=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
https://timesofnepal.com.np/redirect?url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
https://www.mattias.nu/cgi-bin/redirect.cgi?https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://people4success.co.nz/ra.asp?url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.frasergroup.org/peninsula/guestbook/go.php?url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
https://www.swarganga.org/redirect.php?url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://69dom.ru/redirect?url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.domodedovo-city.ru/plug.php?e=go&url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://forum.ink-system.ru/go.php?https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://yubik.net.ru/go?https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://staldver.ru/go.php?go=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.survivalbook.ru/forum/go.php?https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
https://unicom.ru/links.php?go=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
https://bjorkstadensaikido.se/gbook15/go.php?url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://tesay.com.tr/en?go=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
https://kombi-nation.co.uk/execs/trackit.php?user=guest_IuSyD&page=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://teenstgp.us/cgi-bin/out.cgi?u=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://matongthiennhien.vn/url.aspx?id=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://thucphamnhapkhau.vn/redirect?url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
https://compromat.ws/goto?https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://beautynet.co.za/www/RotBannerStatic/redirect.asp?url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://newellpalmer.com.au/?goto=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.enter-web.biz/Redirect.asp?AccID=-17745&AdCampaignID=2708&AdCampaignType=2&AffDuration=30&url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.zhenghe.biz/urlredirect.php?go=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://hammel.ch/includes/asp/gettarget.asp?type=e&id=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://ab-search.com/rank.cgi?mode=link&id=107&url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://alga-dom.com/scripts/banner.php?id=285&type=top&url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.campeggitalia.com/redirect/redirect.asp?sito=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
https://www.chinaleatheroid.com/redirect.php?url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.cloud-campaign.com/Redirect.aspx?companyid=15&scenarioid=4523&type=click&recordid=81a4b988-f110-4a83-8310-07af52db7ce8&&url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://congovibes.com/index.php?thememode=full;redirect=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.cureya.com/kinbaku/out.cgi?id=13854&url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.dcgreeks.com/ad_redirect.asp?url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.eliseerotic.com/cgi-bin/autorank/out.cgi?id=pansuto&url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.extreme-thumbz.com/cgi-bin/atc/out.cgi?id=33&u=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.findingfarm.com/redir?url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://freenudegranny.com/cgi-bin/atc/out.cgi?id=74&u=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.fuckk.com/cgi-bin/atx/out.cgi?id=163&tag=top2&trade=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.hentaicrack.com/cgi-bin/atx/out.cgi?s=95&u=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.humaniplex.com/jscs.html?hj=y&ru=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
https://www.hyiphistory.com/visit.php?url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.imxyd.com/urlredirect.php?go=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://iam.ittot.com/urlredirect.php?go=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://kite-rider.com/0link/rank.cgi?mode=link&id=178&url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://ladyboysurprises.com/cgi-bin/at3/out.cgi?trade=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.letras1.com/out.php?url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
https://www.miratucarta.com/es/redir?url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.nafta-him.com/bitrix/redirect.php?event1=&event2=&event3=&goto=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
