Benzoic Acid: Skin Antiseptic and Food Preservative
Benzoic Acid is a very common ingredient to a chemist. It is known to be the colorless solid which is often times used as a preservative. Benzoic Acid has a chemical formula of C7H6O2, which is composed of a simple aromatic carboxylic acid. It naturally occurs in many plants and can be used as a food preservative. It can also be used as an antiseptic on the skin to help prevent any infection caused by bacteria.
What does Benzoic Acid look like in Chemistry?
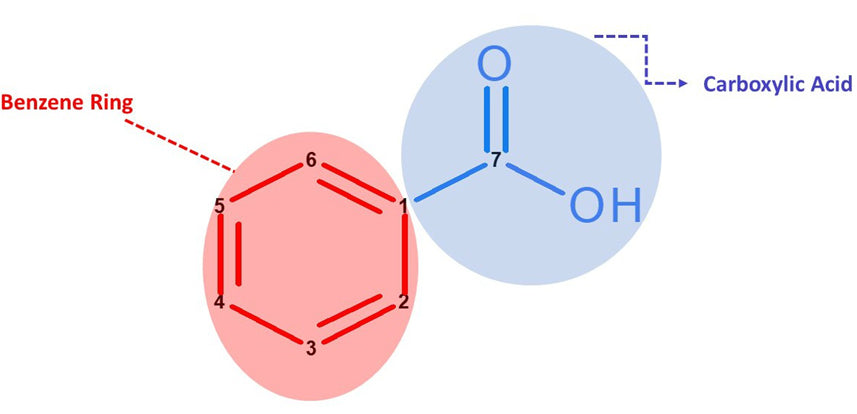
Let’s Get Building!
Using your Student Molecular Set from Duluth Labs let’s create Benzoic Acid! You’ll need:
-
7 Carbon Atoms
-
2 Oxygen atoms
-
6 Hydrogen atoms
-
6 Small connectors (compact small bonds for hydrogen)
-
5 Medium Connectors
-
8 Long connectors
-
Molecular Tool (for Disassembly)
Put aside all the atoms and connectors needed.
Let’s Start Building With Our Benzene (Ring A).
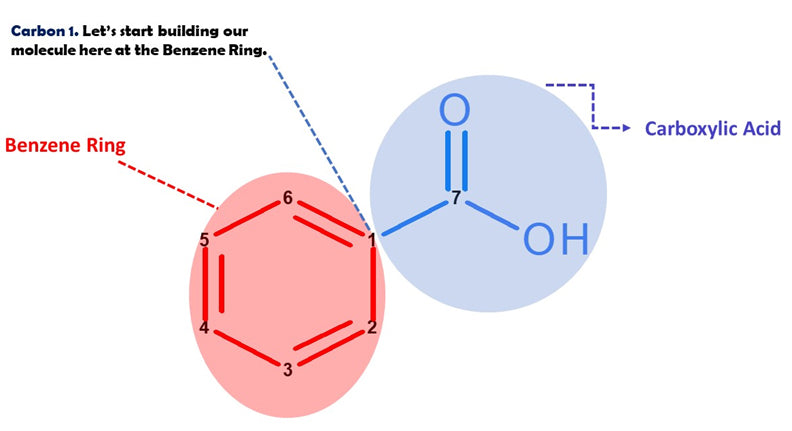
Note: We build this portion in a clockwise direction, starting with Carbon 1.
Let’s start!
Steps:
-
1

1. Get a Carbon atom (Carbon 1) then attach another carbon(Carbon 2)to it using a medium connector. Add a hydrogen atom to Carbon 2 using a small connector.
-
2

2.Attach another carbon atom (Carbon 3) to Carbon 2 using 2 long connectors. Place a hydrogen atom to this Carbon 3 atom using a small connector.
-
3

3.Get another carbon atom (Carbon 4)then attach this to Carbon 3 using a medium connector. Add a Hydrogen atom to Carbon 4.
-
4

4.Then, attach another carbon atom (Carbon 5) to Carbon 4 using 2 long connectors. Add a Hydrogen atom to Carbon 5.
-
5

5.Attach one carbon atom (Carbon 6) to Carbon 5 using 1 medium connector, then using a small connector, attach a hydrogen atom to Carbon 6.
-
6

6.Using 2 long connectors, join both Carbon 6 and Carbon 1 together.
-

Yay! We've just built our Benzene Ring Portion!
Note: Let’s now build the Carboxylic Acid portion of our Benzoic Acid molecule. We will build this portion in a clockwise direction, starting with Carbon 7.
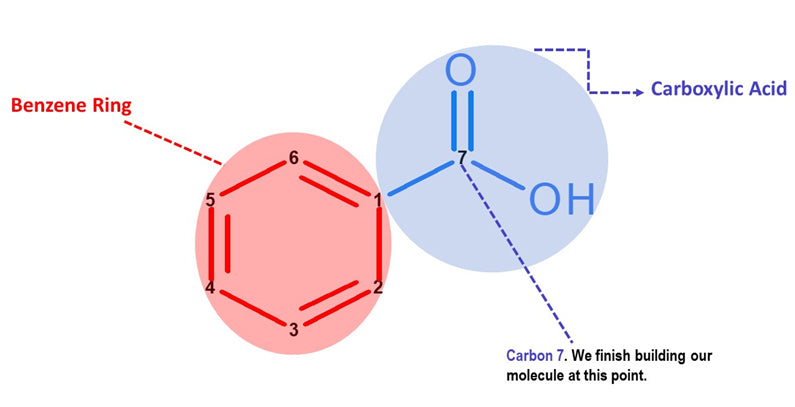
Let’s continue building!
Steps:
-
1

1.Get one carbon atom (Carbon 7) then attach this to Carbon 1(of Ring A) using a medium connector.
-
2

2.Then, place one oxygen atom on Carbon 7. using 2long connectors.
-
3

3.Get another oxygen atom and attach this to Carbon7using a medium connector. Then, add one hydrogen atom on the oxygen atom, forming a hydroxyl (-OH). Group.
-
4

Yay! We've just built our Benzoic Acid Molecule!