https://www.prairieoutdoors.com/lt.php?lt=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.prismotri.com/url.php?url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://prosticks.com/lang.asp?lang=en&url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://lnk.pwwq.com/sm/out.cgi?id=00686&url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://pcr.richgroupusa.com/pcrbin/message.exe?action=REDIRECT&url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.scouttools.com/eu.aspx?url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
https://www.sicakhaber.com/SicakHaberMonitoru/Redirect/?url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
https://ullafyr.com/gjestebok/go.php?url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://vividvideoclips.com/cgi-bin/at3/out.cgi?s=80&c=3&u=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.qlt-online.de/cgi-bin/click/clicknlog.pl?link=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
https://www.schwarzburg-tourismus.de/de/page/mod/url/url.php?eid=44&urlpf=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://slipknot1.info/go.php?url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
https://www.donbosco.it/common/mod_30_conta.asp?id=6&link=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.ndwa.com.jo/goto.php?url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.flowertour.jp/redirect.php?id=mi45f604ec34a55&url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
https://magicode.me/affiliate/go?url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://count.f-av.net/cgi/out.cgi?cd=fav&id=ranking_306&go=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
https://tracker.onrecruit.net/api/v1/redirect/?redirect_to=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://ads.tcshopping.net/usb007/bin/redir.phtml?rid=GOO_AUTORUN&q=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://race.warmd.net/engine.php?do=redirect&url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.pta.gov.np/index.php/site/language/swaplang/1/?redirect=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.logopro.co.nz/ra.asp?url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
https://uniline.co.nz/Document/Url/?url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://warnerdisplays.co.nz/ra.asp?url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.communityweb.org/ASP2/adredir.asp?url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.morozo.org/exec/out.cgi?id=sioman&url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://old.roofnet.org/external.php?link=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.forum-wodociagi.pl/system/links/3a337d509d017c7ca398d1623dfedf85.html?link=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.bucatareasa.ro/link.php?url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://maximov-design.ru/link.php?go=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://mosprogulka.ru/go?https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.mukhin.ru/go.php?https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://ranger66.ru/redir.php?url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://ww.sdam-snimu.ru/redirect.php?url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://t-s-c.org.tw/modules/links/redirect.php?url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
https://fid.com.ua/redirect/?go=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.uktrademarkregistration.co.uk/JumpTo.aspx?url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://fs.co.za/redirect.aspx?id=55&url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.amateurinterracial.biz/cgi-bin/atc/out.cgi?id=34&u=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://e-okinet.biz/rank.php?mode=link&id=1599&url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://kinnyuu.biz/rank/out.cgi?url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://d-click.fmcovas.org.br/u/20636/11/16715/41_0/0c8eb/?url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://www.18to19.com/cgi-bin/atx/out.cgi?s=60&c=1&l=&u=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://t.agrantsem.com/tt.aspx?cus=216&eid=1&p=216-2-71016b553a1fa2c9.3b14d1d7ea8d5f86&d=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
https://prod1.airage.com/cirrata/www/delivery/ck.php?ct=1&oaparams=2__bannerid=150__zoneid=50__cb=27f996991c__oadest=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
https://autoexplosion.com/link-exchange/results.php?url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://districtaustin.com/wp-content/themes/eatery/nav.php?-Menu-=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
http://dresscircle-net.com/psr/rank.cgi?mode=link&id=14&url=https://sportsbetting010.blogspot.com/2022/08/dwcs-week-3-2022-odds-and-predictions.html
https://images.google.com.my/url?sa=t&url=https://sportstoto7.com/
https://images.google.com.mx/url?sa=t&url=https://sportstoto7.com/
https://images.google.com.mt/url?sa=t&url=https://sportstoto7.com/
https://images.google.com.ly/url?sa=t&url=https://sportstoto7.com/
https://images.google.com.lb/url?sa=t&url=https://sportstoto7.com/
https://images.google.com.kw/url?sa=t&url=https://sportstoto7.com/
https://images.google.com.kh/url?sa=t&url=https://sportstoto7.com/
https://images.google.com.jm/url?sa=t&url=https://sportstoto7.com/
https://images.google.com.hk/url?sa=t&url=https://sportstoto7.com/
https://images.google.com.gt/url?sa=t&url=https://sportstoto7.com/
https://images.google.com.gi/url?sa=t&url=https://sportstoto7.com/
https://images.google.com.gh/url?sa=t&url=https://sportstoto7.com/
https://images.google.com.fj/url?sa=t&url=https://sportstoto7.com/
https://images.google.com.et/url?sa=t&url=https://sportstoto7.com/
https://images.google.com.eg/url?sa=t&url=https://sportstoto7.com/
https://images.google.com.ec/url?sa=t&url=https://sportstoto7.com/
https://images.google.com.do/url?sa=t&url=https://sportstoto7.com/
https://images.google.com.cy/url?sa=t&url=https://sportstoto7.com/
https://images.google.com.cu/url?sa=t&url=https://sportstoto7.com/
https://images.google.com.co/url?sa=t&url=https://sportstoto7.com/
https://images.google.com.bz/url?sa=t&url=https://sportstoto7.com/
https://images.google.com.br/url?sa=t&url=https://sportstoto7.com/
https://images.google.com.bo/url?sa=t&url=https://sportstoto7.com/
https://images.google.com.bn/url?sa=t&url=https://sportstoto7.com/
https://images.google.com.bh/url?sa=t&url=https://sportstoto7.com/
https://images.google.com.bd/url?sa=t&url=https://sportstoto7.com/
https://images.google.com.au/url?sa=t&url=https://sportstoto7.com/
https://images.google.com.ar/url?sa=t&url=https://sportstoto7.com/
https://images.google.com.ag/url?sa=t&url=https://sportstoto7.com/
https://images.google.com.af/url?sa=t&url=https://sportstoto7.com/
https://images.google.co.zm/url?sa=t&url=https://sportstoto7.com/
https://images.google.co.za/url?sa=t&url=https://sportstoto7.com/
https://images.google.co.za/url?q=https://sportstoto7.com/
https://images.google.co.ve/url?sa=t&url=https://sportstoto7.com/
https://images.google.co.uz/url?sa=t&url=https://sportstoto7.com/
https://images.google.co.uk/url?sa=t&url=https://sportstoto7.com/
https://images.google.co.ug/url?sa=t&url=https://sportstoto7.com/
https://images.google.co.tz/url?sa=t&url=https://sportstoto7.com/
https://images.google.co.th/url?sa=t&url=https://sportstoto7.com/
https://images.google.co.th/url?q=https://sportstoto7.com/
https://images.google.co.nz/url?sa=t&url=https://sportstoto7.com/
https://images.google.co.ma/url?sa=t&url=https://sportstoto7.com/
https://images.google.co.ls/url?sa=t&url=https://sportstoto7.com/
https://images.google.co.kr/url?sa=t&url=https://sportstoto7.com/
https://images.google.co.kr/url?q=https://sportstoto7.com/
https://images.google.co.ke/url?sa=t&url=https://sportstoto7.com/
https://images.google.co.jp/url?sa=t&url=https://sportstoto7.com/
https://images.google.co.in/url?sa=t&url=https://sportstoto7.com/
https://images.google.co.il/url?sa=t&url=https://sportstoto7.com/
https://images.google.co.id/url?sa=t&url=https://sportstoto7.com/
https://images.google.co.id/url?q=https://sportstoto7.com/
https://images.google.co.cr/url?sa=t&url=https://sportstoto7.com/
https://images.google.co.cr/url?q=https://sportstoto7.com/
https://images.google.co.ck/url?sa=t&url=https://sportstoto7.com/
https://images.google.co.bw/url?sa=t&url=https://sportstoto7.com/
https://images.google.cm/url?sa=t&url=https://sportstoto7.com/
https://images.google.cl/url?sa=t&url=https://sportstoto7.com/
https://images.google.ci/url?sa=t&url=https://sportstoto7.com/
https://images.google.ch/url?sa=t&url=https://sportstoto7.com/
https://images.google.cg/url?sa=t&url=https://sportstoto7.com/
https://images.google.cd/url?sa=t&url=https://sportstoto7.com/
https://images.google.cat/url?sa=t&url=https://sportstoto7.com/
https://images.google.ca/url?sa=t&url=https://sportstoto7.com/
https://images.google.by/url?sa=t&url=https://sportstoto7.com/
https://images.google.bs/url?sa=t&url=https://sportstoto7.com/
https://images.google.bi/url?sa=t&url=https://sportstoto7.com/
https://images.google.bg/url?sa=t&url=https://sportstoto7.com/
https://images.google.bf/url?sa=t&url=https://sportstoto7.com/
https://images.google.be/url?sa=t&url=https://sportstoto7.com/
https://images.google.ba/url?sa=t&url=https://sportstoto7.com/
https://images.google.ba/url?q=https://sportstoto7.com/
https://images.google.az/url?sa=t&url=https://sportstoto7.com/
https://images.google.at/url?sa=t&url=https://sportstoto7.com/
https://images.google.as/url?sa=t&url=https://sportstoto7.com/
https://images.google.am/url?sa=t&url=https://sportstoto7.com/
https://images.google.al/url?sa=t&url=https://sportstoto7.com/
https://images.google.ae/url?sa=t&url=https://sportstoto7.com/
https://images.google.ae/url?q=https://sportstoto7.com/
lstiles135@gmail.com.
https://opviewer.com/
http://www.erotikplatz.at/redirect.php?id=939&mode=fuhrer&url=https://opviewer.com
http://www.imsnet.at/LangChange.aspx?uri=https://opviewer.com
https://www.kath-kirche-kaernten.at/pfarren/pfarre/C3014?URL=https://opviewer.com
http://gs.matzendorf.at/includes/linkaufruf.asp?art=kapitel&link=https://opviewer.com
http://www.nuttenzone.at/jump.php?url=https://opviewer.com
https://cms.oeav-events.at/wGlobal/nessyEDVapps/layout/fancybox.php?link=https://opviewer.com
https://www.oebb.at/nightjet_newsletter/tc/xxxx?url=https://opviewer.com
https://www.gardensonline.com.au/Global/Players/YouTube.aspx?VideoURL=https://opviewer.com
http://www2.golflink.com.au/out.aspx?frm=gglcmicrosite&target=https://opviewer.com
http://www2.golflink.com.au/out.aspx?frm=logo&target=https://opviewer.com
https://www.golfselect.com.au/redirect?activityType_cd=WEB-LINK&course_id=2568&tgturl=https://opviewer.com
https://www.malcolmturnbull.com.au/?URL=https://opviewer.com
http://march-hare.com.au/library/default.asp?PP=/library/toc/lib-12.xml&tocPath=&URL=https://https://opviewer.com
https://www.oliverhume.com.au/enquiry/thank-you/?redirectTo=https://opviewer.com
http://www.parents-guide-illawarra.com.au/Redirect.aspx?destination=https://https://opviewer.com
https://ramset.com.au/Document/Url/?url=https://opviewer.com
https://ramset.com.au/document/url/?url=https://opviewer.com
http://rubyconnection.com.au/umbraco/newsletterstudio/tracking/trackclick.aspx?url=https://opviewer.com
http://southburnett.com.au/movies/movie.php?url=https://opviewer.com
https://www.vicsport.com.au/analytics/outbound?url=https://opviewer.com
https://www.vwwatercooled.com.au/forums/redirect-to/?redirect=https://https://opviewer.com
http://clients3.weblink.com.au/clients/aluminalimited/priceframe1.aspx?link=https://opviewer.com
https://maps.google.lt/url?sa=t&url=https://opviewer.com
https://ref.gamer.com.tw/redir.php?url=https://opviewer.com
https://images.google.com.sa/url?sa=t&url=https://opviewer.com
https://maps.google.com.sa/url?sa=t&url=https://opviewer.com
https://www.google.com.sa/url?sa=t&url=https://opviewer.com
https://images.google.hr/url?sa=t&url=https://opviewer.com
https://www.google.hr/url?sa=t&url=https://opviewer.com
https://maps.google.hr/url?sa=t&url=https://opviewer.com
https://images.google.com.pe/url?sa=t&url=https://opviewer.com
https://www.google.com.pe/url?sa=t&url=https://opviewer.com
https://maps.google.ae/url?sa=t&url=https://opviewer.com
https://images.google.ae/url?sa=t&url=https://opviewer.com
https://www.google.ae/url?sa=t&url=https://opviewer.com
https://www.google.co.ve/url?sa=t&url=https://opviewer.com
https://maps.google.co.ve/url?sa=t&url=https://opviewer.com
https://images.google.co.ve/url?sa=t&url=https://opviewer.com
http://onlinemanuals.txdot.gov/help/urlstatusgo.html?url=https://opviewer.com
https://www.google.com.pk/url?sa=t&url=https://opviewer.com
https://images.google.com.pk/url?sa=t&url=https://opviewer.com
https://community.rsa.com/t5/custom/page/page-id/ExternalRedirect?url=https://opviewer.com
https://www.google.com.eg/url?sa=t&url=https://opviewer.com
https://maps.google.com.eg/url?sa=t&url=https://opviewer.com
https://images.google.com.eg/url?sa=t&url=https://opviewer.com
https://www.google.si/url?sa=t&url=https://opviewer.com
https://maps.google.si/url?sa=t&url=https://opviewer.com
https://images.google.si/url?sa=t&url=https://opviewer.com
http://www.pickyourown.org/articles.php?NAME=Visit+Us&URL=https://opviewer.com
https://maps.google.lv/url?sa=t&url=https://opviewer.com
https://www.google.lv/url?sa=t&url=https://opviewer.com
https://images.google.lv/url?sa=t&url=https://opviewer.com
https://community.cypress.com/t5/custom/page/page-id/ExternalRedirect?url=https://opviewer.com
https://www.google.ee/url?sa=t&url=https://opviewer.com
https://cms.oeav-events.at/wGlobal/nessyEDVapps/layout/fancybox.php?link=https://opviewer.com
https://www.oebb.at/nightjet_newsletter/tc/xxxx?url=https://opviewer.com
https://www.gardensonline.com.au/Global/Players/YouTube.aspx?VideoURL=https://opviewer.com
http://www2.golflink.com.au/out.aspx?frm=gglcmicrosite&target=https://opviewer.com
http://www2.golflink.com.au/out.aspx?frm=logo&target=https://opviewer.com
https://www.golfselect.com.au/redirect?activityType_cd=WEB-LINK&courseid=2568&tgturl=https://opviewer.com
https://www.malcolmturnbull.com.au/?URL=https://opviewer.com
http://march-hare.com.au/library/default.asp?PP=/library/toc/lib-12.xml&tocPath=&URL=https://https://opviewer.com
https://www.oliverhume.com.au/enquiry/thank-you/?redirectTo=https://opviewer.com
http://www.parents-guide-illawarra.com.au/Redirect.aspx?destination=https://https://opviewer.com
https://ramset.com.au/Document/Url/?url=https://opviewer.com
https://ramset.com.au/document/url/?url=https://opviewer.com
http://rubyconnection.com.au/umbraco/newsletterstudio/tracking/trackclick.aspx?url=https://opviewer.com
http://southburnett.com.au/movies/movie.php?url=https://opviewer.com
https://www.vicsport.com.au/analytics/outbound?url=https://opviewer.com
https://www.vwwatercooled.com.au/forums/redirect-to/?redirect=https://https://opviewer.com
http://clients3.weblink.com.au/clients/aluminalimited/priceframe1.aspx?link=https://opviewer.com
https://clients1.google.ad/url?q=https://opviewer.com
https://cse.google.ad/url?q=https://opviewer.com
https://images.google.ad/url?q=https://opviewer.com
https://maps.google.ad/url?q=https://opviewer.com
https://www.google.ad/url?q=https://opviewer.com
https://emaratyah.ae/new-redirect.php?w=https://opviewer.com
http://mbrf.ae/knowledgeaward/language/ar/?redirecturl=https://opviewer.com
http://rafco.ae/container.asp?url=https://opviewer.com
http://for-css.ucoz.ae/go?https://opviewer.com
https://clients1.google.com.af/url?q=https://opviewer.com
https://cse.google.com.af/url?q=https://opviewer.com
https://images.google.com.af/url?q=https://opviewer.com
http://toolbarqueries.google.com.af/url?sa=t&url=https://opviewer.com
https://www.google.com.af/url?q=https://opviewer.com
https://www.snek.ai/redirect?url=https://opviewer.com
http://www.torrent.ai/lt/redirect.php?url=https://opviewer.com
http://avto.al/az/home/redirect?carId=1639612&url=https://opviewer.com
https://clients1.google.al/url?q=https://opviewer.com
https://cse.google.al/url?q=https://opviewer.com
https://images.google.al/url?q=https://opviewer.com
https://images.google.al/url?q=https://opviewer.com
http://toolbarqueries.google.al/url?q=https://opviewer.com
https://www.google.al/url?q=https://opviewer.com
http://tido.al/vazhdo.php?url=https://opviewer.com
http://smile.wjp.am/link-free/link3.cgi?mode=cnt&no=8&hpurl=https://opviewer.com
https://oxleys.app/friends.php?q=https://opviewer.com
http://www.ain.com.ar/openpop.php?url=https://opviewer.com
http://www.ain.com.ar/openpop.php?url=https://opviewer.com
https://www.google.nl/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.mw/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.mv/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.mu/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.ms/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.mn/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.mk/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.mg/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.me/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.md/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.lv/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.lu/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.lt/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.lk/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.li/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.la/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.kz/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.kg/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.jo/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.je/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.it/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.is/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.iq/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.ie/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.hu/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.ht/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.hr/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.hn/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.gr/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.gp/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.gm/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.gl/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.gg/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.ge/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.fr/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.fm/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.fi/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.es/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.ee/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.dz/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.dk/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.dj/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.de/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.cz/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.com/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.com.vn/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.com.uy/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.com.ua/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.com.tw/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.com.tr/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.com.sv/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.com.sg/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.com.sa/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.com.qa/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.com.py/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.com.pr/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.com.pk/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.com.ph/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.com.pe/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.com.pa/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.com.om/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.com.ni/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.com.ng/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.com.na/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.com.mz/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.com.my/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.com.mx/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.com.mt/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.com.ly/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.com.lb/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.com.kw/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.com.kh/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.com.jm/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.com.hk/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.com.gt/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.com.gi/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.com.gh/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.com.fj/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.com.et/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.com.eg/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.com.ec/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.com.do/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.com.cy/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.com.cu/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.com.co/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.com.bz/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.com.br/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.com.bo/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.com.bn/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.com.bh/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.com.bd/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.com.au/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.com.ar/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.com.ag/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.com.af/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.co.za/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.co.ve/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.co.uk/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.co.ug/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.co.tz/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.co.th/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.co.nz/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.co.ma/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.co.ls/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.co.kr/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.co.ke/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.co.jp/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.co.in/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.co.il/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.co.id/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.co.cr/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.co.bw/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.cm/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.cl/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.ci/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.ch/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.cd/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.cat/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.ca/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.by/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.bs/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.bi/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.bg/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.bf/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.be/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.ba/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.az/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.at/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.as/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.am/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.al/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.ae/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://www.google.ad/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://plus.google.com/url?q=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.tn/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.sn/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.sk/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.si/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.sh/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.se/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.rw/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.ru/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.rs/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.ro/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.pt/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.pl/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.no/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.nl/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.mw/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.mv/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.mu/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.ms/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.mn/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.mk/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.mg/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.lv/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.lu/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.lt/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.lk/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.li/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.la/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.kz/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.kg/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.jo/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.je/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.it/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.is/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.iq/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.ie/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.hu/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.ht/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.hr/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.hn/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.gr/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.gm/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.gl/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.gg/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.ge/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.fr/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.fm/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.fi/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.es/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.ee/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.dz/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.dk/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.dj/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.de/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.cz/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.com/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.com.uy/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.com.ua/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.com.tw/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.com.tr/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.com.sv/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.com.sg/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.com.sa/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.com.qa/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.com.py/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.com.pr/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.com.ph/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.com.pe/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.com.pa/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.com.om/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.com.ni/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.com.ng/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.com.na/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.com.mz/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.com.my/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.com.mx/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.com.mt/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.com.ly/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.com.lb/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.com.kw/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.com.kh/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.com.jm/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.com.hk/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.com.gt/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.com.gi/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.com.gh/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.com.fj/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.com.et/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.com.eg/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.com.ec/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.com.do/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.com.cu/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.com.co/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.com.bz/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.com.br/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.com.bo/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.com.bn/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.com.bh/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.com.bd/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.com.au/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.com.ar/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.com.ag/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.co.za/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.co.ve/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.co.uk/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.co.ug/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.co.tz/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.co.th/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.co.nz/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.co.ls/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.co.kr/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.co.ke/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.co.jp/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.co.in/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.co.il/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.co.id/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.co.cr/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.co.bw/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.cm/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.cl/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.ci/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.ch/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.cd/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.cat/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.ca/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.by/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.bs/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.bi/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.bg/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.bf/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.be/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.ba/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.at/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.as/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.ae/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://maps.google.ad/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.tn/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.sn/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.sk/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.si/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.sh/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.se/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.rw/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.ru/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.rs/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.ro/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.pt/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.ps/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.pl/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.no/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.nl/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.mw/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.mv/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.mu/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.ms/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.mn/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.mk/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.mg/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.me/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.md/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.lv/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.lu/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.lt/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.lk/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.li/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.la/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.kz/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.kg/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.jo/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.je/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.it/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.is/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.iq/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.ie/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.hu/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.ht/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.hr/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.hn/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.gr/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.gp/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.gm/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.gl/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.gg/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.ge/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.fr/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.fm/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.fi/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.es/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.ee/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.dz/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.dm/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.dk/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.dj/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.de/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.cz/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.com/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.com.vn/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.com.vc/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.com.uy/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.com.ua/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.com.tw/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.com.tr/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.com.sv/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.com.sg/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.com.sa/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.com.qa/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.com.py/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.com.pr/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.com.pk/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.com.ph/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.com.pe/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.com.pa/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.com.om/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.com.np/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.com.ni/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.com.ng/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.com.na/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.com.mz/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.com.my/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.com.mx/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.com.mt/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.com.ly/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.com.lb/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.com.kw/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.com.kh/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.com.jm/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.com.hk/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.com.gt/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.com.gi/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.com.gh/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.com.fj/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.com.et/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.com.eg/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.com.ec/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.com.do/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.com.cy/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.com.cu/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.com.co/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.com.bz/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.com.br/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.com.bo/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.com.bn/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.com.bh/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.com.bd/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.com.au/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.com.ar/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.com.ag/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.com.af/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.co.zm/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.co.za/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.co.za/url?q=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.co.ve/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.co.uz/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.co.uk/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.co.ug/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.co.tz/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.co.th/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.co.th/url?q=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.co.nz/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.co.ma/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.co.ls/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.co.kr/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.co.kr/url?q=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.co.ke/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.co.jp/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.co.in/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.co.il/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.co.id/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.co.id/url?q=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.co.cr/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.co.cr/url?q=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.co.ck/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.co.bw/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.cm/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.cl/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.ci/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.ch/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.cg/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.cd/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.cat/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.ca/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.by/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.bs/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.bi/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.bg/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.bf/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.be/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.ba/url?sa=t&url=https://opviewer.com/for-what-reason-do-people-need-a-massage/
https://images.google.ba/url?q=https://opviewer.com/for-what-reason-do-people-need-a-massage/